
FUNDS-IN-TRUST
FAO/GCP/CAF/007/NET
 | NETHERLANDS FUNDS-IN-TRUST FAO/GCP/CAF/007/NET |
A report prepared for the Hatchery Production and Research Centre Project
by
E.H. Eding
(Consultant)
This report was prepared during the course of the project identified on the title page. The conclusions and recommendations given in the report are those considered appropriate at the time of its preparation. They may be modified in the light of further knowledge gained at subsequent stages of the project.
The designations employed and the presentation of the material in this document do not imply the expression of any opinion whatsoever on the part of the United Nations or the Food and Agriculture Organization of the United Nations concerning the legal or constitutional status of any country, territory or sea area, or concerning the delimitation of frontiers.
FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1984
Hyperlinks to non-FAO Internet sites do not imply any official endorsement of or responsibility for the opinions, ideas, data or products presented at these locations, or guarantee the validity of the information provided. The sole purpose of links to non-FAO sites is to indicate further information available on related topics.
This electronic document has been scanned using optical character recognition (OCR) software. FAO declines all responsibility for any discrepancies that may exist between the present document and its original printed version.
2. NURSERY OPERATIONS FOR THE PRODUCTION OF LARVAE
2.1.1 Introduction
2.1.2 Feeding of the broodstock
2.1.3 Water supply
2.1.4 Size of the broodstock
2.2 Artificial Reproduction
2.3 Incubation of Large Quantities of Eggs
2.3.1 Introduction
2.3.2 Experiments
2.3.3 Results
2.3.4 Conclusions and recommendations
2.4 Artemia Hatching
2.5 Artemia-fed Larvae
2.6 Larvae Fed with Dry Pelleted Food
2.7 Cost of the Larvae Production on the Basis of Food Consumption
2.8 Scaling-up Larvae Production
3. HANDLING OF EGGS AND LARVAE AND PREVENTION OF DISEASES
3.1 Introduction
3.2 Water Quality Improvement
3.2.1 Oxygen saturation of water entering the hatchery
3.2.2 Oxygen level in the nursery tanks
3.2.3 Removal of sand and organic material
3.3 Improvements in Farming Techniques
3.3.1 Improvement of light intensity
3.3.2 Suggested improvements for daily cleaning of the
nursery tanks
3.3.3 Installation of an air pump
3.3.4 Elimination of stress-causing factors
3.3.5 Fish storage in the hatchery
3.3.6 Prevention of waste caused by uneaten food
3.3.7 A cleaning schedule for nursery tanks
Appendix 1: SCHEDULE OF ACTIVITIES
Appendix 3: GROUND PLAN OF THE HATCHERY
Appendix 4: GROUND PLAN OF THE SETTLING TANK
LIST OF TABLES
1. WATER SUPPLY CALCULATED FOR ADULT FISH IN 1/min
2. FOOD REQUIREMENTS PER TEMPERATURE AND PER WATER SUPPLY
3. PROPHYLACTIC TREATMENTS DURING HATCHING OF EGGS AND LARVAL REARING
4. EFFECT OF MALACHITE GREEN CONCENTRATIONS ON THE HATCHING RESULTS BY
CONTINUOUS EXPOSURE
LIST OF FIGURES
1. Californian rectangular tank
3. Installation position of the light
4. Water inlet of the Californian tank
5. Optimal nursery tank for dry pellet fed larvae
6. Installation scheme for the air pump and air pipes
The Hatchery Production and Research Centre project GCP/CAF/007/NET located at La Landjia, Bangui, is financed by the Netherlands Government and executed by FAO.
The project has the following immediate objectives (Blessich et al., 1983):
The establishment of a hatchery for catfish culture (C. lazera = C. gariepinus) at the National Fish Culture Centre, Bangui-La Landjia, Central African Republic.
The adaptation and optimization of the technology for the controlled propagation and semi-intensive culture of C. lazera under the prevailing conditions in Central Africa.
The development of a diet, based on locally available agricultural by-products which will be suitable to feed C. lazera either as fingerlings or during grow-out.
The establishment of a policy for the distribution of C. lazera fingerlings to the private fishfarmers.
The training of the local staff in these technologies for controlled propagation of C. lazera and its perspectives for fish culture development.
Objectives 2 and 3 should be realized when extension is started in order to ensure a continuous supply of fingerlings. To achieve this aim the conditions for farming C. lazera should be optimal (i.e., water quality and quantity, feed quality, feeding level, reproduction techniques, disease prevention and production systems).
This report summarizes the consultant's findings and recommendations during the period 28 November–22 December 1983, concerning the following terms of reference:
établir une technique d'incubation en grandes quantités des oeufs de Clarias et résoudre les problèmes d'adhésion des oeufs.
suivre les élevages de grossissement d'alevins et en particulier réaliser un élevage complet de l'éclosion jusqu'au stade de fingerling (1-1, 5 g).
assister au développement d'un management pour l'élevage de fingerlings de Clarias.
mettre au point un système d'hygiène et des traitements.
The African catfish C. lazera does not reproduce in captivity. An artificial breeding technique has been developed in Wageningen (Netherlands) to control the propagation of this fish. At La Landjia station this procedure takes place every six weeks, under the supervision of Mr Ndodet (counterpart of Dr Janssen). The broostock in the hatchery which is used for this technique originates partly from fish reared from the egg stage in the hatchery and from fish captured in ponds at an age of 9–12 months.
The farming history of the two broodstocks results in an elimination of the annual reproductive cycle of the hatchery-conditioned broodstock in contrast to the pond-conditioned broodstock which retains the annual natural reproductive cycle.
According to Dr Janssen it is also possible to eliminate the annual natural reproductive cycle of the pond broodstock after ± 1 year of hatchery-conditioning.
The hatchery-conditioning period for broodstock coming from ponds, in order to transform them into non-cyclic fish, probably depends on the time of the year the broodstock is transferred from the ponds to the hatchery. At La Landjia station non-cyclic broodstock produce 15% of their body weight as stripped eggs, throughout the year. To keep the broodstock in this breeding condition optimal farming management is required.
A feeding level of 1% of the body weight should be used when the broodfish reaches 300 g or more. The food should contain a crude protein level between 30 and 55% and a high energy content.
An automatic feeder must be used instead of handfeeding in order to avoid stress. Stress occurs when the covers are taken from the tanks to feed the fish by hand which will result in a lower food intake.
Especially wild fish from the ponds which are conditioned in the hatchery will react in this way. A higher water supply during the first conditioning months may possibly prevent accumulation of uneaten food particles in the tank.
