OBJECTIVE: To be able to describe morphological features of the abalone by observing the appearance and internal structures of the abalone.
PROCEDURE:
1.1 Appearance of abalone
Place the abalone in a glass tank to observe their behaviour. Allow them to creep on the bottom and walls of the tank.
A pair of tentacles protrude from the front end of the body. Observe the length and location of the tentacles and touch their tips. Upon disturbance they disappear under the shell, however they reappear after a short interval. These sensitive and chemotactic organs are protrusile.
Immediately below the tentacles, the pair of eyes are visible. The eyestalk is named after its shape. Observe the tentacles of the mantle protruding from beneath the shell. Identify the location and number of tentacles of the upper foot. Count the number of brown spots on the upper foot.
Observe the abalone moving its foot in an undulating and gliding fashion. Observe the abalone grazing on benthic diatoms attached to the bottom of the holding tank, with the aid of its calyx lobe and radula. The calyx lobe and radula are located on the anterior part of the body.
1.2 Shape of the abalone shell
The abalone shell is shaped like an ear and although it is relatively thin it is strong. The shell length is about one-fifth of the width. The spiral is very small. The soft part of the body is broad and wide and occupies most part of the shell.
As for the respiratory pores, only the last two to three stomas are perforated, while old pores are closed to form bosses. The row of respiratory pores and bosses divides the shell into two parts, the upper part which is wide, and the genital slope which has a rough surface and wave-shaped wrinkles.
The abalone shell has a narrow and thickly laid growth-line which is commonly used to calculate the age of the specimen. The outer surface of the shell is blue-brown or ash-brown in colour, however occasionally it may also be red.
The anterior end of the shell is wide and is of an ovate shape. The outer tip is thin, with a sharp edge, while the inner tip is much thicker. The inner tip has a slimy edge.
The inner part of the shell is blue-white or copper-white in colour, with a nacreous lustre.
1.3 Internal organs
Remove the shell from the body by cutting the ligament. While separating the shell from the body, take care to avoid injury to the intestine and other soft organs.
Cut off the mantle with the aid of dissecting scissors in order to observe the internal organs.
Observe the digestive organs of abalone: the gullet, stomach, intestine and anus.
The liver is surrounded by the genital gland of the abalone. Remove a fragment of the tissue with the aid of a sharp scalpel and place it under a microscope for detailed observation.
The measurement of maturity has a great significance in abalone culture.
1.4 Nervous system
Its central nerve ganglion is placed around the mouth. A little scraping will expose the cerebral nerve ganglion. Abalone also has a pair of nerve nodules (placed at the foot, intestine, etc.) and also a collection of nerves connecting them.
EXERCISE:
After the above observations, follow the instructions below:
OBJECTIVE: To be able to select good abalone adults for seed production.
PROCEDURE:
2.1 Selection of adult abalone from the sea
The optimum size of abalone for spawning ranges between 7–9 cm.
The suggested period for catching adult abalone is between March-May and October-November when the water temperature is low. During these periods the abalones are fairly inactive and can be easily caught. In addition, at higher water temperatures injuries inflicted during the catching process infect easily.
2.2 Transportation of abalone
Frequent transportation is necessary in the culture of abalone. Adult abalone may be transported in buckets containing water or without water. Whichever the method used, the abalone must be in a comfortable state so as to reduce mortality. Care must be given to ensure their firm attachment to the bucket or on the attaching boards during transport.
If the distance is short or if the quantity of abalone to be transported is small, they may be transported in buckets containing water. On the other hand, when abalone are transported over long distances or the quantity is large, they are usually transported in containers without water.
The transported abalone should be moved to the rearing tank as soon as possible.
OBJECTIVE: To know how to obtain sexually mature abalone and to be able to control and ensure spawning as and when required.
PROCEDURE:
3.1 Rearing facilities
To induce maturation in adult abalones, the necessary biological and physical conditions should be met throughout the year, and the rearing system itself should be properly maintained.
Plastic drums with a holding capacity of 1–2 tons are often used in the rearing and conditioning of the abalone broodstock. However, concrete tanks are sometimes used.
