11.1 Description
11.2 Calculation of Fish,
Ice and Water Quantities
The use of insulated containers for stowage and transport of fish in iced sea water offers several advantages over traditional boxed and iced practises. It is particularly suited to small pelagic species that are not normally gutted at sea but which are caught in bulk and require rapid chilling because of their high fat content.
Advantages:
Disadvantages:
The following specification relates to a container developed for use in the UK for stowage and handling of herring and is shown in Figure 40. For similar use in tropical areas the insulation thickness should be increased to a minimum of 75 mm.
| Container dimensions: | 1 170 mm wide x 1320 mm deep x 1950 mm high |
| Insulation: | 50 mm rigid foam polyurethane |
| Construction: | Aluminium inner skin with GRP outer skin; hinged lid at top and drain at bottom |
| Capacity: | 2.1 m� |
| Empty weight: | 175 kg |
| Aeration: | By means of an aluminium lance. built into the container with coupling at top to manifold |
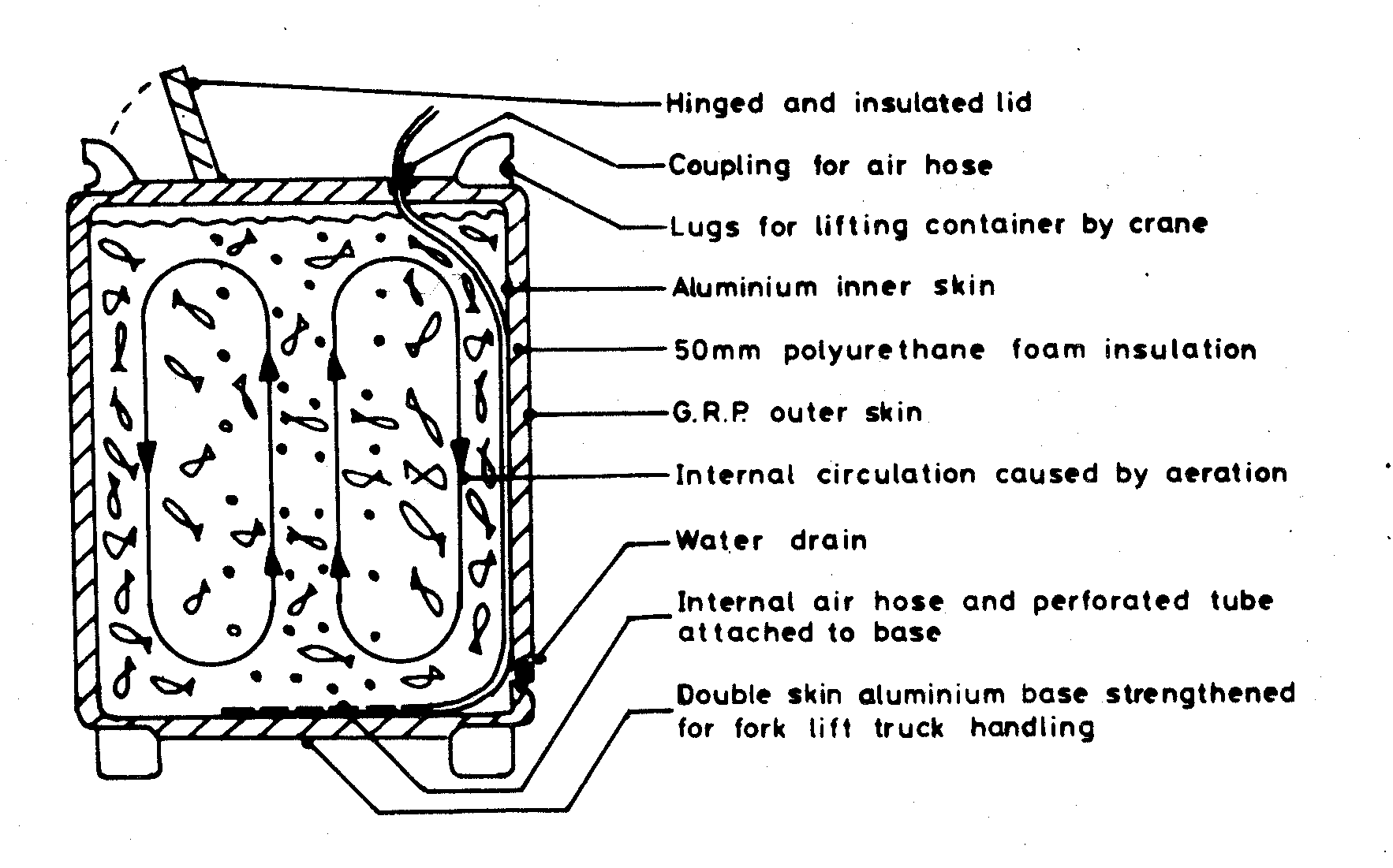
Figure 40. Chilled sea water container
For any given container .the ice:water:fish ratios can be calculated from a knowledge of ambient temperature. sea water temperature, the insulative properties of the container and the period of holding. The requirement of the ice is to cool the sea water and fish to 0�C, to counter the heat input from aeration and to counter the heat gain through the container.
The requirement for water is to provide a medium that will enable rapid heat transfer from the fish and to hold the fish such that they are not subject to crushing.
Table 16 (Section 6.2) shows the weight of ice required to cool, one kilogramme of fish or water to zero degrees centigrade. over a range of temperatures. It is assumed for the purposes of estimation that the specific heats are equal. Table 23 shows the weight of ice required to offset the heat gain through the container previously specified. It assumes a figure of 0.085 kg of ice loss per degree centigrade above zero per hour based on trials with the container.
Table 23 Ice required for each day of stowage to counter heat gain through the walls of the container specified
Ambient temperature |
kg of ice/24h |
35 |
72 |
Example calculation
The following example calculation of ice:water:fish ratios assumes ambient temperature of 20�C. sea water temperature of 15�C. a holding period of three days and a container as previously specified.
Assume 1/5 volume of container for water
0.2 x 2.1 = 0.42 m�.
Ice required to cool 0.42 m� of sea water at 15�C from Table 16
=420 x 0.17 kg .71.4 kg
which occupies a volume of 0.08 m� according to Table 3.
Ice required to counter heat gain through container for 3 days from Table 23
=123 kg
which occupies a volume of 0.13 m�.
For purposes of estimation the heat input by aeration will be assumed negligible.
Volume of remaining space within container
Vr = 2.1 - 0.42 - 0.08 - 0.13 = 1.47 m�.
This volume will be the volume of fish plus the volume of ice required to cool the fish from 15�C to 0�C.
From Table 16 the ratio of fish:ice is approximately 1:0.17
volume of fish = 1.47 x 1/1.17 = 1.26 m�. and
volume of ice = 1.47 x 0.17/1.17 = 0.21 m�.
Total volume water = 0.42 m�
Total volume ice = 0.08 + 0.13 + 0.21 = 0.42 m�
Total volume fish = 1.26 m�
From this follows:
Ratio fish:ice:water = 3:1:1 (by volume).
For higher conditions of ambience and sea water temperatures the above calculation can lead to unacceptably low carrying capacities and in these circumstances what is done in practice is to put in less ice and water initially than calculated but to later drain off some of the water and re-ice. It is important when doing this not to drain off all the water or the fish will no longer be floating in an ice water mix but subject to its dead weight
which can lead to damage by crushing.