To calculate the water supply needed for the broodstock it was assumed that:
adult fish take 50% of their oxygen requirement from the air
a feeding level of 1% is optimal
the oxygen level of the water is 8 ppm
the oxygen level in the tank does not fall below 3 ppm.
To calculate the water supply, the formula of Willoughby (1968) is used and the above assumptions are substituted in this formula.
In principle the volume of the hatching tank is not important in this calculation.
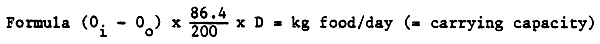
Oi = O2 grade in mg/1 of the water inlet
Oo = O2 grade in mg/1 of the water outlet
86.4 = m3 water supply per day at a rate of 1 l/sec
200 = grammes O2 necessary to digest 1 kg of food (based on experiments with trout)
D = rate in 1/sec
This formula is only used as guide to prepare a water supply table for practical use (Table 1).
| kg of fish/tank | l/min |
| 50–70 | 10a |
| 90 | 13 |
| 110 | 15 |
| 130 | 18 |
| 150 | 21 |
a A minimum of 10 l/min is calculated to obtain at least one tank renewal per hour
The size of the female broodstock depends on the following:
The weight of the fish. A suitable weight to manipulate broodstock at the time of hypophysation and stripping is 500 g.
The age of the fish. The broodstock should consist of 9–12 month old fish from the ponds. Nursing of larvae to broodstock in ponds simplifies hatchery management as only two age groups are kept in the hatchery, adult broodstock and fingerlings up to 1 g.
The 9–12 month old broodstock selected from the ponds has to be conditioned in the hatchery for one year in order to eliminate the annual natural reproductive cycle, after which it can replace the actual broodstock. When all broodstock has been used for one year the maximum age of the broodstock will be ± 3 years.
The larvae production capacity. When a simple production system is used in the hatchery a total production of 216 000 larvae per cycle is possible. To produce this amount of larvae about 18 females of 500 g have to be hypophysized (Section 2.2).
The length of the nursery period for larvae up to a weight of 1 g. Normally the length of this period depends on the temperature and varies between 6 and 8 weeks. Every 6–8 weeks artificial breeding techniques are carried out. In Wageningen it is possible to use the same broodstock every 6 weeks with a water temperature of 25°C and a feeding level of 1.5%. At La Landjia Station this will not always be possible because in December and January temperatures are below 25°C and total development of the gonads will take longer. For this reason it is necessary to keep a larger stock.
Diseases, failures in reproduction technique or during the larvae nursery period. Broodstock diseases and mortality can be caused by:
injection of females for artificially induced breeding. After injection, ulcers can develop if contaminated needles and fluid are used. The infection can cause mortality on the hypophysized broodstock.
after stripping. If the broodstock is not stripped carefully, gonads can break and the kidneys can be damaged. Also, internal bleeding can occur.
poor farming techniques (e.g., rough handling of fish and inappropriate feed).
Failures in reproduction techniques can occur when:
a wrong concentration CPS (Carp Pituitary Suspension) has been used to induce artificial reproduction;
a wrong latency period (time between hypophysation and stripping) has been used;
When the injected females are highly stressed during the latency time (practical experience).
Failures in larvae production can be caused by wrong farming methods and diseases.
Taking into account the above, the following broodstock should be accommodated in the hatchery:
Broodstock tank 191 ± 200 male conditioning stock
Broodstock tank 29 ± 200 female conditioning stock
Broodstock tank 21 ± 200 male actual broodstock for propagation
Broodstock tank 22 ± 200 female actual broodstock for propagation
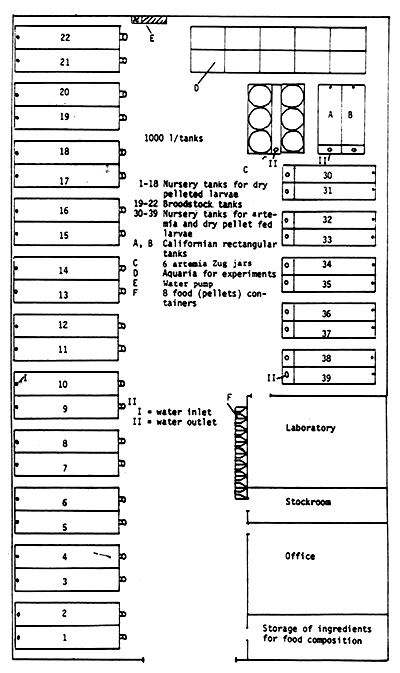
1 A groundplan of the hatchery can be found in Appendix 3.
A reserve broodstock should be maintained in a pond in case of emergency (200 males and 200 females).
The number of females which should be hypophysized for the production of larvae depends on the following characteristics.
The stripping percentage. This is defined as weight of stripped eggs/body weight × 100. At La Landjia this percentage was 15% during the last two years.
This stripping percentage was reached when females were selected according to the development of their belly.
The consultant calculated within 10% to be sure that the egg production per fish was not overestimated.
The number of eggs per gramme. The number varies from 700 to 800/g.
The hatching percentage. This is defined as the percentage of fertilized eggs which develop into normal larvae. At La Landjia Station a hatching-percentage of 80 is normal. The consultant used a hatching percentage of 50 in his calculations to be sure that in the case of slight failures sufficient larvae are still produced.
The production capacity of the hatchery. A production level of 216 000 fingerlings per 6–8 weeks is possible. This production level is calculated by the consultant based on the following assumptions:
larvae are totally dependent on the oxygen in the water
the oxygen level of the water inlet is 8 ppm
a minimum oxygen level of 3 ppm is necessary to maintain growth
the water supply available for the hatchery is estimated at 10 l/sec
According to Dr Janssen the water supply depends on the time of the year and varies between 10 and 20 l/sec. In 1982, in one of the driest seasons ever, no water was available and larvae production was not possible. To prevent water shortage in the future the possibilities of using ground water should be investigated.
a feeding level of 10% is applied at the end of the nursery period when the fingerlings reach a weight of 1 g
the survival rate. Survival rate after hatching up to a weight of 1 g is estimated at 70%.
Substitution of the above assumptions in the Willoughby formula results in a total possible food supply of 21.6 kg at the end of the nursery period. With this amount of food 216 kg of fish can be fed at a feeding level of 10%. Thus the total fingerling production capacity at the end of one production cycle (eggs up to 1 g) will be 216 000 fingerlings.
