by
Sukotjo Adisukresno1
1. INTRODUCTION
Macrobrachium rosenbergii is available in the tropical and subtropical countries in the Indo-Pacific region. The prawn occupied rivers, lakes or swamps, life both in fresh-or brackishwaters. Catching from natural waters without control may cause the extinction of this animal. To overcome these problems, the culture of this prawn in farm should be improved.
The improvement of this culture should be based on the biological characteristics of this animal. Macrobrachium rosenbergii has the following biological characteristics:
Active searching food during night (nocturnal)
2. MANAGEMENT
2.1 Omnivorous
In natural waters, this animal feed on worms, snails, insect larvae, seed, soft stem of aquatic plants, small fish, small crustaceans, detritus, etc. The feed in the culture, all kinds of food might be given. The food should be any kind of material which is available locally and cheap. In the country where trash fish is cheap, feeding Macrobrachium with fish gives satisfactory production, since the food contain high protein. Rice bran, coconut oil cake, waste from food factory, vegetables, soft leaves of aquatic plants might be used to feed the prawn.
Aside from these, the prawn needs also the natural food available in the pond. To provide natural food with enough nutrient, manure and fertilizer should be applied regularly. The amount of manure and fertilizer applied depends on the nature of the pond soil. In general, the manure of cowdung, carabao dung, pig dung, chicken droppings, bat droppings, might be applied at the rate of 500–1000 kg/ha for the first application (on the preparation of pond) and then every month apply 100–300 kg/ha. The fertilizer used might be compound fertilizer, urea, triple superphosphate (T.S.P.), Z.A. (Zwavelzuur Ammonia), etc. The amount is 100–200 kg/ha for the first application and then 25–50 kg/ha every month. The manure and fertilizer will promote the growth of benthic algae, plankton and also the worms, small crustaceans, aquatic insect larvae, etc.
2.2 Moult periodically
Like the other crustaceans, the prawn is moulting periodically. The frequency of moulting depends on:
Sex: the male prawns moult more frequently than the females.
During the culture, the prawn should get enough food whether it is from nature or from the supplemented feed. The newly moulted animals are very weak and this invites the strong animals to attack them as prey (cannibalism).
2.3 Cannibalism
Cannibalism occurs when the food is insufficient, or when there are moulted animals. To prevent the cannibalism, the pond should be provided with shelter or hiding place for the prawn. This device might be:
Branches or twigs: aside from hiding place, it is also the place for the animal searching food of benthic algae which grow on them and prevent pouching by cast net.
2.4 Avoid direct strong light
The pond should be provided with shelter which is mentioned in the above paragraph. Aside from these, the deep pond gives better survival rate and production. The depth suggested is 0.8–1.5 m.
2.5 Nocturnal habit
The prawn actively searches food during the night. The young prawn is active in searching for food all day round. Feeding the prawn in the morning is inefficient, it is better to feed the cultured prawn in the evening. During day time, the prawn stays quietly on the bottom.
Taking advantage of this characteristic, the prawn might be harvested during the evening, with cast net, gillnet or trap net.
3. OTHER MAINTENANCE OPERATIONS
Since the prawn is aquatic animal and it consumes higher amount of oxygen than fish, water maintenance should be maintained properly. Water from the bottom layer should be discharged everyday and replaced with new freshwater. The water replacement should be at least 10% of the volume of the pond everyday. To discharge water from the bottom layer it might be done by:
Using stand pipe or valve in case the pond is provided with water outlet
4. SUMMARY
Feed the animal with supplemental feed available locally which are cheap.
by
Budiono Martosudarmo1
1. INTRODUCTION
Most of the larvae of fish and crustaceans in their natural habitat feed on plankton which consists of phytoplankton and zooplankton. Zooplankton themselves in their life cycle depend also for their existence on phytoplankton.
In hatcheries, usage of certain kinds of planktons as food organisms for fish and prawn larval rearing is of prime importance. Almost every hatchery never eliminate application of food organisms in their larval rearing although artificial food have been widely developed. A number of workers reported the involvement of food organisms in their work of larval rearing which is collected from ponds or natural waters as well as cultured purposely in a separate tank.
In the Macrobrachium larval rearing, the food organisms used mainly so far are Chlorella sp. and Artemia nauplii. Sometimes Moina sp., a genus of cladoceran, is also used in the giant freshwater prawn larval rearing as practised in Thailand. Fujimura (1966) is among the authors who recommended the use of Chlorella water or as it is known “green water” in increasing the efficiency of Macrobrachium larval rearing.
This account is meant for providing an information on the culture of food organisms mainly Chlorella sp.
2. THE ROLE OF ALGAE
In general, algae besides serving as food for the larvae, act also as biological agent in detoxifying the larval rearing medium by incorporating or assimilating the undesired materials in the water mainly ammonia and nitrate. It has been proven by Cohen et al (1976) that the green algae facilitate the growth of Macrobrachium larvae indirectly by removing toxic materials notably ammonia from the medium.
1 Senior staff, Brackishwater Aquaculture Development Center, Jepara, Indonesia
Meanwhile, Manzi, J.J., Maddox and Sandifer (1977) reported that unialgal supplement in the larval tank significantly increased the survival rate of the larvae of M. rosenbergii in both static and recirculating water system. They concluded further that the mechanisms responsible for algae enhancement of this prawn larviculture still remain speculative. Algae could be a direct nutritional source, either by accidental ingestion or active feeding by the larvae and could also reduce nitrogenous compound by utilizing ammonia and nitrate derived from metabolic waste of the larvae.
3. THE PROCEDURES OF CHLORELLA CULTURE IN FIVE-TON TANK
Wash the container thoroughly and flush with 100 ppm chlorine solution to eradicate the undesired micro-organisms left in the container; and then let it be dry for a few minutes.
Fill in a mixture of seawater and freshwater to make a medium culture of 12 ppt. The water should be filtered through 60 u or 100 u filter bag to prevent predators from coming along with the water. Make the water to a depth of 30 cm.
To make sure that there is no predator (mostly rotifers) in the culture medium, add 2 ppm CuSO4 and mix thoroughly.
Prepare Allen Miquel solution which consists of:
| Na2HPO4. 12 H2O | : | 40 g | - | - mix in 800 cc of distilled water |
| CaCl2. 6H2O | : | 20 g | ||
| FeCl3 | : | 20 g | - | |
| HCl | : | 20 co | - |
Fertilize the water medium with urea 50 ppm and 800 cc A/M solution.
Inoculate the water with about 5–10 pails of Chlorella water or green water.
Stock the water with Tilapia sp. of about 10 specimens in order to provide organic fertilizer in the tank through the faecal matter of the Tilapia. Pelletized feed of 5 percent of total body weight is sufficient to be given to Tilapia to maintain their growth.
Provide sufficient aeration to the culture to maintain an even distribution of population of the Chlorella and to supply oxygen at night.
Sunlight as a source of energy for their photosynthesis should not be neglected. Accordingly, before culturing this algae, a location with sufficient illumination should be selected.
After 5 to 7 days, the water colour becomes green indicating that a dense growth of Chlorella have been attained. The density of Chlorella cells obtained in these methods is about 1 million per cc.
To maintain the growth of the algae check always the presence of predators, that is rotifers - Brachionus plicatilis, in the culture. The presence of rotifers can be controlled by application of CuSO4, 2 ppm. This chemical is not harmful to the blue green algae.
4. LITERATURES CITED
Cohen, D., A. Finkel and M. Sussman. 1976 On the role of algae in larviculture of Macrobrachium rosenbergii, Aquaculture 8: 199–207
Fujimura, T. 1966 Notes on the development of a practical mass culturing technique for the giant prawn Macrobrachium rosenbergii. Proceeding: IPFC 12th Session. Honolulu, Hawaii IPFC/C66/WP 47
Manzi, J.J., M.B. Maddox and P.A. Sandifer. 1977 Algal supplement enhancement of Macrobrachium rosenbergii (De Man) larviculture.
by
Ruben Manik1
1. INTRODUCTION
The so-called giant freshwater prawn, Macrobrachium rosenbergii, is widely distributed in the Indo-Pacific region, ranging from Australia to New Guinea to Indus River delta. This species has emerged in the last few years as one of the aquatic animals having a very high potential for aquaculture.
This is based on a number of advantages of this species over many other crustaceans. It adapts to a relatively wide range of temperature from a minimum of 15 to a maximum of 35°C. This species has a relatively short larval life. It is also a fast growing species. Fast growing individuals reach market size in about 7–8 months, and the meat is of high quality in terms of tests and texture.
In Indonesia, the potential area for this farming are paddy-fields, freshwater ponds, rivers (enclosures) and even brackishwater ponds of low salinity.
