


3.1 Collection, assortment and
identification
Investigation on germplasm varieties is one of the most
important part in the maintenance of silkworm germplasm resource, only based on
these basic research results that the germplasm could be effectively utilized in
silkworm breeding and genetic research. Collection, assortment and
identification are usually done in the early stage, and maintenance was strictly
followed the specific rules in China, that is:
- Those newly collected varieties should be separately raised during the
first two generations when newly introduced, and could be raised in the same
room of the existed stocks only after confirmed that they are pebrine disease
free.
- The uniqueness of each variety is maintained only through systematic and
scientific manipulation of the germplasm. Otherwise, it will degenerate to
loose its original characteristics in genetic markers or in its performance
of economic characters, or it will become lost because of drastic reduction
or fertility to the extent that it is no longer able to produce viable offspring.
- Generally, rearing once a year in the congenial season for monovoltine
and bvoltine races, two or three times for diapaused multivoltine, nonstop
rearing for non-diapaused multivoltine.
- In order to prevent inbreeding depression and maintain the specific properties,
the newly hatched larvae should be collected from at least 20 layings: cut
one corner of eggs from each of the 20 layings, and stitch them together,
brush 1500-2000 newly hatched larvae in the form of mixed rearing.
- Strictly maintain the purity of the variety in each stage of egg incubation,
larvae rearing, mounting, cocoon collection, moth emergence, and egg production.
In any case, if the abnormal character which is different from the original
record, delete it, or separate if it could be a new mutation.
- Duplicate set of eggs should be produced so as to prevent the distinction
of the stock in case of unsuccessful reproduction due to some unexpected circumstance.
Investigations on the silkworm germplasm resources are mainly
including following aspects:
3.2 Records of biological features and
characteristics
- Race, voltinism, moltinism;
- Eggs: shape, color, shell color, coherence;
- Larvae: taxis, eating habit, marker, body shape, color; size;
- Cocoon: size, shape, color, wrinkle;
- Moth: size, color, mating habit, egg laying habits, laid egg number;
- Except the word records, photocopies of egg, larvae, cocoon, pupa moth should
be maintained.
3.3 Investigation of economic
characteristics
- Characteristics investigated during rearing: development uniformity, duration,
survival rate of larvae, larva-pupa survival rate, rate of dead worm cocoons,
cocoon weight, cocoon shell weight, cocoon shell ratio.
- Characteristics should be investigated by special inspection machine: filament
weight, filament length and size, reelability, neatness, strength and elongation,
cohesion, degumming loss percentage of cocoon shell, lousiness and etc..
3.4 Identification of resistant
ability
Resistant ability is generally investigated under the
artificially set environment condition to identify the specific property in
different adverse conditions. For example:
- To identify the tolerant ability to high temperature and humidity, larvae
reared under the environment at the temperature 30-32°C, and RH over
90%;
- To identify the resistant ability to certain disease, inoculation of a specific
bacteria or virus.
- To identify the tolerant ability to certain insecticide or fluoride pollution,
by oral administration of miro-concentration of insecticide or fluoride.
3.5 Genetic analysis
- Gene behavior and gene location of the new mutation. There are high rate
spontaneous mutation in silkworm, to identify and study its function, inheritance
and location, enrich and modify the silkworm gene linkage map, are one of
the important aspect of germplasm maintenance.
- Biochemical genetic research. To study isoenzyme pattern of hemolymph,
skin, midgut and silk gland, protein type of blood and digestion juice in
different development stage are also widely concerned in germpalsm investigation.
- Molecular linkage mapping by bio-technique such as RAPD, RELP, SADF, AFLP.
This is the newly developed field and also widely recognized in silkworm germplasm
research, will be the hot spot in the near future.
Methodology of silkworm germplasm maintenance and research
adopted by National Sericulture Institute of China Agriculture Science Academy
refer to Annex 2.
3.5.1 Long term cold storage techniques
for diapaused polyvoltine germplasm strains
The polyvoltine varieties have to be raised for many times a
year in the germplasm maintenance, which not only created more works for the
maintenance but also resulted in fast degeneration for more rearing generation
in a year. The new cold storage technique had made it possible to rear diapaused
polyvoltine varieties once a year. That is to rear the polyvoltine varieties in
summer season, laid eggs (August 1-5) are preserved under 25°C for 35-40
days, then under 20°C during September to October, after that, the
temperature gradually decreased to the natural temperature until 5°C, egg
bath takes place in mid-December, and then be transferred into cold storage
under 2.5 °C or 0°C, take out for intermediate temperature under
10°C-15°C for 7 days in late April, and put back to the cold storage
under 2.5 °C until mid June before incubation starts. Silkworm will be
reared again in summer season. Experiments had shown that there were no
significance difference between the new method and normal rearing cycle in the
rearing performance such as hatching rate, cocoon weight, cocoon shell weight,
shell ratio, survival rate of larva-pupa, and voltinism character.