Corrugated plastic plates (made of vinyl chloride) and/or V-shaped structures (usually made of cement, e.g. roof tiles) are placed in the tanks for the abalones to attach on and seek protection. Abalone may also be matured in plastic netcages which are convenient to maintain
The netcage size should be for easy handling; they are usually square or oval. A one-ton tank with two netcages is recommended as it is fairly convenient for all farm operations including feeding of adult abalone.
3.2 Rearing conditions
Adult abalone sexually mature at water temperatures between 17–21 °C, however proper gonadal growth starts at a water temperature as low as 7.6 °C.
The above temperature minimum is considered the biological zero point in abalone farming, that is, at any temperature above 7.6 °C gonad maturation may be triggered. The sum of the effective accumulated temperature (EAT) is:
(T - 7.6 °C)
where:
T = refers to the average rearing water temperature.
Laboratory observations and trials indicate that the gonad grows at temperatures between 7.6–21 °C.
The formula for the linear relationship between the rate of increase of the gonad index (Y) and water temperature (T) is:
Y = 0.00597T - 0.04537 (T= ≤20 °C)
At an EAT of 800 °C days, the reproductive organs mature, however the spawning rate still remains low. Spawning rate increases up to 98% when an EAT of 1000 °C days is provided. Spawning time can be effectively planned when applying the EAT technique to condition the broodstock.
Laminaria and Undaria are a good feed for the adult abalone. Laminaria can be cultured and has a long rearing period.
During abalone maturation the amount of water change should be ten times the abalone's oxygen consumption. Oxygen stability is normally guaranteed when 100 l/h per kg of abalone are supplied at the rearing water temperature of 20 °C. The formula for calculating the rearing quantity of abalone by the amount of water change is as follows:
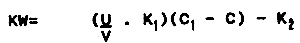
where:
| K | = oxygen consumption by the abalone (ml/kg/hr) |
| W | = possible rearing quantity |
| U | = volume of inflowing water |
| V | = tank capacity (m3) |
| C1 | = dissolved oxygen of inflowing water (ml/l) |
| C | = dissolved oxygen of outflowing water (ml/l) |
| K1 | = coefficient of dissolved oxygen |
| K2 | = oxygen consumption except abalone (ml/l/hr) |
When the abalones are matured, a sudden change in the environmental conditions of the rearing tank may trigger spawning. It is therefore, essential to carefully pay attention to maintain a constant environment.
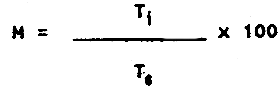
where:
| M = | maturity (%) |
| Ti = | thickness of the middle part of the reproductive sac at the time of investigation |
| Ts = | thickness of the middle part of the reproductive sac before spawning |
The thickness of the middle part of the reproductive sac prior to spawning varies according to the size of the abalone and the prevailing environmental condition at the culture site. Therefore, prior investigation is conducted a relation formula, to ease calculation, should be formulated. In the case of the local (Korea DPR) abalone, the relation formula Y = -4.3 + 1.28 × is used to measure the thickness of the middle part of the reproductive sac before spawning.
EXERCISE:
OBJECTIVE: To be able to perform the technique of controlled spawning.
MATERIALS:
PROCEDURE:
A number of stimulating methods can be applied to spawn mature abalone.
4.1 UV light method
The artificial spawning method should be selected taking into consideration the maturity of adult abalone.
During the stimulation process the female-male ratio should be 4:1. As spawning starts the male and female abalone should be separated and allowed to continue the emission of the genital product.
An abalone with a mature gonad may release its gametes when the UV-sterilized seawater stimulating method is applied. The volume of water to be supplied at the time of ultra-violet exposure is 1/20 of normal sterilizing capacity. Ultra-violet lamps with various capacities and with outputs between 15W-2KW are used. Low-pressure sterilizing lamps are more effective as they emit a great amount of short-waves between 275–180 mm.
The number of adult abalone to be used should be limited according to the number of eggs required. The number of spawned eggs is about 1.5 million per 100 g of gonadal mass of a mature abalone.