Food is normally the largest single item in the running expenditure of a prawn hatchery or prawn farm. If prawns are held in artificial confinement where natural food are absent or limited, an external food source should be added. For example, the external food of freshwater prawn larvae that are held in tanks may consist of live food (nauplii of brine shrimp) or artificial food (fish egg, fish flesh, formulated feeds, etc.)
The production of freshwater prawn stocked in pond depends on the ability of the environment to produce natural food. Various factors such as soil and water fertility, water temperature and intensity of solar radiation affect the production level of natural food in the pond. However, for maximum rate of performance, the supplemental feeding programme is supposed to be important. Hence, the knowledge of nutrient requirements, the preparation of suitable feeds from the local available ingredients, feeding techniques, and the cost effectiveness of prepared feeds is of paramount importance to commercial success.
1 Fisheries Technologist, Provincial Fisheries Service, Central Java Province, Semarang, Indonesia
2. NUTRIENT REQUIREMENTS OF FRESHWATER PRAWN
The nutritional requirements of fish are similar to those of land animals. They need to consume proteins, minerals, growth factors, and energy sources for growth, reproduction and other normal physiological functions. A deficiency of one or more of the essential nutrients results in reduced rate of performance, diseases or even death.
The nutrients may come from natural aquatic organisms or from prepared diets. If prawns are held in artificial confinement where natural foods are absent or limited such as tanks, raceways, their prepared diets must be nutritionally complete; however, where natural food is available and supplemental diets are only fed additionally for growth, the diets may not contain all of the essential nutrients.
In most cases, the fish culturist is interested in maximum rate of performance. However, there may be other objectives in a feeding programme, such as holding fish in a certain desired size for a long period (milkfish stunting programme), reproductive purposes (broodstock diets). Under all of these conditions a nutritionally balanced diet is important.
The nutritional requirements of fish do not seem to vary greatly among species. The quantitative nutrient requirements that have been derived for several species of fish are probably adequate basis for estimating the nutrients need of others.
2.1 Proteins and amino acids
Protein is a major constituent of the animal body, and a liberal and continuous supply is needed throughout life. The primary aim of fish culture is to transform dietary protein into tissue protein efficiently. Since protein is the most costly ingredient or nutrient in fish feeds, optimized utilization of dietary protein is essential for economical production. Exceptionally prepared diets of high protein quality is needed for fish larvae in relatively small quantity.
Generally fish require a higher percentage of protein in their diet than birds and mammals. Protein level in successful fish foods range from 20 to 60 percent to accommodate for differences in the physiological needs of different fishes. For example, the optimum level of protein in prepared diets for warm water food fish is 30–36 percent, whereas in poultry diets the practical level is 16–22 percent. This may be because fishes utilize carbohydrates less efficiently. Therefore, some dietary protein may be metabolized for energy.
The amount of protein that should be provided in prepared diets depend on several factors. Some of these factors are:
Economic considerations
Freshwater prawn have higher protein requirements during early life than during later phase of growth. During the larval stage, the prawn requires high quality protein with higher protein level than juveniles.
The protein quality affects the amount of protein that should be provided in prepared diets. More of a low quality protein is needed in the diet for maximum growth than a high-quality protein. The protein quality is primarily influenced by amino acid composition and digestibility. Qualitatively, the essential amino acids for prawn are similar to those for other fish and animals. The essential amino acids must be provided in the diet. The quantitative requirements of essential amino acids for freshwater prawn are not yet known. But, for practical purposes, the quantitative requirements of essential amino acids for other fishes can be used as guidelines for estimation as shown in Table 1. Higher protein level are required for maximum growth rate in all-plant diets than in the diets containing some fish meal. Inclusion of protein from animal sources in the diets gave better results.
| Amino acid | Channel catfish | Eel (% of protein) | Salmonid |
| Arginine | 4.3 | 3.9 | 6.0 |
| Histidine | 1.6 | 1.9 | 1.8 |
| Isoleucine | 2.3 | 4.1 | 2.2 |
| Leucine | 3.4 | 3.6 | 3.9 |
| Lysine | 5.1 | 4.8 | 5.0 |
| Methionine + Cystine | 2.3 | 4.5 | 4.0 |
| Phenylalanine + Tyrosine | R2 | R | 5.1 |
| Threonine | 2.2 | 2.2 | 3.6 |
| Tryptophan | 0.5 | 1.0 | 0.5 |
| Veline | 2.8 | 3.6 | 3.2 |
Source: Channel catfish, Lovel (1978); Eel, Nose (1978);
salmonid, National Research Council (1973).
R means required but quantity has not been determined.
Data for other species show adding fish meal, which is rich in methionine, to all plant diets low in methionine increased gain per unit of protein fed and reduced the amount of protein required for maximum growth. The percentage of protein in prepared diets is not synonymous with the amount of protein available from the prepared feeds to the prawn. Prawn digest protein in natural food quite well. Protein in some feedstuffs such as fish meal and soybean meal are highly available to fish. Protein in grain and fibrous feeds is less digestible to fish than land animals.
Comparison of digestion coefficient for several feedstuffs for some species of prawn are presented in Table 2.
| Species | Feedstuffs | Digestion coefficient (%) |
| Palaemon serratus | Corn, gluten meal | 93 |
| Shrimp, cannery residue, meal | 88 | |
| Palaemon serratus | Fish, anchovy meal, mechanically extracted | 82 |
| Animal by product, meal rendered (dried slaughter house offal) | 83 | |
| Cotton seed, meal, mechanically extracted | 82 | |
| Peanut, kernels, meal, solvent extracted | 90 | |
| Penaeus japonicus | Algae, diatom, whole, fresh | 63 |
| Shrimp, whole, fresh | 86 |
Source: National Research Council (1977)
In order to utilize protein efficiently for growth, other sources of energy must be present in the diets. All fish have the ability to use fat and carbohydrates to some extent to spare protein as an energy nutrient. Thus, if the diet is deficient in energy the fish will use part of the protein to meet energy needs.
The other factor that affects the level of protein in the prepared diets is the availability of natural food in the environment. The protein level in the formulated diet may be less if natural food contribute significantly to the daily food intake of the fish.
Protein is by far the most costly nutrient in fish feeds. Therefore, optimized utilization of dietary protein is essential for economical viewpoint in growout pond. On the other hand, economical consideration for preparation of prawn larval food is often neglected because the prepared diet is needed in small quantities.
2.2 Lipids and fatty acids
Lipids or fats are stored within the tissues of an organism in a variety of forms, but are most commonly found in the diets in the form of triglycerides. Hydrolysis of triglycerides by lipases result from conversion of the triglycerides to free fatty acids and glycerol.
Understanding the nomenclature of the fatty acids is important because different families of fatty acids have been found to be essential to the well being of different animals.
High level of poly-unsaturated fatty acids (PUFA) with carbon chains 18 to 20 units in length are generally found in the tissues of fishes.
There are three major PUFA families: the oleic (omega 9), linoleic (omega 6), and linolenic (omega 3) acid series. Oleic, linoleic, and linolenic acids are written as 18:1 omega 9, 18:2 omega 6, and 18:3 omega 3, respectively. In the case of oleic acid, there are 18 carbon atoms in the molecule with one double bond located between the ninth and tenth carbon atom from the methyl and of the molecule. Linoleic acid has two double bonds and linolenic acid three, with their positions from the methyl end of the molecule being mix and three carbon atoms, respectively (the omega position).
Fat and oils are high energy components of prepared diets. In most ingredients, they are 85–95 percent digestible. Successful fish feeds contain from 4 to 18 percent fat. Fat as an energy source has a protein sparing action especially useful in foods for young fish where protein needs for growth are a maximum. Lipids or fat contain approximately twice as many calories per gram as do protein and carbohydrates; thus they contribute greatly to the energy level of diets even when present in relatively low quantity.
The level of lipid in prawn feed has not been determined with any precision and will depend in part, on the level of other energy sources, the size of prawn, and environmental conditions such as temperature. Commercial diet for carp at warm water temperature may contain 10 to 15 percent fat, while lower level of fat are used at temperature below 20°C. Eel diet may contain up to 10 percent fat.
Too much lipid in the diet can have negative effects. When lipid is present in excess the fish lay down high level of depot fat, especially in association with the abdominal cavity walls and viscera. Certain nutritional diseases may occur such as fatty livers when excessive dietary lipid level are present.
