Deterioration or spoilage is due mainly to the multiplication of minute organisms called bacteria in the flesh of the fish. These bacteria putrify the fish liberating a bad smell and causing the fish to soften, spoil and become unacceptable. All methods of preservation are in essence an attempt to reduce or limit this multiplication of bacteria between the time when the fish is caught and when it is consumed.
Certain basic facts should be understood:
A living fish has bacteria in its guts and to a less extent in its gills but not in its flesh. When the fish dies the bacteria quickly invade the flesh. Therefore, the sooner the guts and gills are removed the better. When they are removed the fish must be washed so that the smallest possible number of bacteria from the guts spread to the flesh. Time is therefore important.
Bacteria multiply faster in a warm than in a cool environment. Chilling or cooling will limit bacterial reproduction. The temperature at which fish are kept is therefore important.
Bacteria cannot multiply where there is no moisture and so drying limits bacterial reproduction.
Smoke, salt and other preservatives (e.g., vinegar) reduce or prevent bacterial reproduction.
Because all preservation methods are based on limiting the total number of bacteria, it is necessary that all containers, utensils and other gear used in preparing, transporting or storing fish should be kept thoroughly clean. If they contain traces of fish from the time they were previously in use they will also be heavily contaminated with bacteria which will hasten spoilage of the new catch.
The production of fish serves to improve health by providing protein and other nutrients which are frequently deficient in African diets. Fish ponds and dams are therefore important in reducing malnutrition and allied illness, but if not properly managed they may also contribute to ill health. Consequently it is desirable that those concerned with building or managing them should be aware of ways to minimize this danger. The two important diseases which are affected by such bodies of water in central east Africa are bilharzia (schistosomiasis) and malaria.
Bilharzia
Bilharzia is a disease of humans who are infected by contact with water containing the minute organisms responsible for initiating the disease. These free swimming organisms, called cercariae, penetrate the skin and enter the blood stream of the infected person. There they eventually grow into worms which live and feed in the blood vessels of the abdomen causing, after a period of time, various symptoms and signs which characterize the disease. They may include the passing of blood in the urine or feces of the infected person. This human excreta contains the worms' eggs which, if they reach water, hatch out to produce larvae or miracidia which must enter the body of a snail if they are to survive. The snails involved are small rather inconspicuous water snails of several different shapes. The larvae undergo change in the snail and eventually leave the snail as numerous free swimming cercariae which are now capable of penetrating the skin of man and thereby infecting him and continuing the cycle. This cycle involving man, human excreta, water and snails is vital to the bilharzia organism. If persons with the disease are treated then there will be no eggs in their excreta. If human excreta is properly disposed of, the eggs cannot hatch to infect another person. By avoiding contact with water containing cercariae man will not become infected. The water of a fish pond or dam containing no snails will not infect man.
Control of the disease depends on various measures designed to break the chain of infection. These include:
Urination and defecation in places where bilharzia eggs cannot reach bodies of fresh water that might harbor snails.
Medical treatment of all persons with the disease so that they no longer pass out eggs in their feces or urine.
Avoiding or minimizing contact with water which may be infected. Water for drinking, bathing and washing clothes should be drawn from water free of infection or which has been drawn 24–48 hours prior to use. The cercariae will die in water which has been allowed to stand for this length of time.
Destruction of snails by clearing away water plants either mechanically or with fish. There are several kinds of fish which eat snails. For example, in dams containing fish of the species Haplochromis, the snail population will be greatly reduced.
Killing snails with certain chemicals or poisons such as with copper sulfate put into the water. These, however, should only be used after consultation with an expert, because some may kill the fish or the organisms on which fish feed.
These points have been stressed because bilharzia is extremely prevalent and is responsible for a great deal of ill health in Africa. The disease is a major public health problem in Rhodesia and to a less extent also in Zambia.
Malaria
Malaria, which causes much illness in large areas of Africa, is spread from man to man by certain mosquitoes. Mosquitoes all breed by laying their eggs in water. Dams and fish ponds are often cited if there are a lot of mosquitoes in an area. The breeding seldom takes place in the open water of a dam or fish pond but rather in small collections of water in the environs of the pond or dam. Breeding may also take place in areas at the edge of the pond where the water is sheltered by vegetation growing in the pond. Another common breeding site is in water which has collected in the footprints of animals that have drunk from a dam. A properly managed pond or dam, especially if stocked with fish that eat mosquito eggs and larvae, will not cause mosquito breeding. It is important, however, to keep the edges and backwaters clear of vegetation and also to see that water does not stand in puddles in the vicinity of the dam or pond. There are insecticides and oils which can be put into water to stop mosquito breeding but these may sometimes be harmful to the fish in the pond or dam. They can, however, be used on the stagnant pools and seepage not connected with the pond or its inflow.
