Estimations of potential acidity by peroxide oxidation and total pyrite by sulphur
and iron measurements (Table 15) indicate the magnitude of total acid production which
might take place, but indicate relatively little about rates of reaction. The
consultants did a series of short-term leaching experiments (Table 16) using dike
soil and sediment, held on filter paper in large glass funnels. Distilled water or
brackish stream water was poured through ground dry soil or sediment and collected
in beakers. Distilled water leached most of the readily available acid from dike soil
with a quantity of water comparable to the sediment weight. Thus, a second leach
removed substantially less acid than the first. The yield of acid was about 0.4 meq
per 100 g of dry dike soil, or about 4 percent of the potential acidity measured by
peroxide oxidation. Stream water leached a much smaller quantity of acid from
freshly collected wet sediments (<1 percent of the total available). Distilled water
leach of ash produced a strongly basic solution (@pH 9), but the titration alkalinity
of the leach solution was only 1.7 meq/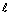 . Thus, a mixture of equal volumes of ash
leach and dike soil distilled water leach was still fairly acidic (pH 4.8). The
total base-yielding potential of the ash is probably comparable to the values obtained
from the short-term leaching experiment, whereas the total acidity potential of dike
soil is more than an order of magnitude greater than from the short-term leaching
experiments. Leaching of dike soil with distilled water which had been used
previously to leach ash yielded an even greater quantity of acid (alkalinity = -10.6
meq/
. Thus, a mixture of equal volumes of ash
leach and dike soil distilled water leach was still fairly acidic (pH 4.8). The
total base-yielding potential of the ash is probably comparable to the values obtained
from the short-term leaching experiment, whereas the total acidity potential of dike
soil is more than an order of magnitude greater than from the short-term leaching
experiments. Leaching of dike soil with distilled water which had been used
previously to leach ash yielded an even greater quantity of acid (alkalinity = -10.6
meq/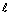 ) than with distilled water alone (alkalinity = -6.2 meq/
) than with distilled water alone (alkalinity = -6.2 meq/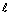 ).
).
Water collected less than half an hour after a rain shower from standing puddles
on the dikes had even higher total acid (alkalinities of -12 and -15 meq/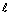 ) than from
the consultants' laboratory distilled water leaching experiments with dike soil
(Table 17). Surface water from Pond 20 after the same rain shower had an alkalinity
of +1.30 meq/
) than from
the consultants' laboratory distilled water leaching experiments with dike soil
(Table 17). Surface water from Pond 20 after the same rain shower had an alkalinity
of +1.30 meq/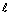 , whereas alkalinity in this pond five days earlier was +1.71 meq/
, whereas alkalinity in this pond five days earlier was +1.71 meq/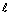 .
.
Brackish water (salinity = 25‰) from ponds at a new private aquaculture project
in southeastern Johore State (Santee Estates) also had significant loss of alkalinity
(Table 17) from expected values (1.6 meq/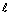 ), with essentially no alkalinity in a pond
left without water exchange for six months.
), with essentially no alkalinity in a pond
left without water exchange for six months.