If the diet contain high level of poly-unsaturated fatty acid will be easily oxidized into peroxides and other toxic compounds. The prawn fed with this diet may result in poor growth and survival rate, and other adverse effects. The addition of some antioxidant substances can retard the oxidation of fat. The kinds and level of antioxidant that recommended in feed are shown in Table 3.
| Common name | Generic name | Level (ppm) |
| Ethoxyquin | 1, 2 dihydro-6-ethoxy-2, 2, 4-trimethylquinoline | 150 |
| BHT | butylated hydroxytoluene | 200 |
| BHA | butylated hydroxyanisole | 200 |
Source: Rumsey (1978) personal communication
There is evidence that fat and fatty acids have an additional role in providing a dietary source of essential fatty acids, linoleic and lonolenic series, required for maximal growth and normal tissues deposition. Most fishes require relatively high level in omega 3 (linoleic group) than omega 6 (linolenic group) fatty acids series.
2.3 Carbohydrates and fiber
Carbohydrates, refers specifically to the “nitrogen-free extract” portion of feed, are found in most ingredient of plant origin. Carbohydrates are the least expensive form of dietary energy for man and domestic animals, but most fishes appear to make much less use of them. One reason may be that the natural food of wild fishes are not rich in digestible carbohydrates.
If their digestion occurs, product of hydrolysis are assimilated to the blood stream where their only known function is to provide energy, thus they have a protein sparing action.
Carbohydrates may also serve as percusors for the various metabolic intermediates such as non-essential amino acids, chitin synthesis, glycogen storage, fatty acid (muscular fat).
The range of carbohydrates found in prepared feeds is 10 to 50 percent and the efficiency of utilization as energy varies from 40 to 99 percent. The ability of fish to assimilate carbohydrates depend on their ability to elaborate amylase. This is closely related to food habits of the species. Principally, all species of fish have been shown to secrete alpha-amylase. Carnivores, like the yellowtail fish, deal with little carbohydrates. Omnivores, such as the common carp, are able to digest fair amounts of carbohydrates in their diets. The grass carp, a herbivore, subsists primarily on a vegetarian diet. In carnivorous fish, amylase is primarily of pancreatic origin, whereas in herbivorous the enzyme is widespread throughout the entire digestive tract. Thus the activity of this enzyme is greatest in herbivores.
Caridean prawn assimilate carbohydrates with varying degrees of efficiency, according to their source and the species the prawn fed. Wheat starch is assimilated better than potato starch by Palaemon serratus. The freshwater prawn, M. rosenbergii, as omnivorous animal, is expected to assimilate carbohydrates better than penaeid shrimp. Strong carbohydrates activity has been shown in the herbivorous prawn, Macrobrachium dayanum.
Indigestible dietary material, such as plant cell wall material, which are usually referred to as fiber, is not a necessary component in production rations for optimum rate of growth or nutrient digestibility. Fiber may serve a role in fish rations as a diluent for other nutrients and as an extender in the ration to help insure equitable distribution of nutrients to all fish. Level as high as 21 percent reduce nutrient intake and impair digestibility of nutrients. Fiber in less than 8 percent may add structural integrity to pellet diets, but larger amounts may impair pellet quality.
2.4 Vitamins
Besides the three major classes of nutrient (carbohydrates, protein, fat) fish needs the non-energy nutrients such as vitamins and minerals. Vitamins are organic compound other than carbohydrates, fats, and protein that are essential to animal and human life. Not all species of animals require the same vitamins in their diets. Most of the vitamins are required by fish.
Although the amounts of vitamins needed by the body are small compared with the amounts required of carbohydrates, fats and protein, deficiency of these accessory food factors result in serious diseases specific for each vitamin. One of the first symptoms of a deficiency of any of the 13 to 15 essential vitamins for warm water fish is depressed appetite and reduced growth rate. Other common symptoms are abnormal color, lack of coordination, nervousness, fatty livers, and increased susceptibility to bacterial infections.
Fish from the wild seldom show signs of nutritional diseases because natural aquatic foods are fairly nutritious, especially in the essential growth factors like vitamins and minerals, and the fish growth rate is limited to the amount of energy and protein in the natural foods.
Traditionally, nutritional diseases are not a serious problem in pond culture of fish, primarily because with moderate stocking conditions the natural food in ponds satisfactorily supplement the artificial feeds with any deficient growth factor. However, when fish were reared intensively in ponds, or artificial conditions (raceways, tanks, cages, etc.) the nutritional adequacy of the prepared feed become more critical, because the availability of the natural foods decreases or is absent. In this case, the supplementation of vitamins in prepared feeds become important. Vitamin requirements for fishes are affected by size, age, environment conditions, water temperature and nutrient relationship.
The recommended level of vitamins for catfish, carp, and eel may serve as model for vitamin requirements for freshwater prawn diets. Based on this information, the recommended allowances are given in Table 4. Requirements may be different for brood fish, fish growing at different temperature, or fish reared at different cultural conditions.
| Vitamin | Amount (per kg) in dry diet | |
| Supplemental diet | Complete diet | |
| Vitamin A activity | 2 000 IU | 5 500 IU |
| Vitamin D activity | 220 IU | 1 000 IU |
| Vitamin E | 11 IU | 50 IU |
| Vitamin K | 5 mg | 10 mg |
| Choline | 440 mg | 550 mg |
| Niacin | 17–28 mg | 100 mg |
| Riboflavin | 2–7 mg | 20 mg |
| Pyridoxine | 11 mg | 20 mg |
| Thiamine | 0 mg | 20 mg |
| D-calcium-pantothenate | 7–11 mg | 50 mg |
| Biotin | 0 mg | 0.1 mg |
| Folacin | 0 mg | 5 mg |
| Vitamin B 12 | 2–10 ug | 20 ug |
| Ascorbic acid | 0–100 mg | 30–100 mg |
| Inositol | 0 mg | 100 mg |
Source: National Research Council (1977)
Formulated diet (supplemental of complete diet) contain these vitamins as a premix (Aquamix, Pfizer Premix A, B, etc.) additive compounded by special formulation. Many prawn/fish farmers in Indonesia add at 1.0 to 2.5 percent of premix (Aquamix of Pfizer Premix) per kg of feed.
2.5 Minerals
Minerals or inorganic elements required by fish for tissue formation, various metabolic process, and to maintain osmotic balance between fluids in their body and the water.
Mineral in the water can make significant contributions to the fish requirements for some minerals, such as calcium, but may be insignificant for others, especially in freshwater environment.
Fish require large amounts of calcium and phosphorus for growth and development. Most fish appear to be able to absorb enough calcium from the water across the gills, for normal growth except when water is low in calcium (soft water).
Unlike land mammals, the ratio of calcium to the phosphorus in the diet is not critical unless the dissolved calcium in the water is very low. Fish appear to have an ability to balance Ca/p ratios by controlling the absorption and excretion of calcium for optimal utilization of both mineral elements.
Levels of dissolved phosphorus are very low in natural waters in relation to calcium; consequently, the water in fish culture environments is not a significant source of phosphorus. Dietary deficiencies in phosphorus have caused reductions in growth rate, body content of calcium and phosphorus, and appetite in fish. Deformed backs (lordosis) and heads have been associated with phosphorus deficiency in carp in Japan.
The minimum requirements in phosphorus of available phosphorus in the diets for freshwater prawn have not been determined. For other species, such as channel catfish, the minimum requirements of phosphorus is 0.8 percent using practical diet ingredients.
Feed ingredients vary widely in their calcium and phosphorus content. Fish meal, a principal ingredient in fish feeds, is rich in both calcium and phosphorus. On the other hand, feed ingredients of plant origin usually lack calcium, and despite a fairly high content of phosphorus: the latter is predominantly in the form of phytin of phytic acid (e.g. soybean meal) which is not readily available for absorption by fish. Phosphorus availability of common feedstuffs varies from 33 percent for grains to 50 percent for fish meal or animal by-products. Soybean meal has an intermediate phosphorus availability of 40 percent. Dicalcium phosphate has the highest availability (80 percent).
Natural food as well as most artificial feed ingredients are usually adequate in potassium, magnesium, sodium and chlorine for normal growth of animals unless there is a high rate of mineral loss. These elements are probably available in sufficient quantity in prepared feed without mineral supplementation.
2.6 Energy-protein relationship
Like other animal fish or prawn require energy for growth, activity, and reproduction. The biological process utilizing energy is defined as metabolism. The rate at which energy utilization occurs is called metabolic rate. Metabolic rate in fishes is influenced by temperature, species, age or body size, activity, physical condition, and water chemistry parameters, such as oxygen or carbon dioxide saturation, pH and salinity.
The energy needs of fish are supplied by fats, carbohydrates and proteins. Protein are used very efficiently by fish as a source of energy. On the other hand, the function of dietary protein is to serve as a source of amino acids for synthesis of body protein.