Other malaria control measures not directly related to fish culture include the prevention of mosquito breeding in empty tin cans and holes which collect water, the spraying of insecticides in houses to kill mosquitoes which enter, the screening of houses to prevent the entry of mosquitoes, the medical treatment of all cases of malaria and the regular taking of certain drugs which prevent malaria even in persons bitten by infected mosquitoes.
Every fish culturist should have an adequate knowledge of the chief physical, chemical and biological factors which, in one way or another, influence the production of fish. To this end, it is suggested that observations be made regarding the temperature, oxygen content, pH and alkalinity of the water, as well as the quality and density of plankton and the benthos.
Temperature
Any reliable thermometer, which has a graduation scale from 0.0°C (32°F) to 40°C (104°F) can be used for recording water temperatures. The thermometer is kept at about 6 to 12 in. under the water surface until the mercury comes to a standstill. At that point the temperature is recorded. In pond intake furrows the temperature must be taken near the inlet into the pond. In reservoirs, where the water is more than about 5 ft deep, the temperature of the deep water must also be taken. To that end, a water sampler, as described in the section on “oxygen content,” is used. The water sampler is kept at the required depth for a few minutes. It is then quickly lifted to the surface of the water and the temperature is taken by inserting the thermometer into the water sampling bottle. It is essential that temperatures be taken shortly after sunrise, and again some hours before sunset.
Application
Water temperature is a decisive factor when making the choice of the kind of fish to be reared in a particular place. For instance, temperatures below 11°C (52°F) are not suitable for Cichlid culture but trout can be bred, provided the temperature of the water never rises above 21–24°C (70–75°F). Carp, however, can be bred in both cases. The water temperature also has a great influence on the biological production, determination of the spawning season, etc.
 | Figure 72. Water sample bottle. |
Oxygen content
To ascertain the amount of oxygen (O2) dissolved in a water is a somewhat elaborate proceeding and should be undertaken only by persons who have a basic knowledge of chemistry.
To ascertain the oxygen content in surface water, a water sample bottle is filled by dipping with water from the surface. To determine the oxygen content at depth, however, a water sample is taken by means of a water sampler. In taking samples, the sample bottle should be filled to overflowing and the stopper then pushed in. This is essential to avoid air bubbles under the stopper.
A 500-cc bottle with a glass stopper is used to make a simple water sampler. A weight of about 1½lb, such as a stone or a length of metal, is tied firmly to the lower part of the bottle. A sinking string is tied to the neck of the bottle and, a little higher up, a glass stopper is tied to the same string. The length of the string between the edge of the bottle neck and the stopper must be at least twice the length of the stopper (Figure 72).
To take a sample the stopper is firmly pushed into the bottle neck, so that no water can get into the bottle while it is sinking. The bottle is sunk to the desired depth. Then the stopper is pulled out of the bottle neck by means of a sharp jerk at the sinking string. The bottle starts filling with water while air bubbles appear on the water surface. When the air bubbles no longer appear, the bottle is full. The bottle containing the water sample is then quickly pulled out and closed with the stopper.
To determine the amount of dissolved oxygen the following items are needed:
One 500-cc glass bottle with a glass stopper for water sampling.
The necessary number of 100-cc glass bottles for water samples.
One 50-cc bottle of manganous chloride solution: 100 g pure crystalline MnCl2.4H2O dissolved in 200 cc of distilled water.
One 50-cc bottle of alkaline-iodide reagent: 100 g potassium hydroxide (KOH) dissolved in 200 cc of distilled water plus 60 g potassium iodide (KI).
Three 1-cc graduated pipettes.
A color chart for the identification of the amount of dissolved oxygen.
The determination of the oxygen content in a water sample is conducted in the following way: by means of separate pipettes, first add 1 cc of manganous chloride solution and then 1 cc of alkaline-iodide reagent to the water sample by placing the tip of the pipette in the water and allowing the solution to flow into the bottle. Then the bottle is closed with a stopper and the contents shaken. At this stage, the water sample will turn into shades from white to dark brown in color. The white color indicates the presence of dissolved oxygen. The more dissolved oxygen the water contains the darker is the shade of brown. For practical purposes the interpretation of the value of the coloring is done by means of a special color chart. The coloring of the test water is compared with the color specimen on the chart, on which the value of each shade is indicated.