EXERCISE:
Select a number of adult abalone and separate them by sex. Measure the shell length and mass, and calculate the gonad index. Remove the abalone from the water and allow them to dry under the shade; the time of air exposure will vary depending on the maturity level of the broodstock, from 30 minutes to 2 hours.
After air-drying, clean the abalone in UV-treated seawater always keeping the sexes separated. Then place them in a tank filled with UV-irradiated seawater. One to two adult abalone are placed in a 20-litre plastic tank while a larger number can be collected in tank with a larger capacity (500–1000 l). When the above operations have been completed the following ones should be carried out.
When the abalone have been released into the tank, the tank should be covered so as to simulate night time. This is because the amount of the genital product released will be at its highest when light exposure is reduced. After one hour check the abalones at intervals of 15–30 minutes and note the beginning of the spawning activity as well as record the number of specimens which have been induced.
When an abalone begins spawning, observe the dispersion of the gametes in the water; siphon out the genital product by using a glass tube cleaned with filter-sterilized seawater to avoid contamination of the eggs and sperms. At this stage the fertilization process can be clearly observed under the microscope. Observe the spawning behavior of the male and female abalone and note the frequency of release.
When the male abalone have ejaculated sperms for an hour or so, move the sperm suspension to another container to estimate its concentration. The sperm concentration can be observed under microscope using a haemocytometer. If the concentration is too thick to be calculated, proceed to dilute the suspension. Another way of evaluating the concentration of sperms is through the use of photo-electric colorimeter. This method is very simple and practical.
When the female abalone have released eggs for one hour or so, transfer the egg suspension to another container and calculate the number of eggs. The number of eggs can be calculated through the volumetric method.
OBJECTIVE: To acquire the techniques of artificial egg fertilization and early embryo handling.
MATERIALS:
PROCEDURE:
5.1 Fertilization
Fertilization rate is increased when it is carried out within one hour from egg release. The quantity of semen required for fertilization is calculated based on its concentration. The recommended egg density during the fertilization process is a maximum of 40 eggs/ml, while the ratio of eggs and sperm should be 1:1000. At a higher ratio, polyspermy usually occurs.
Place the calculated amount of sperm into the bucket containing the freshly released eggs and stir gently in order to facilitate dispersion of the sperm and aid egg fertilization. Examine under the microscope the number of sperms sticking on the membrane of one egg. Three to five sperm heads on one egg is an indication of correct egg:sperm ratio. Too many sperms cause egg shrinkage and consequent abnormal development. The normal rate of fertilization is above 95 %.
5.2 Egg washing
Successfully fertilized eggs sink to the bottom of the tank. The sinking speed of the eggs is 2 cm/sec. Unfertilized and abnormal eggs sink two to three times slower than the fertilized ones.
The upper water layer is removed by suction or by slightly tilting the tank to one side after the eggs have sunk to the bottom. This procedure is carried out to remove unwanted matter such as abnormal and unfertilized eggs. When drawing out the water, at least 15 cm from the bottom should be left in order the prevent the eggs from being sucked out or damaged.
At this stage freshly filtered and sterilized seawater is added to the tank containing the developing embryos. The newly added seawater should have the same temperature as water and the fertilized eggs should soon start floating uniformly. When pouring the additional seawater, attention should be taken not to pile the eggs on one side of the tank as more than one egg layer may induce some eggs to develop abnormally.
The eggs should be washed five to six times until they hatch. If fertilized eggs are piled on the bottom of the tank for more than three hours, their growth is affected as oxygen supply is generally not sufficient. Care should be taken when washing the eggs as handling may affect the subsequent survival of the abalone larvae.
Examine the developing embryos under the microscope. Observe normal and deformed embryos as well as the unfertilized eggs. Measure the diameter of the normal growing eggs and calculate the rate of fertilization. When the eggs have been thoroughly washed add fresh seawater. At this stage fill a 100 ml graduated cylinder by dipping it in five different places of the tank, to be fixed under formalin.