However, if inadequate glucose or fatty acids are present, then the fish will break down amino acids to produce energy. If this occurs, additional protein must be consumed by the fish in order to furnish the intact amino acids necessary for making the body proteins. Excessive dietary energy intake may restrict protein consumption and subsequent growth. Therefore, it is difficult to determine a specific energy or protein requirement per se without considering the relationship of one to the other.
For certain species, such as channel catfish fingerling, the optimum protein-to-energy ratio (P/E) had been determined. The P/E ratio of 88 mg protein/kcal is optimum for channel catfish fingerling. As a rule of thumb, the P/E ratio range between 100–125 mg protein/kcal. The ratio will vary according to availability of natural food in the environment.
Fat contains more energy per unit weight than any other biological product. Generally, fats are well digested and utilized by fish. It is usually estimated that fat provides 8.5 kcal metabolizable energy (ME) per gram.
Carbohydrates in feed material range from easily digested sugar to complex cellulose molecules which cannot be digested by animals. There is controversy as to the value of carbohydrates in fish feeds. It appears however that digestible carbohydrates can be well utilized as an energy source if it is kept in proper balance with other nutrients. The ME value of carbohydrates range from near zero for cellulose to about 3.8 kcal/g for easily digested sugars. Raw starch range from 1.2 to 2.0 kcal ME/g. Cooking of starch can increase the ME about 3.2 kcal/g. Heat and moisture associated with pelleting process improves the digestibility of starch feed materials. As a conclusion, the value of carbohydrates in fish feeds depend on the source and type of carbohydrates and the processing to which it has been subjected.
Protein has a ME value of about 4.5 kcal/g for fish, which is higher than that for mammals and birds. Protein is used very efficiently by fish as a source of energy but for economic reason should be kept to a minimum consistent with good growth and cheaper carbohydrates and fats should be used to supply most of the energy.
3. DIET FORMULATION
3.1 Systematic method of formulating diet
As mentioned before, the nutritional requirements of freshwater prawn are not known with any reliability and only few data are available.
Therefore, much of the feed formulation and development is accomplished by trial and error. A systematic method of formulating a diet should be established by a feed formulator.
First, the parameters for the diet should be defined:
Is the diet to be prepared for the larvae, juvenile, or stock growout pond? If for stock growout pond, is the diet as supplemental or complete?
Are there any special conditions to be considered such as water quality, etc.?
Is there another negative effect with the use of this diet? For example, some ingredients contain natural toxin substances, or some contamination that will retard the growth of fish.
3.2 Criteria for ingredient selection
The quality of prepared diets is governed initially by the selection of high quality ingredients. Consequently, the feed formulator should be concerned and informed about all aspects of feed quality. In relation to the feed quality, there are several criteria concerning the selection of feed ingredients:
Determination of chemical composition or nutritive value of the diet of ingredients. The general composition of the diets include:
Vitamin potency
For practical purpose, the information from research agency is very useful for estimation of nutritive value of ingredients or prepared diets. But direct determination of nutritive value of ingredients is better than estimation.
The other ingredients are selected to balance the diet in terms of the general composition.
The percentage of various ingredients is altered for obtaining an amino acid balance.
The ingredients should not contain toxic substances that will retard the growth of fish or prawn. If certain ingredients contain toxic substances or other undesirable features, the feed formulator should establish the maximum level of that ingredients in the diets.
The ingredients are acceptable and palatable to the prawn.
The ingredients can be digested by the prawn. Although the digestibility coefficient of ingredients are limited for freshwater prawn, data for other species can be used as a guide.
Since economics of the diets must also be considered, the following criteria are also very important, i.e.:
The cost of ingredients are compared with others which also could be used to determine a least cost formula.
The basic ingredients in the diet is not available seasonally only.
All ingredients are expected to be locally available and not competitive for human consumption.
3.3 Toxins and antigrowth substances in feeds
Toxic materials affecting fish feed ingredients are mycotoxins, toxic products in plant materials, bacteria, residues of pesticides, and agricultural and industrial chemicals.
These toxic and antigrowth substances can be grouped into:
Biological toxins
3.3.1 Chemical contaminants
Some research workers found that the most common dietary contaminants have been the organo-chlorine pesticides, DDT, dieldrin, endrin and industrial chemicals such as polychlorinated biphenyls (PCB) and hexachlor benzene (HCB). Sources of residues can often be traced to the field where the foodstuffs was produced. Accidental contamination with various toxic materials may occur during processing such as fish meal, fish oil, etc.
Other sources of chemical contaminants such as boiler water additives, lubricants (on pellet mill rolls and other processing equipment) are examples of material that could become feed additives during feed processing.
3.3.2 Biological toxins
Salmonella - This bacteria is a very serious source of contamination caused by rodent contamination of stored ingredients. Animal byproducts and fish meals are the worst offenders and the primary source of Salmonella contamination in fish feed.
Mycotoxin - Mycotoxin of mold toxin affect many animals, including fishes. The mold Aspergillus flavus, which produces aflatoxin, is found in many plant materials including cottonseed, peanuts, soybeans, farm grain, and by-products of processed oil seeds. Certain other molds also produce toxins to animals.
Natural toxins and inhibitors - There are many plants that may cause toxic reactions when ingested by animals, including fish, but only a few of these are used in feeds that are important to the fish. Table 5 lists several feedstuffs that sometime cause toxic reactions. The feedstuffs that are most likely to be a problem in the feed are cottonseed meal, kapok seed meal, soybean meal, and legume leaves.
| Feedstuffs | Inhibitors | Method of reduction or elimination of activity |
| Cottonseed meal and kapok seed meal | goosypol: cyclopropene fatty acids | prepressed solvent or limit amount of feed |
| Soybean meal | trypsin inhibitor: lectins | heat treatment |
| Ground nuts | trypsin inhibitor | limit amount of feed |
| Leaves of legume plants | mimosine | limit amount of feed |
Most all plant protein source contain phytic acid which can interfere with both mineral and protein availability. This problem must be eliminated by special processing techniques to make them of maximum nutritional value.
3.4 Some typical formulated feeds for freshwater prawn
Under natural conditions, freshwater prawns are believed to be omnivorous, feeding on various plant and animal materials, including detritus, grass roots, insect larvae and fishes.
3.4.1 Larval food
Live food, such as Artemia nauplii, is a high quality of larval food. But in some countries, the eggs of this brine shrimps are expensive and frequently in short supply. Zooplankton may be used instead of Artemia nauplii. Some workers have suggested that the other organisms can be used as live food (Moina, Daphnia, or Brachionus) for M. rosenbergii larvae. Prepared feed is also used for larval stage with the following typical combination:
small amount of vitamin A + D, B complex, and C (ascorbic acid)
All ingredients are mixed together in a blender and then steamed until cooked. This feed should be kept in a refrigerator where it can stand for two days.
3.4.2 Food for juvenile stage
Usually, farmers hold the juveniles in tank or nursery pond before release to growout ponds. One of the series of formulated feeds for juvenile is:
| Ingredient | Percent |
| Fish meal | 20 |
| Soybean meal | 9 |
| Rice bran | 45 |
| Coconut oil cake | 20 |
| Tapioca | 5 |
| Pfizer premix or aquamix | 1 |
The vitamin or premix is optional depending on the availability of the natural food in the tank or nursery pond.
3.4.3 Feed for stock in growout ponds
Supplemental feeding program is important in intensively culture or in ponds with poorly conditions. The typical formula for stock in growout pond is as follows:
| Ingredient | Percent |
| Shrimp head meal or fish meal | 20 |
| Rice bran | 40 |
| Coconut oil cake | 20 |
| Tapioca | 20 |
The vitamins may be included in the diet according to condition of pond and cultural techniques.
4. FEED PROCESSING AND STORAGE
Compared to other livestock feeds, fish feeds are unique in that the particle size of the feeds is critical. Also the feed is subjected to water during feeding. The feeds are not chewed completely by the fish as are those for other animals. All these factors must be considered when making fish feeds.
In relation to this consideration, the feed formulator must know the general processing principles in fish feeds:
Packaging and storage
4.1 Grinding or particle size
The objectives of grinding are:
for efficient feed mixing
to improve pellet quality (if processed in pellet form)
to facilitate and increase the ingredient acceptance and ingestion by fish, and
to increase the digestion and utilization of feeds
4.2 Weighing and mixing
Accuracy in weighing is essential to ensure that the feed ingredients are in the proper proportion as formulated in the diet. The size and accuracy of scales must be suitable for the job.