Based on the above, the number of females needed for hypophysation can be calculated by using the following procedure:
| Production capacity hatchery | 216 000 | fingerlings |
| Survival rate (70%) 100/70 × 216 000 | 308 571 | larvae |
| Hatching percentage (50%) 100/50 × 308 571 | 617 142 | eggs |
| Number of eggs/g (700) 617 142/700 | 881.6 | g of eggs |
| Stripping percentage (10%) = 100 g eggs/kg fish 881.6/100 | 8.82 | kg fish |
| Number of fish necessary (500 g) 8.82/500 | 18 | females |
The females which are selected on the basis of the development of the belly, are injected with 4 mg carp pituitary suspension per 1 kg body weight. After hypophysation the females are put together in one 1 000-1 tank. Low densities of hypophysized fish will cause fighting which results in stress; and this will impair egg quality.
The latency period depends on the actual average water temperature during the conditioning of the broodfish.
The milt is collected by sacrificing a male. The milt is stored in a 0.9% NaCl solution and verified on motility under the microscope by activating the sperms with tap water.
The stripped eggs are mixed with milt and fertilized.
Two methods can be used to incubate eggs of the catfish C. lazera.
Method I:
Eggs are incubated in a Californian rectangular tank (Fig. 1). Each tank contains four trays on which the eggs are spread in a single layer. The purpose of the trays is to lead the waterflow along the eggs to assure a sufficient oxygen supply.
In practice a maximum of 200 g of fertilized eggs can be incubated per Californian tank. This method is used at the hatchery at La Landjia.
Method II:
Eggs are incubated in Zug jars (Fig. 2) after treatment with the enzyme maxatase (Verreth, 1982). Fertilized eggs are treated with this enzyme in order to remove the sticky coat which is a prerequisite for the incubation of eggs in Zug jars.
The consultant examined and compared the advantages and disadvantages of both methods in order to decide which could be introduced at La Landjia.
Method I:
Two Californian rectangular tanks with four trays each were prepared (Fig. 1). By means of an air pump a recirculating system was established in one of the tanks. This was done to investiage the possibility of using boiled water to prevent contamination with pathogens and to extend exposure time during treatments. The other tank was a flow-through system. In each tank 200 g of fertilized eggs were incubated. This is equivalent to 140 000 eggs.
Method IIa:
For this method one Zug jar was prepared as the incubation unit (Fig. 2). A condition for the incubation of eggs in Zug jars is the absence of stickiness. The eggs of C. lazera have a coat which swells and becomes sticky shortly after water is added. The enzyme maxatase can eliminate this problem if added to the eggs before stickiness occurs.
The following procedure has been developed in Wageningen:
one minute after fertilization 500 ml maxatase 175 ppm should be added to the eggs in a bowl
the total exposure time of maxatase to the eggs should be 6 min
after 2 and 4 min the maxatase solution should be replaced by the same quantity and concentration of maxatase
after the total exposure time of 6 min the maxatase is removed. The eggs are subsequently washed with tapwater for 2 min and then placed in the Zug jar.
The adjusted waterflow was 1–2 l/min and was increased at the end of the incubation period. The average water temperature was 24°C. The influence of maxatase and waterflow in the Zug jar (mechanical damage) was investigated. A control group was formed by taking two fertilized egg samples just before maxatase was added. Both samples were incubated in Petri dishes with stagnant water. The influence of maxatase and waterflow was investigated by taking two egg samples from the Zug jars just before hatching. Hatching percentages in both Petri dishes were calculated.
Method IIb:
The same as IIa, except that there was no renewal of the maxatase solution after 2 and 4 min. The consultant also increased the average temperature of the water to 30°C in order to obtain a better activity of the maxatase. Directly after the eggs were washed with tapwater they were treated with 2.5 ppm malachite green during 5 min to prevent fungus development.
Method I:
Hatching percentage in both tanks was about 80%. In the flow-through system a thin layer of sand and organic material settled on the bottom of the tank and partly on the eggs. This was caused by rain during the night which resulted in dark-coloured water in the hatchery.
Normal larvae production per incubation tank was calculated at:
| (g) | (eggs) | (hatching%) | ||
| 200 | × | 700 | × | (0.80) = ± 112 000 larvae |
The following comments are made:
Advantages
The hatching percentages are equal to those obtained in Wageningen.
The method is simple.
The incubation tank can be made recirculating by the installation of a simple air/water pump.
The use of such a system has the following advantages.
Prevention of contamination with pathogens using boiled water to incubate the eggs. In this case eggs should be treated with Wescodyne 25 ppm to prevent vertical contamination (broodfish eggs).
A longer exposure time by prophylactic treatments.
Settlement of sand and organic materials is not possible.
Disadvantages
Requires a lot of space in comparison to the Zug jar method.
Requires a lot of labour at the moment of hatching.
The number of normal larvae is difficult to estimate.
Method IIa:
Stickiness of the eggs occurred after 2 min of maxatase treatment. Groups of 3–5 eggs stuck together. This situation continued until they were incubated in the Zug jar. Within 5–10 min all the eggs stuck together. Just before hatching (± 29 h after fertilization) stickiness had partially disappeared. The following hatching results were obtained:
| Control group | Zug jar incubated eggs | |
| Percentage of normal larvae | 40a | 30 |
Method IIb:
The same results were obtained as in Method IIa, except that stickiness occurred just after the eggs were incubated in the Zug jar. This stickiness control procedure gave the following hatching results:
| Control group | Zug jar incubated eggs | |
| Percentage of normal larvae | ± 80 | ± 40–50 |
2.3.4.1 Incubation in Californian rectangular tanks
The incubation of eggs in Californian rectangular tanks is recommended because high hatching percentages can be obtained and prophylactic treatments can be easily applied.
Disadvantages of this method are the sticky coats and the dead eggs which remain in the tank after the egg trays have been removed. The consultant observed fungus development on this waste. To prevent this, the mesh size of the wire of the egg trays should be replaced by a smaller mesh size. However, this results in the problem that the diameter of the mesh size is too small for the larvae to swim through. This can be solved by making holes in the bottom corners of the trays (i.e., where the larvae gather after hatching). These holes should be closed by plugs at the moment the eggs are incubated in order to prevent “egg-contamination” of the Californian tank. Just before hatching the plugs can be removed. The plugs should be replaced when all larvae swim around in the tank. Subsequently the egg trays should be removed. The Californian tank now functions as a nursery tank in which larvae are fed with artemia. This should start two or three days after yolk-sac absorption.
At La Landjia Station there are two Californian rectangular tanks. A production of 216 000 larvae was calculated (Section 2.2). To produce this amount of larvae it is necessary to incubate 1 kg of eggs, which implies the need for 6 Californian rectangular tanks (150–200 g of eggs/tank).