Application
The oxygen determination discloses the amount of dissolved oxygen in the water, which is needed, for instance, by the fish for breathing. An oxygen content of less than approximately 3 mg per liter will cause the Tilapia to move close to the water surface gasping for air instead of feeding, and prolonged deficiency in oxygen may cause the death of fish. In the deep parts of the water the oxygen content can be very low, or even absent, in which case there will be very few fish or none at all.
Through the intensive growth of submerged plants and phytoplankton, the deficiency in oxygen can be noticeable, especially at night as by then much of the oxygen has been absorbed by the plants.
pH
The pH is a quantitative expression for the acidity or alkalinity of a water. To determine the pH the use of a Hellige glass comparator is suggested, with comparator disks ranging from 5.2 to 8.4. Instructions for use are always included with the set, which consists of the comparator, disks and indicators.
For a simple pH determination litmus paper is used. This is obtainable at chemists in ready-cut blue and red strips. If a strip of blue litmus paper changes its color from blue to red when dipped into water it shows that the pH content of the water is below 7.0 (acid reaction). If a strip of red litmus paper is dipped into water, and the red color turns to blue, the pH content of the water is over 7.0 (alkaline reaction). If the blue and red strips do not change their color in water it means that the pH content of the water is 7.0 (neutral).
Application
The pH scale ranges from 0–14; pH 7 is neutral, below 7 acid and above 7 alkaline. For fish culture the best water is neutral, or a pH slightly on the alkaline side. Generally waters with a pH value of from 6.5 to 8.0 are regarded as “normal waters” for fish culture. Waters with a pH below 5.0 and over 9.0 are usually fatal to the life of fish.
Alkalinity
For the detailed determination of alkalinity of a water some skill and laboratory equipment are needed. It is, therefore, advisable to send a water sample to a recognized laboratory for analysis. Any clean bottle filled with a water sample taken from the surface of the water can be used for this purpose.
The total alkalinity of fresh waters can be as high as 350 ppm, although the figure is generally somewhere between 45 and 200 ppm.
The total hardness of water based on calcium-carbonate is classified as:
very soft water (0–5 ppm)
soft water (5–10 ppm)
medium soft water (10–20 ppm)
medium hard water (20–30 ppm)
hard water (more than 30 ppm)
Application
Hard waters are biologically more productive than soft waters.
Plankton
Plankton are minute organisms floating in the water. The density of plankton is easily seen by measuring the transparency of a water, while the kind of plankton can be recognized, to some extent, by the coloring of the water. For more detailed determination of the quality, a sample is taken by sieving a specific amount of water through a plankton net (Figure 73).
For measuring the transparency of a water, a white painted metal disk of 25 cm (12 in.) in diameter is used (Figure 73). The disk is fixed to a cord and sunk into the water until it disappears from sight. Further sinking is stopped at the moment of disappearance and the depth measured on the sinking cord.
If the water is not very muddy, the limit of transparency generally indicates the density of plankton. The coloring of the water, clearly seen on the white disk, is caused mainly by the presence of plankton. Different kinds of plankton give the water their specific coloring.

Figure 73. A. Plankton net. B. Secchi disk.
To determine the quality and quantity, or composition, of plankton in a water, a sample is taken in the following way. Ten liters (approximately 18 pt) of water are poured through an extremely fine-meshed (No. 20) silk plankton net. The plankton caught in the net is then transferred to a 100-cc wide-necked jar and conserved in a 5 percent formalin solution. This is achieved by adding 40 percent formalin in a quantity of 1/8th of the sample volume.
The following day, the plankton conserved in the jar is poured into a (preferably narrow) graduated cylinder and left there for 24 hours to set. After 24 hours the volume of the set plankton is read from the graduation scale of the cylinder. If in the example given, the graduation scale of the measuring cylinder showed, say, 5 cc plankton, this would mean that the density of plankton in the tested water was 5 cc to every 10 liters of water.
The sample taken for quantitative analysis can be used for the analysis of the quality, or composition, of plankton, or a new sample taken by dragging the plankton net through the water. Plankton so collected is then transferred to a bottle and fixed with 5 percent formalin. A plankton quality determination sample should always be sent to a specialist for analysis.