The mixing operation is the most important one in feed processing. Addition of a number micro-ingredients such as vitamins, oil, to the fish feed and the mixing operation has increased in complexity. A uniform mixture is necessary for each unit of feed to contain all nutrients in the proper ratio.
4.3 Pelleting of fish feed
Formulated feed can be prepared in several forms, depending on the stage of the fish. The larval feed is usually prepared in suspension form; for juveniles in crumble or granule form; and for stock in growout ponds in paste or pellet form. The advanced form of fish feed is in pellet form. After mixing, the feed goes to the pellet mill where steam in small amount (less than 6%) is delivered in constant pressure. Water stability of the pellet is important because pellet should remain intact in water before pellet is ingested. Some workers suggested that tests for water stability for crustacean diets should be done for at least 24 hours. The water stability depends primarily on the binding material included in the prawn diets. The binding material used in prawn diet for practical purposes are hydrocolloid materials such as starch, wheat flour, etc.
4.4 Sizing of fish feed
If the feed must be processed in pellet form, the sizing of pellet is very important according to the prawn size. In the most commonly used pellet size is approximately 3 mm in diameter by 6 to 10 mm in length. Smaller pellets, crumbles or granules are available for juvenile or postlarvae.
4.5 Packaging and storage of fish feeds
To protect feed quality, packaging materials should be multi-wall bags or have plastic liners to retard moisture uptake and help protect flavor, aroma and color, particularly for tropical areas.
Low relative humidity is important because moisture enhances mold growth, and attracts insects. Molds which may produce aflatoxin and other toxins grow when more than 13 percent moisture is present. High temperature may cause rancidity of oils and deterioration of vitamins in fish feeds. Rancid oil can be toxic, may destroy some nutrients and will cause off-flavor of feeds.
Generally, 90 days is the maximum storage time recommended for fish feed storage in ambient temperature. Moist fish feed should be kept in refrigerator.
5. FEEDING
Feeding is one of the most important steps in rearing of prawn larvae or prawn culture. Prawn culturist must consider several factors related to feeding programme including the following:
Water temperature and environmental factors
6. REFERENCES
Adisukresno, S. and A. Purnomo. 1977 Mass production of Macrobrachium rosenbergii fry in Indonesia. First Asean Meeting Experts on Aquacult., Technical Rep., Semarang, Indonesia, 31 Jan. to 6 Feb. 1977: 55–67.
Manik, R. 1976 Preliminary studies on the effect of different pelletized formulated feeds on the growth of Macrobrachium rosenbergii. Bull. Shrimp Cult. Res. Centre, Jepara, Vol. 2 (1 & 2) July 1976: 187–193.
Manik, R., K. Mintardjo and S. Adisukresno. 1977 Potential protein sources of supplementary feeds formulated for shrimps and prawns in Jepara. Bull. Brackishwater Aquacult. Dev. Centre, Jepara, issued August 1979, Vol. 3 (1 & 2) Jan.-July 1977: 223–226.
National Research Council. 1977 Nutrient requirements of fishes, Nat. Acad. Sci. 1977: 78p.
New, M.B. 1976 A review of dietary studies with shrimp and prawns. Aquaculture 9: (1976) 101–144.
Orme, L.E. 1970 Trout feed formulation and development. Rep. of 1970 Work. Fish Feed Tech. and Nut., US Dep. Int. Fish and Wildlife Service.
Stephenson, E.L. 1972 Processing feed to destory natural toxins and inhibitors. Effect of processing on the nutrient value of feeds, Proc. Symp. Gainesville, Florida, Jan. 11–13, 1972.
Stickney, R.R. and R.T. Lovell. 1977 Nutrition and feeding of channel catfish. Southern Cooperatives Bull. 218, October 1977. 66p.
Anonymous. 1978 Fish feed technology, Lectures presented at the FAO/UNDP Training Course in Fish Feed Technology, held at the Coll. Fish., Univ. Wash., Seattle, Washington, 9 Oct. to 15 Dec. 1978.
by
A. Sunaryanto1 and K. Mintardjo2
1. OXYGEN AND BIOCHEMICAL DEMAND
Oxygen is required by organisms for their respiration. For its activities in extended period, approximately 3.0 ppm dissolved oxygen (DO) is needed by fish. Shrimp/prawn generally need higher oxygen in the water. Oxygen present in the water come from diffusion from air and as a result of photosynthesis in natural waters.
Biochemical oxygen demand (BOD) is a test to know the rate of removal of O2 by organisms using the organic matter in water. It is especially useful as an index of organic pollution.
There are two methods for the analysis of D.O.:
Winkler or Iodometric method and its modifications (also called titrimetric method) which is based on the oxidizing property of D.O.
Membrane electrode procedure, a method based on the rate of diffusion of molecular oxygen across a membrane.
Titrimetric method is the one most precise and reliable procedure for D.O. analysis.
1.1 Procedure of analysis
a. Collection of sample
Collect the sample in a narrow-necked glass-stoppered bottle of 250– 300 ml capacity (standard BOD bottle) by means of an apparatus designed to avoid the entrapment or absorption of any oxygen from the atmosphere. Note the temperature.
1 Fishery Biologist, Brackishwater Aquaculture Development Center, Jepara, Indonesia.
2 Chief, Shrimp Culture Section, Brackishwater Aquaculture Development Center, Jepara, Indonesia.
1.2 Titration
The method given in this manual is standard winkler method with sulphamic acid modification. The procedures are as follows:
Remove the stopper and add 1 ml of sulphamic acid solution. Replace stopper and mix by inverting bottle for 30 seconds.
Add 1 ml of manganous sulphate solution and 3 ml of alkaline potassium iodide solution. Mix by rapid inversion for 30 seconds. Allow the precipitate to settle (for seawater, alternately shake and allow to settle for a 10-minute period).
Add 1 ml of concentrated sulphuric acid and mix by shaking.
Measure a volume of the sample, carefully pour the measured sample into an Erlenmeyer flask.
Titrate with standard thiosulphate until the sample become a faint yellow. Add about 2 ml of starch indicator and continue the titration until the blue colour disappears.
Calculation:

1.3 The use of D.O. meter
At present the only D.O. meter available at our center is the YS I model 51 B. It is a simple and portable instrument, suggested to be used for daily measurement of field. With a proper calibration, the instrument has an accuracy better than 0.2 ppm. Procedure for operating can be read at the rear part of the instrument.
1.4 B.O.D. measurement
If the D.O. content of the original sample is very low, the sample should be aerated for 5 to 10 minutes.
Place portions of sample into 2 BOD bottles, immediately determine the D.O. concentration in one bottle.
Incubate the remaining bottle in the dark at standard temperature (e.g. 20°C) or at the temperature of the original sample for 1 to 5 days.
Determine the remaining D.O.
Subtract the value from the original value of D.O. to give B.O.D.
2. FREE CARBON DIOXIDE
Carbon dioxide is present in the air at concentrations of approximately 3 parts per 10 000. Consequently, pure water at 1 atmosphere air pressure at 25°C contains only 0.4 ppm free CO2 when in the equilibrium with CO2 in the air. CO2 concentrations are also built up because of release of CO2 by respiration and decomposition. Prior to the analysis of free CO2, the pH of the water sample should first be determined. Waters more acid than 5.5 are also caused by the content of strong mineral acids.
2.1 Procedure of analysis
Pretest two small water samples of approximately 20 ml by adding several drops of methyl orange to one and several drops of phenolphthalein to the other. If the sample is acid (red) to methyl orange, acidity is due to acids stronger than CO2. If the sample is alkaline (yellow) to methyl orange and acid (colourless) to phenolphthalein, the acidity is assumed to be due to carbon dioxide. In the latter case:
Carefully pipette 200 ml of the water sample into an Erlenmeyer flask.
Add 10 drops of phenolphthalein (the solution should remain colourless).
Titrate to a faint pink (approximately pH 8.3) with standard sodium carbonate.
(4) Calculation:

3. HARDNESS
Hardness is principally the total of soluble Ca and Mg salts present in the water, expressed as its CaCO3 equivalent. Hardwater (< 120 ppm Ca CO3) plus other unidentified factors are believed to be the cause of calcium plaque deposits an appendage and body of larvae. Typically, crystal matrix formation begins at the base of the appendages and spreads proximately into the body of the animal. Hardwater disease has been associated with heavy larval mortality.
3.1 Procedure of analysis
Pipette 50 ml water sample into a 250 ml Erlenmeyer flask.
Add 1 ml buffer (to keep the pH at approximately 9–10) and mix.
Add 4 drops indicator Eriochrome black T.
Titrate with standard sodium EDTA. At this end point, the solution should be clear and should change from wine-red to pure blue.