It is recommended to purchase 4 Californian tanks and 16 egg trays for this purpose or to adjust four 300-1 nursery tanks.
2.3.4.2 Incubation in Zug jars (Methods IIa and IIb)
This can only be applied in the hatchery if:
the same hatching percentages (± 80%) are obtained as in Wageningen (Verreth, 1982).
it is certain that batches of maxatase have the same enzyme-concentration. As the maxatase at La Landjia was of a different batch than that in Wageningen it should be checked with the firm who sells the chemical if there is a possible difference in enzyme-activity between batches, as this could be a reason why the consultant did not succeed in eliminating the stickiness.
the effect of the water flow in the Zug jars on hatching percentages has been investigated.
a simple method for the application of prophylactic treatment has been developed.
The artemia hatching procedure starts one day before yolk-sac absorption is finished. The procedure begins with the decapsulation of artemia (Janssen, 1983). Subsequently 10 g of artemia is incubated in a 3% salt solution, which is well aerated during ± 18 hours (depending on the temperature). In this way optimal hatching percentages should be obtained.
According to Janssen,± 50 mg of artemia/1 000 larvae is necessary before dry-pellet feeding can be started. To produce 216 000 larvae of 1 g 310 000 larvae should be available at the moment artemia-feeding starts. During the first 4 days a peak of dying larvae will occur (± 10%). Thus, 280 000 larvae will be left which should be fed 14.0 kg artemia. To hatch this quantity of artemia during 14 days 6 more Zug jars are necessary.
The artemia hatching capacity at this moment consists of:
6 Zug jars of 15 l each
3 Zug jars for the decapsulation of artemia
3 Zug jars to hatch the artemia.
A maximum of 450 g artemia can thus be hatched every day. Especially at the end of the artemia-feeding period, the hatching capacity is too low.
The consultant recommends the purchase of 6 Zug jars in order to obtain an artemia hatching capacity which corresponds better to the larvae production.
Exogenous feeding starts from 2 to 4 days after hatching the larvae (this depends on the temperature). Artemia is fed four times a day at saturation level.
Feeding is stopped when the larvae gather in the dark corners and lie on the bottom of the tank. During the night the larvae were not fed resulting in hungry larvae in the morning.
At the end of the artemia-feeding period it was noticed that larvae had damaged tails (no bacteria were found). At the same time larvae were not saturated during the day because insufficient artemia was produced. It is possible that the damaged tails were caused by cannibalism, especially because there was a tendency for only the smaller fish to be damaged.
To obtain a continuous artemia supply the consultant installed 6 artemia pumps. These pumps have the following advantages:
continuous food supply
larvae can be concentrated near the water outlet by feeding there, where they find the best oxygen supply and the cleanest part of the tank
the larvae can be fed during the night.
After 2 to 4 days of artemia-feeding an estimate should be made of the number of larvae in the tank. At this age it is easier to estimate them. The larvae which are redundant can be transferred to the ponds, or are otherwise eliminated.
At the moment dry food is administered (16–23 days after hatching, depending on the temperature) the larvae should be divided over the nursery tanks.
The following nursery tanks are available at La Landjia:
18 tanks of 1 000 l contents (tanks 1–18)
10 tanks of 300 l contents (tanks 30–39) (see Appendix 3)
The number of larvae to be distributed over these tanks is calculated at 266 000 of which 20% die during this operation. Thus, an end-production of 216 000 larvae will be possible. The amount of dry food necessary to produce 216 000 larvae can be assessed as follows:
an equal mortality during the nursery period.
The amount of food necessary to produce 216 000 larvae of 1 g is:
216 000 + 25 000 = 241 000 g (± 245 kg dry food)
Two kinds of food should be used to feed the larvae:
Trouvit 00 from ± 40 mg to 150 mg
Trouvit 0 from ± 150 mg to 1 g
Automatic feeders should be used to administer the food (Section 2.1.2).
Given the information of the previous section, the minimum cost of the larvae on the basis of food consumption can be calculated.
Example: Food costs to produce 220 000 larvae
| FCFA | |
| 14 kg artemia | 155 555 |
| 42 kg salt for artemia hatching | 8 400 |
| 245 g Trouvit 00 and 0 | 745 652 |
| Total | 909 607 |
Price of one larva on the basis of food requirements 4.13 FCFA
The most important influence on the price of the larvae is Trouvit. Dr Janssen produces a larval feed at the hatchery which is much cheaper than the Trouvit food (one tenth of the Trouvit price). Nevertheless the quality of the food is lower than the quality of the Trouvit food, e.g., the taste of the food is inferior to Trouvit and the food conversion rate is estimated at 1.5.
It is not known if the hatchery-produced food can be administered directly after artemia-feeding.
It is recommended to start “replacing experiments” (of Trouvit by “hatchery food”) only when all production aspects are optimal (i.e., increasing larvae production per year in order to reduce the uncalculated costs like labour and the writing-off of the hatchery).
The nursery operations described above make possible a yearly production of 1.3 million larvae of 1 g. Increasing larvae production in the future can be realized as follows:
Using a management system with different age-classes (artemia- and dry pelet-fed larvae)
Lowering the final larvae weight. This will increase the number of larvae per production cycle and the number of production cycles per year. Therefore it is recommended to execute “survival experiments” to determine the optimal age at which larvae should be transferred to the ponds.
Making a better use of the total available water during the seasons. A greater water supply in the hatchery will increase the carrying capacity (Section 2.1.3). Thus during the rainy season more larvae should be produced per production cycle than during the dry season.
In intensive farming systems such as the hatchery at La Landjia disease prevention is a very important part of fingerling production.
Preventive measures can exist in:
water quality improvement
improvements in farming techniques
prophylactic treatment.
Findings and recommendations concerning hygiene and prevention of diseases are summarized below.