3.5.2 Maintenance of lethal
genes
There are many lethal genes discovered in silkworm, and these
genes require special techniques to be maintained in the germplasm resource. The
small egg mutation sm, when the female moth is an homozygous sm,
it lays 100% small eggs which are only half of the normal size and normal
embryo cannot be developed and become dead eggs. Usually, to keep this gene,
rear 5-10 layings of heterozygous sm separately, and select the layings
with a ratio small egg: normal egg 1:1 as the seed layings, and 5-10 laying are
reared separately again at the next generation. This will cost much more man
power and material for maintenance, and moreover, very hard to be used for
linkage analysis as marker gene. Now, take the dominant gene of larval zebra
marking, which is in the same linkage as the marker, make the Ze
+/+sm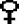 × +
sm/+sm
× +
sm/+sm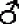 system to keep the
sm gene, just use zebra marking
system to keep the
sm gene, just use zebra marking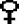 × non-zebra marking
× non-zebra marking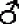 , can produce
the heterozygous sm eggs, therefore, only one batch of mixed rearing is
required for the next generation. Here, the complete linkage of female silkworm
was used, it is easy to distinguish that the zebra marking individual is
heterozygous sm, to produce normal eggs, and non-zebra marking individual
is homozygous sm, to produce small eggs. This principal can also be used
in the maintenance of second small egg sm-2, kidney shape egg ki,
shriveled egg Grc01, by the system of ++/ch
sm-2
, can produce
the heterozygous sm eggs, therefore, only one batch of mixed rearing is
required for the next generation. Here, the complete linkage of female silkworm
was used, it is easy to distinguish that the zebra marking individual is
heterozygous sm, to produce normal eggs, and non-zebra marking individual
is homozygous sm, to produce small eggs. This principal can also be used
in the maintenance of second small egg sm-2, kidney shape egg ki,
shriveled egg Grc01, by the system of ++/ch
sm-2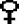 × ch sm-2/ ch
sm-2
× ch sm-2/ ch
sm-2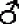 , E
+/+ki
, E
+/+ki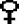 × + ki/+
ki
× + ki/+
ki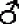 , +
+/pGrc01
, +
+/pGrc01 ×
pGrc01/pGrc01
×
pGrc01/pGrc01 .
Other lethal genes like larval lethal gene l-nl, small non-molting larva
nm-d, white dead egg al, stinky larva sku etc. can also
make similar system to be maintained in a much easier method.
.
Other lethal genes like larval lethal gene l-nl, small non-molting larva
nm-d, white dead egg al, stinky larva sku etc. can also
make similar system to be maintained in a much easier method.



 × +
sm/+sm
× +
sm/+sm system to keep the
sm gene, just use zebra marking
system to keep the
sm gene, just use zebra marking × non-zebra marking
× non-zebra marking , can produce
the heterozygous sm eggs, therefore, only one batch of mixed rearing is
required for the next generation. Here, the complete linkage of female silkworm
was used, it is easy to distinguish that the zebra marking individual is
heterozygous sm, to produce normal eggs, and non-zebra marking individual
is homozygous sm, to produce small eggs. This principal can also be used
in the maintenance of second small egg sm-2, kidney shape egg ki,
shriveled egg Grc01, by the system of ++/ch
sm-2
, can produce
the heterozygous sm eggs, therefore, only one batch of mixed rearing is
required for the next generation. Here, the complete linkage of female silkworm
was used, it is easy to distinguish that the zebra marking individual is
heterozygous sm, to produce normal eggs, and non-zebra marking individual
is homozygous sm, to produce small eggs. This principal can also be used
in the maintenance of second small egg sm-2, kidney shape egg ki,
shriveled egg Grc01, by the system of ++/ch
sm-2 × ch sm-2/ ch
sm-2
× ch sm-2/ ch
sm-2 , E
+/+ki
, E
+/+ki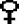 × + ki/+
ki
× + ki/+
ki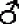 , +
+/pGrc01
, +
+/pGrc01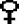 ×
pGrc01/pGrc01
×
pGrc01/pGrc01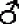 .
Other lethal genes like larval lethal gene l-nl, small non-molting larva
nm-d, white dead egg al, stinky larva sku etc. can also
make similar system to be maintained in a much easier method.
.
Other lethal genes like larval lethal gene l-nl, small non-molting larva
nm-d, white dead egg al, stinky larva sku etc. can also
make similar system to be maintained in a much easier method.