Calculation:
If 1 ml standard Na - EDTA = a mg Ca CO3,

4. NITROGEN
In its various forms especially ammonia and nitrite it can deplete dissolved oxygen level in receiving waters, stimulate aquatic growth, exhibit toxicity toward aquatic life, affect chlorine disinfection efficiency, present a public health hazard, and affect the suitability of wastewater for reuse. Documentation of mortality in prawn larvae in production culture systems due to ammonia and nitrite toxicity is lacking. Experimental 96 hour LC 50 for ammonia at pH 8.3 is reported to be 14 ppm. The 192 hour LC 50 for nitrite has been shown to be 4.5 ppm. Ammonia toxicity is one of the suspected causes of larval mortality of Macrobrachium rosenbergii. Since ammonia is the principal excretory product of most crustaceans, its potential increase within more intensified culturing systems has been suspected to slower larval development and lower production.
4.1 Procedure of analysis
4.1.1 Ammonia
The method given in here is Nesslerization procedure of the Colorimetric method, which has an adeuqate accuracy within ammonia concentration of 20/mg/1 to 5 mg. The most reliable results are obtained in fresh samples. In the event that a prompt analysis is impossible, add 0.8 ml concentration H2SO4/1 sample and store at 4°C to preserve the ammonia concentration. Neutralize sample acidity with NaOH or KOH immediately before making the determination.
a. Pretreatment
Add 1 ml ZnSO4 solution to 100 ml sample and mix thoroughly.
Add 0.4 to 0.5 ml NaOH solution to obtain a pH of 10.5 as determined with a pH meter, mix thoroughly.
Let the treated sample stand for a few minutes, whereupon a heavy flocculent precipitate should fall, leaving a clear and colourless supernate.
Filter sample, discarding the first 25 ml filtrate.
b. Colour development
Use 50.0 ml sample or a portion diluted to 50.0 ml with ammonia free water in a Nessler tube.
Add 1 drop EDTA or 1 to 2 drops potassium sodium tartrate (if sample contains sufficient concentration of calcium, magnesium, or other ions that produce turbidity or precipitate with Nessler reagent.
Add 2.0 ml Nessler reagent if EDTA reagent is used, or 1.0 ml Nessler reagent if potassium sodium tartrate is used.
Mix by inverting the tube at least six times. Let the reaction proceed for at least 10 minutes.
c. Visual comparison
Compare the colours produced in the sample against those of the ammonia standards. Prepare a series of visual standards in Nessler tube by adding the following volumes of standard NH4 Cl solution and diluting to 50 ml with ammonia free water: 0.0, 0.1, 0.2, 0.3, 0.4 and 0.5 ml (1 ml of standard NH4Cl contains 0.1 mg NH3).
4.1.2 Nitrite
Since nitrites may be oxidized readily to nitrates, sample of water should be analyzed promptly. The sulfanitic acid modification method is used.
Pour 50 ml of water sample into a Nessler tube.
Add ± 0.5 gr or one small spoon of a mixture of tartar acid, sulfanilic acid, and alpha naphtylamine (90:10:1). This gives a pink to read colour within 10 minutes if nitrite is present.
Compare with standard dilute from the standard NaNO2 solution (1 ml= 0.005 mg N - NO4) similarly treated.
| Number of ml in standard NaNO2 | ppm N when diluted to 50 ml |
| 1 | 0.01 |
| 2 | 0.02 |
| 3 | 0.03 |
| 4 | 0.04 |
| 5 | 0.05 |
5. REFERENCES
APHA - AWWA - WPCF 1975 (14th Ed.) Standard methods for the examination of water and wastewater.
Golterman, H.L. and R.S. Clymo. 1971 (Rev) Method for chemical analysis of freshwaters. IPB Handbook No. 8. Blackwell Scientific Publications, Oxford and Edinburgh
Swingle, H.S. 1969 (Rev) Method of analysis for waters, organic matter, and pond bottom soils used in fisheries research in Auburn University.
by
M. Sachlan1
1. INTRODUCTION
It is a well-known fact, that about 90 percent of the crustacean larvae in nature which live as mero-plankton are lost due to the lack of food or consumed by other animals. Specially for the larvae of Macrobrachium, it is really a struggle for life to become a mature animal.
According to Ling, there are 8 (eight) larval stages and 11 (eleven) ecdysis for the larvae to become juvenile stage, where every larval stage has their own kind of food; in other words, they have to change their food during the successive larval stages.
The fecundity of Macrobrachium, according to the data from Palembang (South Sumatra) vary from 20 000 to 96 000 eggs. The eggs hatch in fresh or brackishwater, but the first ecdysis which occurs four days after hatching takes place in brackishwater with a salinity of 8 to 15 ppt.
Therefore, to reduce the mortality of the larvae in nature, a special hatchery and a special food for every larval stage have to be provided; otherwise, the larvae will lose in the new environment. But there are also new problems with man-made hatchery.
In the hatchery the larvae are cultivated until juvenile in a small environment in comparison with the sea or river. The ecological conditions between those two environments are quite different, and a little mistake in the handling of the hatchery, will be fatal for the larvae, e.g. the larvae will be a victim of a parasite or a lack of oxygen. The important things in hatchery is a hygienic rearing method for the larvae in order to avoid parasites or a bad condition, so the larvae will grow up until juveniles.
The purpose of this paper is to summarize what is known as the parasites and pests of the Macrobrachium sp and their control; not only occuring in the hatcheries, but also in the brackishwater ponds and in the open waters.
2. SYSTEMATIC LIST OF THE PARASITES AND PESTS
2.1 Virus
Cause several diseases in the thoracic region.
1 Ex-Director of the Inland Fisheries Research Institute, Bogor, Indonesia.
2.2 Bacteria
Vibrio, Pseudomonas, Aeromonas: Attack the gills and cause red spots on them. While other bacteria are chitinovorous, attacking the edges of exoskeletal parts; the bacteria sometimes breaks open the chitin and enter the body.
According to Nurjana, this disease is found also in broodstock, that were kept in concrete tanks.
2.3 Fungi
Lagenidium, Serolipidium, Dermocystidium and Leucothris mucor; specially the last species can cause the dangerous black-gill diseases of the prawn in the ponds. The three other species cause the skin disease (mycosis) of the penaeid nauplius, until become immobile and settle to the bottom of the tank until they die. All the four species of the fungi belongs to the class: Phycomycetes. As we know, Saprolegnia and Achlya belongs also to the Phycomycetes, which attack the freshwater fishes and their eggs; these species are saprophytic.
2.4 Algae
Licmophora, Nitzchia, Skeletonema, Chaetoceroas (Diatoms) and blue-green algae (Cyano-phyta). According to Nurdjana, Licmophora as an epibiont attach to the larvae of zoea stage, after metamorphosis, 30 specimens can attach a zoea and hinder the movement of the larvae until it die. In the BADC, Jepara, Licmophora usually occurs in June and August. Specially the species L. abbreviata attach to the shrimp larvae in the Philippines. Blooming of Skeletonema costatum, Nitzschia closterium and Chaetoceras calcitrans as food is very harmful for the larvae especially under state of “crowding”.
It causes overfeeding of the larvae, especially for the zoea stage of penaeids, and will result in a long stiff faeces behind the larvae which can entangle the body of the larvae and cause much mortality. For an example, the blooming of C. calcitrans could attain a density of 6 million specimens per cc which will result in increase of the pH above 8.5 and causes high mortality. The blue-green algae, usually consist of the genera Phormidium and Oscillatoria that attach to the crustacean and prevent the molting process.
2.5 Protozoa
Zoothamnium (Ciliata, Peritricha, Vorticellidae-coloni)
Epistylis (Ciliata, Peritricha, Vorticellidae-coloni)
Vorticella (Ciliata, Peritricha, Vorticellidae-solitair)
Acineta (Ciliata, Suctoria)
Gregarina (Sporozoa)
Normally the 4 genera of Ciliata are epibiont and according to Nurdjana the ciliate attach to the larvae, especially of Macrobrachium.
When they attach on the gills of the penaeid larvae, they cause high mortality. The ciliate occurs in larval rearing tanks, where the larvae are overfed with Brachionus plicatilis (Rotatoria). Gregarina belongs to the class: Sporozoa which attacks the guts of the earthworm and shrimps. It is interesting to know that these genera of the ciliate also attach to the Lernaea cyprinacaea, an ecto-parasite (Copepoda) of freshwater fishes.
2.6 Coelenterata
Medusae stage of Hydrozoa. They prey directly on the post-larval shrimp according to Nurdjana. The occurrence of this undesirable creature is due to the bad water filter system of the hatchery.