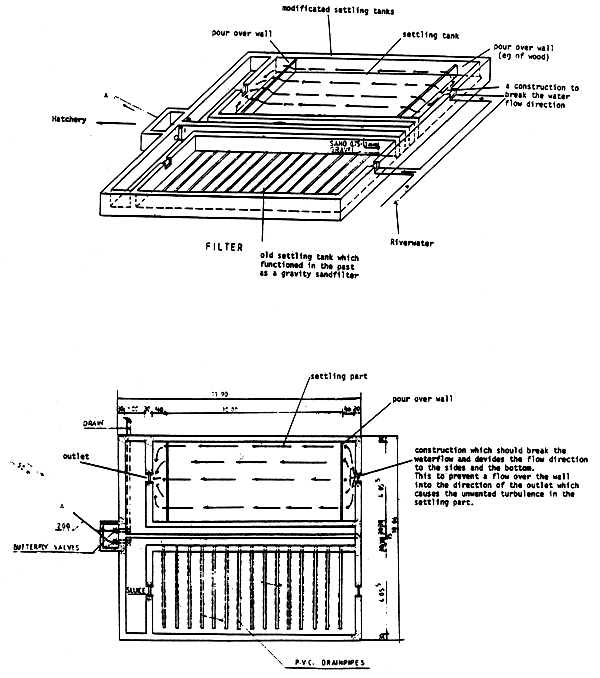
The water at La Landjia station is obtained from a small river. Before entering the hatchery the water goes through a water treatment tank. This tank was constructed to function as a gravity sand filter, but as the filter caused many problems concerning the passage and oxygenation of the water, FAO expert, Dr Janssen, removed the sand from the filter tank.
Before the removal of the sand the oxygen concentration of the outflowing water was 50% or less. This has, without any doubt, resulted in anaerobic places in the filter tank, thus impairing hygiene.
Consequently removal of the filter-sand also had a positive influence on the level of infection-pressure in the hatchery.
Oxygen saturation of the incoming water is currently ± 95%.
As it is unknown whether larvae of the African catfish (C. lazera) up to a weight of 1 g can utilize oxygen from the air, the consultant made the assumption that they are completely dependent on the oxygen in the water. To maintain growth, a minimum oxygen level of 3 ppm is therefore required.
In order to obtain a proper oxygen supply from the water the consultant developed a table for practical use in the hatchery (Table 2). The values in the table were calculated with the formula presented in Section 2.1.3 and were based on 100% saturated water at different temperatures.
| Watersupply (1/min) | 22°C | 24°C | 26°C | 28°C |
| Maximum food requirements (kg) | ||||
| 6 | 0.238 | 0.227 | 0.216 | 0.205 |
| 8 | 0.319 | 0.302 | 0.287 | 0.274 |
| 10 | 0.398 | 0.378 | 0.359 | 0.342 |
| 12 | 0.478 | 0.454 | 0.431 | 0.410 |
| 14 | 0.557 | 0.529 | 0.503 | 0.479 |
| 16 | 0.637 | 0.605 | 0.575 | 0.547 |
During major water intake by the hatchery the settling tank does not function. According to Dr Janssen the maximum water intake is 20 1/sec. With lower water intake and after rainfall, sand and organic material settle in the nursery tanks.
Although the tank was not constructed as a settling tank (Section 3.2.1), it would function better with a few modifications. Nevertheless the colloid particles which occur in the water after rainfall will not settle. The effect of this substance on the gills, and consequently on the growth, is unknown.
The following modifications are recommended in order to reduce waste in the nursery tanks:
change the water inlet such that turbulence and flow speed are minimized and a laminair flow is created;
the inflow and the outflow at the surface of the tank, both over the total width;
both tanks should be set parallel in order to achieve a longer settling time;
placing a drain in the tank to remove the slime.
These modifications are clarified in Appendix 4.
An obvious disadvantage at La Landjia Station is the lack of light intensity. It is not possible to see the fish clearly in the tanks.
Light is required for:
timely identification of diseased fish
checking the presence of food particles and/or excrements at the bottom of the tank
determining if fish are saturated after feeding.
Light also gives the possibility to concentrate larvae at the water inlet (Fig. 3).
Larvae, just after hatching, are photonegative, which means they concentrate at the darkest parts of the nursery tanks. By using covers these dark places can be created near the water inlet where the oxygen supply is optimal and waste settlement is minimal. It also facilitates clearing the outlet part of the tank because deformed and/or dead larvae and waste will remain there.
3.3.2.1 Water inlet
To improve the self-cleaning ability of the nursery tanks which contain artemia-fed larvae the consultant changed the position of the water inlet. This was done because of the slight flow in the tank which caused settlement of waste at the inflow of the tank.
The water inlet should start 20–30 cm from the beginning of the tank. This will create a calm spot with optimal water refreshment. In combination with light and dark cover (Section 3.3.1) larvae will concentrate at these calm spots (Fig. 4). This is also the area where artemia is administered by handfeeding and by automatic feeding pumps (Section 2.5).
In order to remove waste more efficiently from the nursery part of the tank it is recommended to move the small meshnet construction to another part of the tank. This place depends on the number of larvae. Air can also be used to prevent settlement of waste in the tank.
When larvae start eating dry-pellet food, automatic feeders are used. Because of the shape of the automatic feeders, food must be available at the middle of the nursery tank; therefore, at the moment of dry-pellet feeding the water inlet should be at the back of the tank (Fig. 5).
The above-mentioned measures concerning settlement of waste can also be taken for nursery tanks which contain dry pellet-fed fish.
3.3.2.2 Water outlet
A small meshnet-frame situated in front of the level regulator (water outlet) prevents the escape of larvae. The mesh can easily be clogged by excrements and uneaten food. To prevent this the following modifications can be made:
The purchase of frames with nets of different mesh sizes in order to adjust the mesh size to the larvae size. This adjustment of the mesh size is necessary because of the increasing amount of excrement and waste during the growing period.
Changing the angle of the frames in the tanks to 45°. This is the optimal angle to create a good aeration through the frame and an optimal flow through the gauze.
The precautions mentioned in A and B result in the faeces settling behind the frame. The excrement partly settles in front of the frame, but the largest amount settles behind the frame or is washed out of the nursery tank.
The excrement which settles in the nursery tank should be removed (by siphoning) twice a day by the hatchery personnel.
The water outlet pipes (level regulators) should be provided with handles. The technicians at the hatchery should be taught to use these handles and avoid touching the water in order to prevent the dispersion of pathogens from one nursery tank to another.
An air pump should be installed in the hatchery. The consultant installed an air compressor to provide oxygen. After two and a half weeks it broke down because of a too low capacity. The installation of a low-pressure air pump is recommended because of four objectives:
to increase oxygen supply in the Zug jars for better hatching results of artemia
to improve hygiene in the nursery tanks (Section 3.3.2)
to aerate the containers of the automatic artemia pumps which contain living artemia
to lengthen the time of prophylactic and therapeutic treatment for larvae up to an age of ± 21 days.
An installation scheme for the air pump is proposed in Figure 6.
In the hatchery the following stress-causing factors can be eliminated:
The pump in the back of the hatchery must be removed. With its present location, larvae, fingerlings and broodfish are stressed by vibrations every time the pump starts working. Even after three weeks the fish were not accustomed to the vibrations. Therefore, the pump should be placed outside the hatchery, and not be connected with the foundations of the hatchery.