When fixing with formalin, the upper 15–20 ml in the cylinder is removed after the eggs have sunk to the bottom and the same volume of formalin added. The water in the cylinder is stirred and a 0.5–1.0 ml sample is collected by using a pipette for observation under the microscope using a simplified cell counter. At this stage the number of normally developed, unfertilized and deformed eggs can be observed and counted. The count is carried out five times to obtain the average value. The ratio of successful egg fertilization and the ratio of normal egg development can be calculated as follows:
Ratio of fertilization (%) 
Ratio of normal cleavage (%) 
It is important to maintain a constant water temperature during egg cleavage, as even small differences may cause abnormal egg development. The most suitable temperature level for culturing abalone eggs is above 30 °C.
OBJECTIVE: To acquire the techniques of optimizing the culture conditions for rearing free-swimming larvae.
MATERIALS:
PROCEDURE:
The free-swimming period of larvae is usually between 4–10 days, however this may vary under specific conditions.
6.1 Larval appearance
In rearing free-swimming larvae it is important to provide the appropriate environmental conditions to reduce to the minimum the free-swimming period. Newly hatched larvae swim freely with the help of the velum and move to the upper layers of the rearing water as they are actively phototrophic.
6.2 Rearing parameters
Transfer all swimming larvae in the upper layers of the water column to the rearing tank by siphoning with a hose.
Constant water temperature is an important factor in the rearing of free-swimming larvae.
The volume of water to be regularly changed varies according to the larval rearing density. At densities above 1000/l, the entire water volume should be changed once every 6–8 hours, while at 300/l, the entire water volume should be changed daily.
Observe on a regular basis the developmental stages of the larvae and note their activity, shape and mobility as well as calculate the survival rate.
EXERCISE:
a. Observe and compare the activity and behaviour of normal and abnormal larvae.
OBJECTIVE: To acquire the techniques of rearing juvenile abalone.
MATERIALS:
PROCEDURE:
7.1 Major rearing technical and management problems
The physiological and morphological changes in the abalone larvae, such as the growth of the foot and the appearance of the tentacles, take about four days under optimum culture conditions. During this period the larvae search for a suitable surface to initiate their benthic life. If they are unable to locate a bio-chemically or physically suitable surface, they will continue their planktonic life, and the larval period may last up to 10–20 days.
When a suitable surface is found the larvae move from the free-swimming to the creeping stage. This process is timed with the disappearance of the velum. After several metamorphic stages, they become juvenile abalones.
The major management and technical problems at this stage involve the provision of rearing conditions suitable for survival and growth after metamorphosis.
7.2 Preparation of rearing boards
Plastic materials such as polyethylene, polyesterol, acrylic, and PVC are commonly used for manufacturing the rearing boards.
The size and shape of the boards differ according to the rearing facilities available, however wave-shaped plastic plates are usually used. The size of a rearing board is about 30×30 or 40×40 cm. The boards are assembled in frames that hold 10–20 boards, arranged at 3–5 cm intervals in order to facilitate their handling.
The provision of a biologically suitable surface to the benthic larvae is an important factor in terms of their survival and growth. In addition, live food of suitable size and appropriate density should be cultured on the surface of the rearing boards in order to provide nutrients to the creeping larvae.
| Nitrogen | = | 5 ug - nitrogen atom weight |
| Phosphorus | = | 1 ug - phosphorus atom weight |
| Silicon | = | 5 ug - silicon atom weight |
7.3 Rearing of larvae
Collect and transfer free-swimming larvae at the point of metamorphosis, into the rearing tank in which the diatom coated boards have been placed, and adjust the density to about 300 larvae/l.
At this stage the rearing water should be static, as the time taken by the larvae to settle on the boards ranges between few hours to one whole day.
Following the completion of metamorphosis into a creeping larvae, the water should be regularly changed and aeration supplied.
Between 10–20 days after setting, the abalone larvae begin to feed on diatoms about 20 um in size. If feed deficiency on rearing boards is determined through observations under the microscope, the above-mentioned diatom fluid should be added and the water allowed to be static for about eight hours. Diatom propagation in the rearing tank takes place when the light intensity is about 3000 lux.
Attention should also be drawn to the biological management of the rearing tank.
EXERCISE:
Observe metamorphosis and the various developmental stages of the cultured larvae, and estimate the survival rate by counting the number of abalones on at least 20 rearing boards.
OBJECTIVE: To acquire the technique of intermediate abalone rearing.