2.7 Diptera (Hexapoda)
The larvae of this chironomids species: Tendipes longilobus is also undesirable, but is not harmful to the shrimps larvae; when this chironomids larvae are in great quantities they can create oxygen-carbon, carbon dioxide tension during the evening, which will lead to larval suffocation.
2.8 Copepoda (Calanus sp, Balanus sp)
The author has seen a blooming of Calanus sp, in the larval pond of P. monodon, and can reduce the production of the larvae. The ponds are fed with soybean cake (extract). The Calanus is known as a pest among the zoo-plankters. Balanus larvae attack the broodstock. If this fouling animal is removed, a hole may be created on the exoskeleton or the shrimp. Balanus belongs to the sub-order Cirripedia.
2.9 Isopoda
It is well known, that many genera of this Malacostracan, are parasites of the crustaceans and fishes. In 1977 and 1978 there was an epidemic of Isopoda on the gills of Macrobrachium (according to M. Nasjiruddin, Ar) near Palembang (South Sumatra). The length of the infected Macrobrachium is between 5 to 13 cm, and the specimens are smaller in comparison with the specimens free from the isopod. The author has seen several freshwater river prawns infected with isopoda, and also freshwater and seawater fishes, the Puntius binotatus and Stromateus cinereus infected by Ichthyoxenus jellinghausii and Cymothoi stramotei. Usually the parasites, who live between the gills and carapace are dorsoventrally depressed. Isopods of the sub-order Epicarida are all parasites on prawns and crabs.
2.10 Helminthes
Nematoda, and especially the genus Thynnascaris and also the Cestoda, the genus Opecochilus, according to the literature are also parasitic of the penaeids.
3. CONTROL OF THE PARASITES AND PESTS
The effective methods of controlling the pest and diseases of prawns, are as follows:
Therapeutic measures
To avoid undesirable organisms in the hatcheries or ponds such as: Medusae, Calanus, it is advisable to use the best of the several kinds of filters, e.g. sand or screen filters, etc. But to avoid diseases of the larvae and broodstock caused by bacteria or fungi, it is suggested to use the following measures:
To clean the broodstock by bathing for a few minutes in freshwater containing one of the following chemicals and well aerated. After bathing the prawn should be cleaned with running water.
| Chemical | Concentration | Time |
| 1. Malachite green | 0,5 ppm | 15–20 minutes |
| 2. KMnO4 | 50 | 15–20 minutes |
| 3. Formaline | 50 | 15–20 minutes |
| 4. Ca-hypochloride | 15 | 15–20 minutes |
| 5. Furanace | 10 | 15–20 minutes |
To clean the larvae before stocking in the rearing ponds, treat with one of the following chemicals:
| Chemical | Concentration | Time |
| 1. Streptomycin | 1,5 – 2 ppm (to avoid diseases) | 20 minutes |
| 2. CuSo4 | 0,6 ppm (as curing method) | 20 minutes |
In the case of competitors prevailing in the hatchery, e.g. blooming of Licomphora and other diatoms as food (Chaetoceras, Skeletonema, Nitzschia), we must apply the prophylactic and strengthening methods. According to Nurdjana, the reducing of the quantity of Licomphora density is done by covering the rearing tanks with bamboo-screen, but the result in this case is not satisfactory. Other methods to strengthen the larvae is by feeding with a dense culture of Chlorella (1 million cells per cc) to filter out the sunlight; the result is more satisfactory. Another method is to give the larvae a high-protein food like soybean, to accelerate the growth of the larvae, so that by ecdysis, the diatom with the silicate cell wall will settle down at the bottom of the tank and could be cleaned by siphoning; this method gives more satisfactory result. When the larvae are attacked by ciliate, such as Zoothamnium and others, it should be treated by the therapeutic method. The treatment is by using quinine-bisulphate at 1 ppm and after the third treatment, nearly all ciliates will die. Other better treatment is with the application of 1 ppm CuSO4. The infection of Lagenidium in the nauplius-zoea of penaeid larvae could be controlled by application of 0.05 ppm malachite green for good result while the medusa of Hydrozoa and chironomids larvae can be reduced with 0.5 ppm of the same chemical.
In the case of Isopod-infection, as in Pelembang, South Sumatra province, no curing methods are known, but the author has experience with river-prawns infected by isopod. The isopod can be taken away with the pincer or forcep carefully and this method does no harm to the prawn.
4. REFERENCES
Fritsch, F.E. 1948 The structure and reproduction of the algae. Vol. 1 Cambridge, the Univ. Press.
Frederick, A.W. and T.W. Frederik. 1949 The fungi. Vol. 1. New York: John Wiley & Sons, Inc.
Haniah, H.S. and A. Ismail. 1980 Reproduction-technic of the Macrobrachium rosenbergii de Man (in Indonesian) Working paper
Nurjana, M.L. and B. Martosudarmo. 1978 Observation on diseases affecting shrimp at the Brackishwater Aquaculture Development Centre, Jepara, Indonesia. Working paper at the First Workshop on Tropical Fish Diseases.
Nasjiruddin, A.R. Parasite of Macrobrachium rosenbergii in the open waters in South Sumatra (in Indonesian) Working paper. Inl. Fish. Res. Station, Palembang
Ling, S.W. 1969 The general biology and development of Macrobrachium rosenbergii de Man. FAO Fish. Rep. 3(57)
Ondara. 1974 The fecunditas of Macrobrachium rosenbergii de Man in South Sumatra. Report No. 14. Inl. Fish. Res. Station, Bogor
Sachlan, M. 1973 Notes on parasites of freshwater fishes in Indonesia. Contributions of Inl. Fish. Res. Station, Bogor, No. 2
Sachlan, M. 1978 The occurrence of fish parasites in the Indonesian waters. Working paper at the First Workshop on Tropical Fish Diseases.
Ward, H.B. and G.C. Whipple. Freshwater biology, edited by W.T. Edmonson, New York London, John Wiley & Sons, Inc.
by
Sukotjo Adisukresno1
1. INTRODUCTION
Prawn farming generally is practiced by the small holder farmers, who usually own a small area of pond. The industry of prawn farming has not fully developed except only in some regions like Hawaii and Thailand, where it has been developed to some extent. In Thailand, over 30 commercial farms of prawn have been established. Since it is an industry which spends a lot of money for construction, operational costs, etc., all of the expenditures and benefits should be accounted carefully. For small holders, most operations are unrecorded. What the farmer do is to make notes on how much money is spent for the preparation and operation of his farm and how much money he earns, so that he can find how much profit he gets but since it is family business, usually does not take into account the salary for the workers (his family), which should be recorded as expenditures.
Method of evaluation of the farm business is discussed in this paper.
2. METHOD OF EVALUATION
2.1 Pay back period
Pay back period method based on the duration of a farm/business to recover its capital outlay.
| P | = | |
| P | = | pay back period in year |
| C | = | capital outlay |
| E | = | yearly income before depreciation and after tax service |
1 Director, Brackishwater Aquaculture Development Centre, Jepara, Central Java, Indonesia
For example:
There are six farms which have the same capital outlay, but their yearly income are different, it is of interest to determine which farm has highest benefit from the data presented below:
| Farms | Capital outlay | I N C O M E | ||
| Year 1 | Year 2 | Year 3 | ||
| A | 10 000 000 | 4 000 000 | 5 000 000 | 5 000 000 |
| B | 10 000 000 | 5 000 000 | 5 000 000 | 5 000 000 |
| C | 10 000 000 | 3 000 000 | 5 000 000 | 7 000 000 |
| D | 10 000 000 | 6 000 000 | 6 000 000 | 6 000 000 |
| E | 10 000 000 | 6 000 000 | 5 000 000 | 5 000 000 |
| F | 10 000 000 | 7 000 000 | 6 000 000 | 4 000 000 |
The pay back period relative rating for the above farms are indicated below.
| Pay back period (year) | Grade/number |
| 2 1/5 | 5 |
| 2 | 4 |
| 2 2/7 | 6 |
| 1 ⅔ | 2 |
| 1 4/5 | 3 |
| 1 1/2 | 1 |
The disadvantage of this method is that the inflation rate is not taken into account.
2.2 Proceeds per dollar outlay
This method is one step better than the pay back period, since the total income during the life period of the business is accounted. The calculation is based on the comparison between the total income during the period of the business and the total capital outlay.
| Farms | Capital outlay | Total income | T.I. | Grade number |
| T.C. | ||||
| A | 10 000 000 | 14 000 000 | 1.4 | 5 |
| B | 10 000 000 | 15 000 000 | 1.4 | 4 |
| C | 10 000 000 | 15 000 000 | 1.5 | 4 |
| D | 10 000 000 | 18 000 000 | 1.8 | 1 |
| E | 10 000 000 | 16 000 000 | 1.6 | 3 |
| F | 10 000 000 | 17 000 000 | 1.7 | 2 |
Likewise, the disadvantage of this method is that inflation rate is not taken into account.