The doors of the stockroom, laboratory and office should be provided with stoppers to prevent stress caused by slamming of the doors.
The hatchery must be frequented only by the hatchery personnel who should have a key of the hatchery door. The hatchery should be provided with an automatically closing door which can be opened without a key from the inside, but only with a key from the outside.
The consultant observed the introduction of wild fish, which were contaminated with bacteria and fungus from the ponds, into the hatchery. The fish were stored in the hatchery for sale and for pond experiments.
A second negative aspect of introducing wild fish into tanks is the non-domesticated behaviour of these fish, i.e., a continuous swimming against the walls near the water inlet. This behaviour results in an easy dispersion of pathogens. The noise caused by this behaviour had the same disturbing effect on the fish in the tanks as the slamming of doors.
The consultant suggests storing pond fish in tanks outside the hatchery. Storage of pond fish in the hatchery should be forbidden.
The sale of pond fish should be an activity which is completely separate from the hatchery activities in order to prevent contamination of larvae by the labourer. Furthermore, it disturbs hatchery labourers during work.
The hatchery at La Landjia Station is a flow-through system in which water temperatures fluctuate during the day and season because the water is taken from a small river.
Highest water temperatures occur at 16.00 h and lowest at 8.00 h. For feeding larvae, highest food intake lies somewhere between 14.00 and 22.0 h. The peak of feeding should therefore lie within this period.
Average daily temperatures measured by Dr Janssen show a seasonal temperature cycle in which the lowest temperature is reached in January (± 22/23°C) and highest in June/July (28/29°C).
For the rearing of larvae this temperature cycle implies different feeding levels during the year, which influences the length of the nursery period. Especially at the time of the year when the average temperature is decreasing and when high feeding levels are used overfeeding can happen. This can cause waste by uneaten food. To prevent this, feeding should take place at a level at which fish continuously gather under the automatic feeder. This will result in optimal food conversions when Trouvit is used.
The feeding levels thus obtained should be compared with the theoretic values. If wide deviations occur one should check if something is wrong.
In view of the fact that the lowest water temperatures are reached in the morning (see Section 3.3.6), metabolism of the fish will then be at a lower level. For this reason cleaning and fish-handling should take place at this time (to minimize stress).
The following cleaning-schedule is suggested:
Check the tank for uneaten food
Check the fish on appetite by handfeeding without disturbing them
Stop aeration of the gauze
If there is uneaten food in the tank and the fish have no appetite, check:
for dead fish in the tank. If present remove and register
fish with abnormal behaviour due to disease
the water supply in relation to food quantity
the water quality (O2, pH, NH4+/NH3)
the water temperature and the estimated feeding level of the day before
at what time feeding started and how the feed was spread on the automatic feeder
if the feed was replaced by food of another composition
if none of the above-mentioned points were at fault replace the feed by fresh feed of a high quality (Trouvit from the refrigerator)
Clean the gauze in front of the level regulator (outlet)
Increase the water supply
Remove the level regulator of the tank
Regulate the water outlet speed in relation to the size of the larvae, otherwise they will be damaged by the mesh in front of the outlet
Clean the area behind the gauze with a disinfected brush
If necessary clean the walls and the bottom of the nursery tank
Replace the level regulator and remove the most recent waste from the gauze
Adjust the calculated water supply
Restart aerating the gauze
Disinfect hands and used material before cleaning the next tank.
Every two tanks have their own bucket with disinfectant at La Landjia. The disinfectant should be checked once a day on activity and level. The level of disinfectant per bucket can become too low through evaporation, and it is thus impossible to immerse nets and other material.
Although Dr Janssen has developed a good system of prophylactic treatment (Table 3), daily exposure to antibiotics, formol and malachite green is detrimental to the health of the larvae. Constant use of antibiotics can lead to the development of resistent bacteria.
To minimize these disadvantages, experiments should be carried out in which prophylactic treatment is reduced (e.g., prophylactic treatment should be restricted to the eggs to prevent vertical contamination).
The removal of the filter sand, which improves the oxygen level and hygiene, supports the execution of such experiments.
| Treatment | Conc. (ppm) | Exposure time | Prophylactic against | ||
| (a) | eggs | Wescodyne | 25 | 5–10 min | vertical contamination |
| (b) | larvae | Malachite green | 0.05 | 30 min | fungus |
| 0–4 days | Furaltadone | 10.00 | 30 min | Flexibacter | |
| (c) | larvae | Malachite green | 0.1 | 30 min | fungus |
| 5–14 | Furaltadone | 10.00 | 30 min | Flexibacter | |
| days | Formol | 15.00 | 30 min | ectoparasites | |
| (d) | larvae | Malachite green | 0.1 | 1 h | fungus |
| 14+days | Furaltadone | 10.00 | 1 h | Flexibacter | |
| Formol | 15.00 | 1 h | ectoparasites |
Eggs are disinfected with Wescodyne as a prevention against yolk-sac oedema, and with malachite 5 ppm during 5 min against fungus. According to Dr Janssen the latter method did not give satisfactory results. For this reason the consultant experimented with different concentrations of malachite green with a continuous exposure during incubation and hatching (Table 4). The eggs were obtained from two females.
| ppm | % dead eggs | % deformed larvae | % normal larvae | occurrence of fungus on eggs |
| 0.5 | 46.7 (7.2) | 6.9 (2.8) | 46.0 (5.2) | no |
| 0.1 | 44.2 (0.1) | 8.2 (0.5) | 47.7 (0.4) | no |
| 0.05 | 50.1 (3.7) | 8.1 (1.0) | 41.8 (4.7) | no |
| 0.02 | 49.1 (1.8) | 5.0 (1.9) | 46.0 (3.8) | yes |
| 0.005 | 53.0 (9.6) | 4.8 (1.8) | 42.1 (7.2) | yes |
| control | 55.4 (4.9) | 4.6 (2.0) | 39.8 (5.7) | yes |
The table indicates that a continuous prophylactic treatment of 0.05 ppm against fungus, in an recirculation system for egg incubation, is necessary.
Three years ago Flexibacter infections occurred in the hatchery in Wageningen. In a short time many fish died. Under present Wageningen hatchery conditions disease problems are very rare and this is mainly due to a strict hygienic approach to hatchery operations.