Application
The analysis taken from different waters in different seasons will give an idea of the plankton production. Plankton is of great value to fish as food, especially in the younger stages of their lives. Some fish are mainly plankton eaters. The amount and quality of plankton indicate the fertility of a water on which fish production, either directly or indirectly, generally depends.
The best waters for fish culture are waters with a greenish color. This indicates that they are rich in green and blue-green algae and are, usually, also rich in zooplankton.
Width and length of ponds in yards for ponds from 1/16 to 1/4 acre in surface area
| Width (yd) Length (yd) | 10 | 15 | 20 | 25 | 30 | 35 | 40 | 45 | 50 | 55 | 60 |
| 10 | 1/16 | 1/14 | 1/12 | 1/11 | 1/10 | 1/9 | 1/8 | ||||
| 15 | 1/16 | 1/12 | 1/11 | 1/9 | 1/8 | 1/7 | 1/6 | 1/6 | 1/5 | ||
| 20 | 1/16 | 1/12 | 1/10 | 1/8 | 1/7 | 1/6 | 1/5 | 1/5 | 1/4 | 1/4 | |
| 25 | 1/12 | 1/10 | 1/8 | 1/6 | 1/6 | 1/5 | 1/5 | 1/4 | |||
| 30 | 1/16 | 1/11 | 1/8 | 1/6 | 1/5 | 1/5 | 1/4 | 1/4 | |||
| 35 | 1/14 | 1/9 | 1/7 | 1/6 | 1/5 | 1/4 | |||||
| 40 | 1/12 | 1/8 | 1/6 | 1/5 | 1/4 | ||||||
| 45 | 1/11 | 1/7 | 1/5 | 1/5 | 1/4 | ||||||
| 50 | 1/10 | 1/6 | 1/5 | 1/4 | |||||||
| 55 | 1/9 | 1/6 | 1/4 | ||||||||
| 60 | 1/8 | 1/5 | 1/4 |
Capacity of water flow in square furrows
| Grade | Gallons per second | |||
| 1 in 1 000 | 1.8 | 3.9 | 4.6 | 10.9 |
| 1 in 500 | 2.6 | 5.5 | 6.5 | 15.5 |
| 1 in 300 | 3.3 | 6.7 | 8.0 | 19.0 |
| Size of furrow | ||||
| Inches | ||||
| Width | 9 | 9 | 18 | 18 |
| Depth | 6 | 10 | 6 | 10 |
Capacity of water flow in v-shaped furrows1
| Grade | Gallons per second | |||
| 1 in 1 000 | 0.9 | 2.2 | 3.6 | 5.4 |
| 1 in 500 | 1.3 | 3.2 | 4.3 | 7.8 |
| 1 in 300 | 1.6 | 3.9 | 6.5 | 9.6 |
| Size of furrow 1 | ||||
| Inches | ||||
| Width of bottom | 0.0 | 4½ | 9 | 14 |
1 All furrows with 9-in. sides and a slope of 135°.
| A. | 1/10th-acre pond; 3 ft to 1 ft in depth; made in firm soil. | ||
| Hand labor: | 50–80 man days | ||
| Tractor and dam scoop: | 15–20 hours | ||
| Minimum task for digging: | 1 × 3 × 18 ft | 54 cu/ft | |
| or | ½ × 3 × 36 ft | ||
| Minimum task for carrying: as much as one man digs. | |||
| B. | 1/10th-acre pond; 3 ft depth all over; made in wet dambo soil. | ||
| Hand labor: | 60–100 days | ||
| Tractor and dam scoop usually not possible | |||
| Minimum task for digging: | 1 × 3 × 18 ft | 54 cu/ft | |
| or | ½ × 3 × 36 ft | ||
| Minimum task for carrying: as much as one man digs. | |||
Mixed irrigation unit
(a) The unit comprises:
6 fish ponds (approx. 0.5 acre total area)
Vegetable garden (approx. 0.4 acre)
Fruit trees, bananas and sugarcane
Dwelling house
20–30 breeding ducks
Duck houses