2.3 Average rate of return
This method is almost the same as the proceeds per dollar outlay. Total income during live period divided by the live period (in year) multiplied 100% and then the result is divided by total capital outlay.
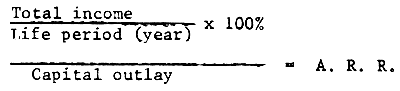
Sample: Farms A
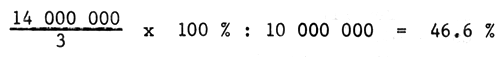
| Farms | Capital outlay | A.R.R Total income | Grade (%) | number |
| A | 10 000 000 | 14 000 000 | 46.6 | 5 |
| B | 10 000 000 | 15 000 000 | 50 | 4 |
| C | 10 000 000 | 15 000 000 | 50 | 4 |
| D | 10 000 000 | 18 000 000 | 60 | 1 |
| E | 10 000 000 | 16 000 000 | 53.3 | 3 |
| F | 10 000 000 | 17 000 000 | 56.6 | 2 |
Likewise, the disadvantage of this method is that the inflation is not taken into account.
2.4 Net present value
With this method, all of the benefits are evaluated at the beginning of the establishment, so that the value of the benefit is standardized. The benefit at the end of the first year is multiplied by present value factor of the first year, the benefit at the end of the second year is multiplied by present value factor of the second year and so on. Present value factor also called as the discounted factor, and there is available table of discounted factor which is arranged from 1 to 50 percent for one up to 50 years.
In case no table is available, the discounted factor can be calculated with the use of a suitable calculator.
For example, d.f. at 12% interest:
And so on, just push the button = to find out the following d.f.
Above is based on the following formula:
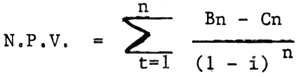
| Where: | Bn | = | Benefit |
| Cn | = | Cost | |
| n | = | Period (year) | |
| i | = | Interest rate (%) |
Some data is analyzed with N.P.V., for example the interest rate is 12%.
Farms A
| 1st year | 4 000 000 × 0.8929 | = 3 571 600 | |
| 2nd year | 5 000 000 × 0.7972 | = 3 986 000 | |
| 3rd year | 5 000 000 × 0.7118 | = 3 559 000 | |
| Total discounted benefit | 11 116 600 | ||
| Cost | 10 000 000 | ||
| Net present value | 1 116 600 | ||
With the same method, calculate the net present value of Farms B, C, D, E, and F, as shown below.
Net present value at 12% of six farms
| Farms | Total discounted benefit | Discounted cost |
N.P.V. | Grade number |
| A | 11 116 600 | 10 000 000 | 1 116 600 | 5 |
| B | 12 009 500 | 10 000 000 | 2 009 500 | 4 |
| C | 10 223 700 | 10 000 000 | 223 700 | 6 |
| D | 14 511 400 | 10 000 000 | 4 511 400 | 1 |
| E | 12 902 400 | 10 000 000 | 2 902 400 | 3 |
| F | 13 880 700 | 10 000 000 | 3 880 700 | 2 |
This is more realistic than the other methods
2.5 Internal rate of return
The main difference between IRR and NPV is that in IRR, the interest rate should be computed; it has been known in NPV. IRR is the interest rate which make the discounted benefit same as the discounted cost.
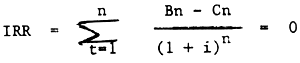
| Where: | Bn | = Income |
| Cn | = Cost | |
| n | = Period (year) | |
| i | = Interest rate (%) | |
| Benefit = Cost - break even point | ||
Data needed:
Income (output) table
Input table
Cash flow table
IRR analysis
INCOME TABLE
| Remarks | Y E A R | ||||
| 1 | 2 | 3 | 4 | 5 | |
| a) Production of prawn (in kg) | 5 000 | 6 000 | 6 500 | 7 000 | 7 200 |
| b) Price unit (kg) | 2 000 | 2 200 | 2 800 | 3 000 | 3 200 |
| c) Total price (× 1 000) | 10 000 | 13 200 | 18 200 | 21 000 | 23 040 |
INPUT TABLE
(× 1 000)
| Remarks | Y E A R | ||||
| 1 | 2 | 3 | 4 | 5 | |
| a) Investment | 8 000 | - | - | - | - |
| b) Operational cost | 4 000 | 5 000 | 6 500 | 7 200 | 8 500 |
| c) Interest 20% | 2 400 | 1 400 | 200 | - | - |
| d) Principal* | 5 000 | 6 000 | 1 000 | - | - |
| e) Depreciation 20% | 1 600 | 1 600 | 1 600 | 1 600 | 1 600 |
| Total input | 21 000 | 14 000 | 9 300 | 8 800 | 10 100 |
*See cash flow table (data from cash flow table)
CASH FLOW TABLE
(× 1 000)
| Remarks | YEAR | ||||
| 1 | 2 | 3 | 4 | 5 | |
| Cash inflow: | |||||
| Sales | 10 000 | 13 200 | 18 200 | 21 000 | 23 040 |
| Loan | 12 000 | - | - | - | - |
| Total cash inflow | 22 000 | 13 200 | 18 200 | 21 000 | 23 040 |
| Cash outflow: | |||||
| Investment | 8 000 | - | - | - | - |
| Operational cost | 4 000 | 5 000 | 6 500 | 7 200 | 8 500 |
| Total cash outflow | 12 000 | 5 000 | 6 500 | 7 200 | 8 500 |
| Cash balance before depth service | 10 000 | 8 200 | 11 700 | 13 800 | 14 540 |
| Loan at beginning of year | 12 000 | 7 000 | 1 000 | - | - |
| Interest 20% | 2 400 | 1 400 | 200 | - | - |
| Principal | 5 000 | 6 000 | 1 000 | - | - |
| Loan at end of year | 7 000 | 1 000 | - | - | - |
| Cash balance after depth service | 2 600 | 800 | 10 500 | 13 800 | 14 540 |
| Cummulated cash | 2 600 | 3 400 | 13 900 | 27 700 | 40 240 |
| Year | Output | Input | Benefit | N.P.V. at | |||
| 40% | 50% | ||||||
| D.F | N.P.V. | D.F | N.P.V. | ||||
| 1 | 10 000 000 | 21 000 000 | 11 000 000 | 0.714 | -7.854 | 0.666 | -7.326 |
| 2 | 13 200 000 | 14 000 000 | -800 000 | 0.510 | - 408 | 0.444 | - 355 |
| 3 | 18 000 000 | 9 300 000 | 8 900 000 | 0.364 | 3.239.6 | 0.296 | 2.634 |
| 4 | 21 000 000 | 8 800 000 | 12 200 000 | 0.260 | 3.172 | 0.197 | 2.403 |
| 5 | 23 040 000 | 10 100 000 | 12 440 000 | 0.186 | 2.406 | 0.138 | 1.708 |
| 555 | - 936 | ||||||
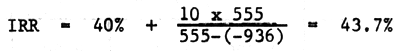
If the common rate of interest is lower than the IRR - profitable.
Benefit cost ratio Using the same data above:
| Year | Output | Input |
| 1 | 10 000 000 | 21 000 000 |
| 2 | 13 200 000 | 14 000 000 |
| 3 | 18 200 000 | 9 300 000 |
| 4 | 21 000 000 | 8 800 000 |
| 5 | 23 040 000 | 10 100 000 |
| Total | 85 440 000 | 63 200 000 |
| Average | 17 088 000 | 12 640 000 |
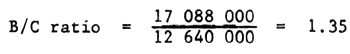
It means it is profitable.
Financial rate of return Same data as the benefit cost ratio
I.R.R
| Year | Investment | Income | Operational cost | Benefit |
| 1 | 8 000 000 | 10 000 000 | 4 000 000 | 6 000 000 |
| 2 | - | 13 200 000 | 5 000 000 | 8 200 000 |
| 3 | - | 18 200 000 | 6 500 000 | 11 500 000 |
| 4 | - | 21 000 000 | 7 200 000 | 13 800 000 |
| 5 | - | 23 040 000 | 8 500 000 | 14 040 000 |
| Total | 8 000 000 | 54 240 000 | ||
| Average | 10 848 000 |
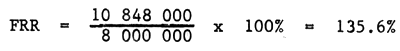
Remarks
Items which may be recorded and accounted in the Macrobrachium farms:
Miscellaneous expenses