Disease prevention for this problem exists in:
disinfection of the hatching tanks before use
disinfection of material such as nets, etc., during the nursery period
removal of fish (weighing less than 100 g) which fall on the floor or jump out of the tank
minimizing fish handling, especially of young fish
disinfection of the floor when the weight of the fish is measured
intensification of disease control for 2–4 days after handling of the fish
separation of larvae from adult fish (fish up to a weight of 100 g are sensitive to Flexibacter while larger fish are more or less immune)
a proper removal of waste and feed particles from the tank
the stopping of feeding the day before the fish are weighed. This precaution is suggested because it has been noticed in practice that under such conditions fish eat better after weighing.
Nowadays when Flexibacter infections occur, it generally ensues that one of the above-mentioned rules had not been observed.
When disease breaks out Dr Janssen uses antibiotics as therapeutic treatment (Clooramphenicol, Ampiciline, Tetracycline or Penicillin). During the consultancy there was an infection of Flexibacter columnaris in two tanks. The average weight in one of the tanks was 50 g and in the other 10 g.
In the Netherlands it is possible to eliminate this disease at this weight by eliminating the infected fish.
The consultant tried the same method at the hatchery at La Landjia Station, but without any effect. The number of dead fish per day showed that Flexibacter columnaris was more virulent at La Landjia than in the Netherlands.
There are some possible explanations for this:
Because of the continuous treatment with antibiotics previously, the immunization system of the fish works on a low level. Till now no resistance to antibiotics has been found.
Because of frequent horizontal infections in the hatchery more virulent bacteria have developed.
Farming conditions are sub-optimal.
Interaction between (a), (b) and (c).
When the hatchery is operating at full capacity two labourers should work on the larvae production (excluding counterpart) and one labourer be assigned to pond operations. At this stage one of the two hatchery labourers is engaged in both hatchery and pond activities. This often results in situations in which the larvae nursery receives too little attention. The employment of an extra labourer would make better terms of reference for each labourer possible, and result in a better management of both pond and hatchery activities.
Although some techniques were already carried out under the supervision of Mr Ndodet (counterpart to Dr Janssen), he requires training in total hatchery management. It should be done in such a way that Mr Ndodet has to propose the planning of the larvae production, Dr Janssen only assisting him if necessary. In this way he will be trained to take initiative.
The consultant also recommends the preparation of a manual for all labourers on the project. This manual should serve as a reference to explain standard procedures (i.e., hatchery and pond techniques, and food technology). For the counterpart, this manual should also contain all information on how to obtain food components, spare parts, hatchery equipment, medicines, etc. This should speed up the process of independent managing of the hatchery by the counterpart.
Considering the amount of work and experiments which still have to be carried out in the hatchery and in extension services, Dr Janssen will certainly need the assistance of either students and/or hatchery employees (UN Volunteer). As the recruitment of students is difficult at present, the latter suggestion should be considered. Without this assistance it will be difficult to fulfil the heavy work programme.
The following recommendations with regard to the hatchery activities are made:
Handfeeding (pelleted food) should be replaced by automatic feeding. The only exception is when it is necessary to check on appetite (see Section 2.1.2).
The peak feeding of broodfish and larvae should take place during the day when highest temperatures occur.
The hatchery should only be used for conditioning of broodstock and nursery of larvae up to a weight of 1 g.
The purchase of four rectangular tanks and 16 egg trays in order to make possible incubation of large quantities of eggs, or the purchase of material to modify some of the nursery tanks for this purpose (see Section 2.3.4).
The purchase of another artemia hatching unit of 6 Zug jars (see Section 2.4).
The larvae should be fed during the night with artemia pumps (see Section 2.5).
The installation of light above the nursery tanks (see Section 2.3.1).
The concentration of artemia-fed larvae near the water inlet by using the husbandry methods mentioned (see Sections 2.5, 3.3.1, 3.3.2).
The installation of a low pressure air pump in order to improve husbandry (see Section 3.3.3).
Elimination of stress-causing factors, such as the pump in the hatchery, slamming of doors, unwanted visitors (see Section 3.3.4).
Fish storage of wild pond fish in the hatchery should be forbidden (see Section 3.3.5).
Reducing prophylactic treatment. The frequency of prophylactic treatment is much too high. The effect of daily treatments with antibiotics (Furaltadone), formol and malachite on the health and consequently the growth of the fish is unknown. Experiments should be carried out in which only eggs are prophylactically treated in order to prevent vertical infections (see Section 3.4).
The employment of an African labourer for the ponds (see Section 4).
The development of a manual for hatchery and pond activities (see Section 4).
The assistance of students or a UN Volunteer to assist Dr Janssen in the heavy work programme (see Section 4).
The modification of the settling tank in order to reduce waste in the nursery tank (see Section 3.2.3).
The execution of survival experiments with different weight classes of larvae in ponds (see Section 2.8).
The determining of the feeding level (pelleted food) by means of appetite tests in order to prevent overfeeding (see Section 3.3.6).
The installation of four to six 1 000–1 tanks outside the hatchery for fish storage (see Section 3.3.5).
Blessich, D., E.H. Huisman and L.J.K. Klein. 1983 Report of the evaluation of the project GCP/CAF/007/NET. Bangui, FAO, 31 p. (unpublished) (Issued also in French: République Centrafricaine. Rapport d'évaluation (23 juin-7 juillet 1983) du Projet Production en écloserie et Centre de recherches. Rome, FAO, FAO/GCP/CAF/007/NET, 28 p.)
Janssen, H. 1983 Perfectionnement de la technique de décapsulation de cystes d'Artemia. Bangui, FED-DGIS/FAO, Note technique 20, 4 p.