MATERIALS:
PROCEDURE:
8.1 Rearing facilities and conditions
Land-based intermediate rearing facility
| 1) | ammonia | = | <50 ppb |
| 2) | pH | = | >7.9 |
| 3) | salinity | = | >30 ‰ |
| 4) | DO | = | 70–100 % saturation. |
Sea-based intermediate rearing facility
A sea-based intermediate rearing facility usually consists of a series of floating netcages located in protected sites and kept in position by a system of floats, ropes and weights. When using the above facility it is important to pay particular attention to the selection of the site, as the entire rearing facility may be destroyed and lost during bad weather conditions.
8.2 Intermediate rearing
Rearing density. The rearing density varies according to the initial size of the abalone juvenile, as well as on the type of the rearing device. However, it is normal to stock 5 kg and 3 kg per m3 in land- and sea-based facilities, respectively. Higher stocking densities usually cause the growth rate of abalone to drop, as well as the piling of abalones on top of one another, which then need to be manually separated. When the mass of the cultured abalones duplicates, thinning should be carried out by dividing the abalones in half. Thinning is easily achieved by removing half of the rearing boards and replacing them with new ones on which the abalone will soon settle.
Feed supply. The amount of feed supplied varies according to the size of the abalone juvenile as well as on the prevailing seawater temperature. Feeding rate increases with increase in water temperature and it is usually high among small abalone specimens. The food is usually provided at intervals of 5–10 days, and the amount should be twice the actual feeding capacity of the stocked abalones. Major seaweeds fed to abalone include species such as Laminaria, Undaria and Ulva. Seaweeds used as feed should be cut into small pieces the size of the abalone, cleaned and removed of their roots and fouling organisms.
Abalones of different sizes are usually fed as follows:
If intermediate rearing is carried out on land-based facilities, abalone faeces and leftover feed should be removed regularly. Cleaning of the rearing facilities should also be carried out on a regular basis. When the seawater temperature is above 20 °C, cleaning should be done every 6–7 days while once every 10–15 days at temperatures below 20 °C. Fouling organisms on sea-based rearing installations should be removed as they hinder water flow. Netcages should be regularly cleaned or changed at the time of feeding.
On land-based rearing facilities, a 30 mm abalone may double in size within one year, while on sea-based installations, abalone growth occurs mainly from May to November. During the intermediate rearing period, the major factors influencing growth rate are water quality, mechanical disturbance (eg. wave action in sea-based facilities), excessive rearing density or unsuitable rearing structures.
It is important to bear in mind that regular investigation on environmental conditions and on the performance of the abalone is needed. The survival rate of abalones during intermediate rearing is usually about 80 %, attaining a shell length of 30 mm.
PUBLICATIONS AND DOCUMENTS OF THE
REGIONAL SEAFARMING DEVELOPMENT AND DEMONSTRATION PROJECT
RAS/90/002 (RAS/86/024)
Working Papers
RAS/86/024
NACA-SF/WP/87/1. Lovatelli, A. Status of scallop farming: A review of techniques. 22 pp.
NACA-SF/WP/88/2. Lovatelli, A. Status of oyster culture in selected Asian countries. 96 pp.
NACA-SF/WP/88/3. Lovatelli, A. and P. B. Bueno, (eds). Seminar report on the status of oyster culture in China, Indonesia, Malaysia, Philippines and Thailand. 55 pp.
NACA-SF/WP/88/4. Lovatelli, A. Status of mollusc culture in selected Asian countries. 75 pp.
NACA-SF/WP/88/5. Lovatelli, A. and P. B. Bueno, (eds). Seminar report on the status of seaweed culture in China, India, Indonesia, ROKorea, Malaysia, Philippines and Thailand. 79 pp.
NACA-SF/WP/88/6. Lovatelli, A. and P. B. Bueno, (eds). Seminar report on the status of finfish culture in China, DPRKorea, Indonesia, ROKorea, Malaysia and Singapore. 53 pp.
NACA-SF/WP/88/7. Lovatelli, A. Seafarming production statistics from China, Indonesia, ROKorea, Philippines, Singapore and Thailand. 37 pp.