Verreth, J.A. 1982 Rapport de mission au Centre piscicole de La Landjia (RCA), 3–11 juillet 1982. Wageningen (Pays-Bas), Department of Fish Culture and Inland Fisheries, Agricultural University, 12 p. (unpublished)
Willoughby, H. 1968 A method of calculating carrying capacities of hatchery troughs and ponds. Progr.Fish Cult., 30:173–4
FIG 1 Californian rectangular tank
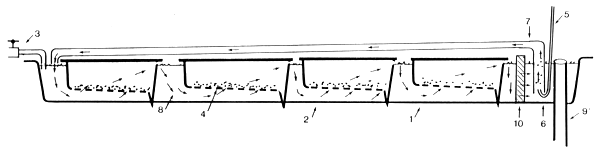
Californian rectangular tank (contents 150 l)
Egg tray
Water inlet (the tap is opened in case of the flow-through tank)
A single layer of fertilized eggs
Air
Airstone
Air/water pump (used in case of recirculation)
Waterflow direction in the tank
Water outlet (water-level regulator)
Meshnet frame
Fig 2 Zug jar
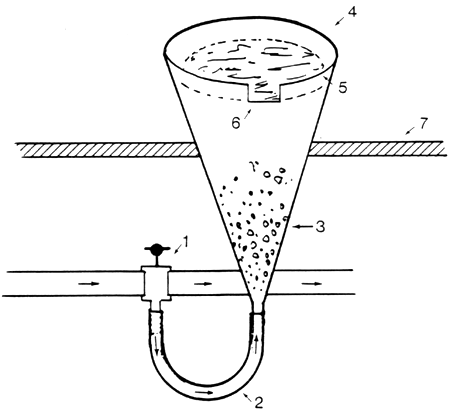
Tap
Waterflow direction
Eggs
Zug jar
Water level
Water outlet
Table in which the Zug jar is placed
FIG 3 Installation position of the light
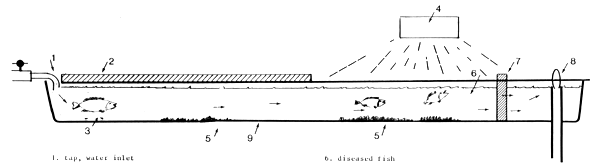
tap, water inlet
cover
concentration of healthy fish
light source
settled uneaten food and excrements
diseased fish
meshnet frame
water outlet with handle
Californian rectangular tank
FIG. 4 Water inlet of the Californian tank
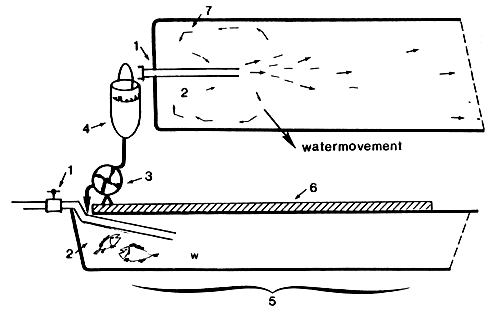
tap, water inlet
concentration of larvae
automatic artemia pump
artemia container
water inlet part
dark cover
waterflow
FIG. 5 Optimal nursery tank for dry-pellet fed larvae
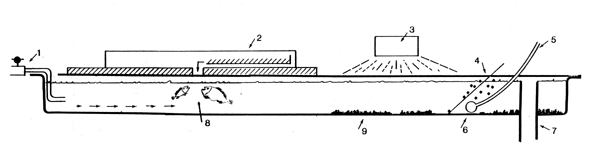
water inlet
automatic feeder
light source
meshnet frame
air
aeration pipe
water outlet
concentration of fish
waste
Fig. 6 Installation scheme for the air pump and air pipes
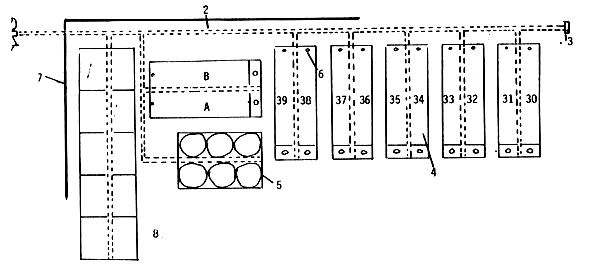
air pump
air pipes
pressure valve
nursery tanks (A, B, 30 to 39)
Zug jars
. taps
hatchery wall
aquaria for experiments
| A. | Itinerary | |
| 29.11.1983 | Departure Amsterdam | |
| 29.11.1983 | Arrival Rome | |
| 30.11.1983 | Departure Bangui | |
| 01.12.1983 | Arrival Bangui | |
| 22.12.1983 | Departure Bangui | |
| 23.12.1983 | Arrival Rome | |
| 23.12.1983 | Arrival Amsterdam | |
| B. | Persons met | |
| Mrs Bentein | Junior Professional Officer UNDP | |
| Mrs Blessich | FAO Project Operations Officer (Africa Group) | |
| Mr Deceuninck | Director Project CAF/80/002 | |
| Mr Chow | Fish Feed Technologist, ADCP, FAO Rome | |
| Mr Demoulin | FAO Associate Expert at Bouar | |
| Ministre Gipi | Ministre des Eaux, Forêts, Chasses, Pêches et du Tourisme | |
| Dr Janssen | FAO Fish Culture Expert, Project GCP/CAF/007/NET | |
| Mr Koudogbo | FAO Representative at Bangui | |
| Mr Koumis | Deputy FAO Representative at Bangui | |
| Mr Marquet | FAO Expert at La Landjia | |
| Mr Mievis | FAO Expert at La Landjia | |
| Mr Ndodet | Counterpart of Dr Janssen | |
| C. | Activities | |
| 29.11.1983 | Arrival at Rome | |
| Briefing at Fisheries Department, FAO | ||
| 30.11.1983 | Continuation of briefing | |
| Departure for Bangui | ||
| 30.11/ 22.12.1983 | Arrival at La Landjia Station to discuss terms of reference | |
| 6.12.1983 | Meeting with FAO Representative | |
| 7.12.1983 | Meeting with UN Representative (UNDP) | |
| 21.12.1983 | Meeting with FAO Deputy Representative | |
| Meeting with Representative (UNDP) | ||
| 23.12.1983 | Arrival at Rome | |
| 23.12.1983 | Arrival at Amsterdam |
| Quantity | ||
| Air pump | - tubes (55 mm external diameter) | |
| - connection pieces (55 mm internal diameter) | 10 | |
| - effervescent stones | 100 | |
| - taps | 100 | |
| - pressure valve | 1 | |
| Drift-thermometers | ||
| Pipettes (100 ml) | 15 | |
| Pipettes (50 ml) | 10 | |
| Pipettes (5 ml) | 20 | |
| Measure retort (3 1) | 4 | |
| Measure retort (5 1) | 4 | |
| Suckers | 1 000 | |
| Perforated rustproof steel to adjust the hatching tank | ||
| for incubation of eggs (180 × 40 cm) | 4 | |
| Fuse for Olympus BHB-F-3 1.5 A | 20 | |
| Mettler AC 100 | 20 | |
| PC 4000 | 20 | |
| Gauze for the racks of the hatching tanks (different diameters) | ||
| Tubes for artemia pumps | 8 | |
| Thermographer | 2 | |
| Stoppers to slow down closing of the doors | 4 | |
| Rena air pumps | 10 | |
| Repair of the oxygenmeter | ||
| - gauze of oxygen electrode | ||
| - electrode brush | ||
| - tablets for zero solution | 100 | |
| - nitric acid to clean the oxygen electrode | ||
| Bottles with pH-solution 6.3 | 10 | |
| Bottles with pH-solution 9.0 | 10 | |
SANDFILTER