NACA-SF/WP/88/8. Lovatelli, A. Site selection for mollusc culture. 25 pp.
NACA-SF/WP/88/9. Lovatelli, A. and P. B. Bueno, (eds). Seminar report on the status of finfish netcage culture in China, DPRKorea, Indonesia, ROKorea, Malaysia, Philippines, Singapore and Thailand. 56 pp.
NACA-SF/WP/86/10. Chong, K. C. Economic and social considerations for aquaculture site selection: an Asian perspective. 17 pp.
NACA-SF/WP/89/11. Chen J. X. and A. Lovatelli. Laminaria culture - Site selection criteria and guidelines. 30 pp.
NACA-SF/WP/89/12. Chen J. X. Gracilaria culture in China. 18 pp.
NACA-SF/WP/89/13. Seafarming Project, RAS/86/024. Site selection criteria for marine finfish netcage culture in Asia. 21 pp.
NACA-SF/WP/89/14. Lovatelli A. Seafarming production statistics from China, India, Indonesia, ROKorea, Philippines, Singapore and Thailand. 47 pp.
NACA-SF/WP/89/15. Chong K. C. and D. B. S. Sehara. Women in aquaculture research and training. 20 pp.
Working Papers
RAS/90/002
SF/WP/90/1. Chen J. X. Brief introduction to mariculture of five selected species in China. 32 pp.
Bibliography
NACA-SF/BIB/88/1. Selected bibliography on seafarming species and production systems. 20 pp.
NACA-SF/BIB/88/2. Selected bibliography on seafarming species and production systems. 52 pp.
NACA-SF/BIB/89/1. Selected bibliography on seafarming species and production systems. 49 pp.
Training Manuals
Manual on seaweed farming: Eucheuma spp. (Training manual No. 1). 25 pp.
Culture of the pacific oyster (Crassostrea gigas) in the Republic of Korea. (Training manual No. 2). 64 pp.
Culture of the seabass (Lates calcarifer) in Thailand. Training manual No. 3. 90 pp.
Training manual on marine finfish netcage culture in Singapore. (Training manual No. 4). 275 pp.
Culture of Kelp (Laminaria japonica) in China. (Training manual No. 5). 204 pp.
Training manual on Gracilaria culture and seaweed processing in China. Training manual No. 6. 155 pp.
Meeting Reports
Report of the First National Coordinators' Meeting of the Regional Seafarming Development and Demonstration Project, 27–30 October 1987, Bangkok, Thailand. 71 pp.
Report of the Second National Coordinators' Meeting of the Regional Seafarming Development and Demonstration Project, 20–23 September 1988, Singapore. 102 pp.
Report of the Third National Coordinators' Meeting of the Regional Seafarming Development and Demonstration Project, 24–27 August 1989, Qingdao, China. 103 pp.
Workshop Reports
Report of the FAO Asian Regional Workshop on Geographical Information Systems: Applications in Aquaculture, 5–23 December 1988, Bangkok, Thailand. FAO Fisheries Report No. 414, FIRI/R414. 13 pp.
Report of the Workshop and Study Tour On Mollusc Sanitation and Marketing, 15–28 September 1989, France. FAO/UNDP Regional Seafarming Development and Demonstration Project RAS/86/024. 212 pp.
General Reports
Progress report on the 1988 Regional Training/Demonstration Courses organized under the Regional Seafarming Development and Demonstration Project (RAS/86/024). 26 pp.
Report of the Seafarming Resources Atlas Mission. Regional Seafarming Project RAS/86/024, July 1989. 74 pp.
Audio-visual Materials
Culture of the Pacific Oyster (Crassostrea gigas) in the Republic of Korea. 71 slides.
Culture of the seabass (Lates calcarifer) in Thailand. 40 slides.
Marine finfish netcage culture in Singapore. 37 slides.
Culture of Kelp (Laminaria japonica) in China. 30 minutes video.
Seafarming Atlas Series
Regional Seafarming Resources Atlas. FAO/UNDP Regional Seafarming Development and Demonstration project (RAS/86/024), January 1990. (Atlas series No. 1.). 83 pp.