Total area of plot 2 acres
(b) Capital cost
Fencing £70
Buildings £80
Ponds £60
Furrows, piping £30
Tools and equipment £15
Breeding ducks £15
Total £270
(c) Balance sheet:
| Debit | Credit | ||
| First year: | £s.d | £s.d | |
| Seeds, etc. | 8.18. 0 | Fish sales | 29.15. 4 |
| Duck food - young stock | 25.15. 4 | Vegetable sales | 54. 1.10 |
| Duck food - breeding stock | 32.10.0 | Duck sales | 55.10. 8 |
| Capital repayment | 15. 0. 0 | ||
Totals | 82. 3. 4 | 139. 7.10 | |
| Net profit to owner | 57. 4. 6 | ||
| Second year: | £s.d | £s.d | |
| Seeds | 4.5.11 | Fish sales | 18. 2. 6 |
| Duck food - young stock | 26. 7. 6 | Vegetable sales | 63. 1. 6 |
| Duck food - breeding stock | 30. 9. 0 | Duck sales | 28.10. 0 |
| Capital repayment | 10. 0. 0 | ||
Totals | 71. 2. 5 | 109.14. 0 | |
| Net profit to owner | 38.11. 7 | ||
| Third year: | £s.d | £s.d | |
| Seeds | 6. 3. 1 | Fish sales | 19. 5. 8 |
| Duck food - young stock | 52.18. 6 | Vegetable sales | 84.17. 0 |
| Duck food - breeding stock | 26.14. 1 | Duck sales | 124.19. 0 |
| Capital repayment | 10. 0. 0 | ||
Totals | 95.15. 8 | 229. 1. 8 | |
| Net profit to owner | 133. 6. 0 | ||
(d) Notes:
All ducks sold at 10/- each if over 4lb liveweight and 7/6 d each if under 4 lb liveweight.
Average price obtained for vegetables approx. 4d per lb.
Fish sold at 6d to 8d per lb.
| Inches | Centimeters | Feet | Meters | Acres | Hectares | Gallons | Liters | Pounds | Kilograms | |||||
| 0.39 | 1 | 2.54 | 3.28 | 1 | 0.30 | 2.47 | 1 | 0.40 | 0.21 | 1 | 4.55 | 2.20 | 1 | 0.45 |
| 0.79 | 2 | 5.08 | 6.56 | 2 | 0.61 | 4.94 | 2 | 0.81 | 0.44 | 2 | 9.09 | 4.41 | 2 | 0.91 |
| 1.18 | 3 | 7.62 | 8.84 | 3 | 0.91 | 7.41 | 3 | 1.21 | 0.65 | 3 | 13.64 | 6.61 | 3 | 1.36 |
| 1.57 | 4 | 10.16 | 13.12 | 4 | 1.22 | 9.98 | 4 | 1.62 | 0.88 | 4 | 17.18 | 8.82 | 4 | 1.81 |
| 1.97 | 5 | 12.70 | 16.40 | 5 | 1.52 | 12.46 | 5 | 2.02 | 1.10 | 5 | 22.72 | 11.02 | 5 | 2.26 |
| 2.36 | 6 | 15.24 | 17.68 | 6 | 1.83 | 14.83 | 6 | 2.41 | 1.30 | 6 | 27.28 | 13.23 | 6 | 2.72 |
| 2.76 | 7 | 17.78 | 20.96 | 7 | 2.13 | 17.30 | 7 | 2.82 | 1.52 | 7 | 31.82 | 15.43 | 7 | 3.17 |
| 3.15 | 8 | 20.32 | 26.24 | 8 | 2.44 | 19.97 | 8 | 3.24 | 1.76 | 8 | 36.37 | 17.63 | 8 | 3.62 |
| 3.54 | 9 | 22.86 | 29.52 | 9 | 2.74 | 22.44 | 9 | 3.64 | 1.95 | 9 | 40.91 | 19.84 | 9 | 4.08 |
| 3.94 | 10 | 25.40 | 32.80 | 10 | 3.05 | 24.71 | 10 | 4.04 | 2.20 | 10 | 45.46 | 22.05 | 10 | 4.53 |
| 4.33 | 11 | 27.94 | ||||||||||||
| 4.72 | 12 | 30.48 | ||||||||||||
Conversion of Centigrade to Fahrenheit 1
| °C | °F |
| 0 | 32.0 |
| 1 | 33.8 |
| 2 | 35.6 |
| 3 | 37.4 |
| 4 | 39.2 |
| 5 | 41.0 |
| 6 | 42.8 |
| 7 | 44.6 |
| 8 | 46.4 |
| 9 | 48.2 |
| 10 | 50.0 |
| 20 | 68.0 |
| 30 | 86.0 |
| 40 | 104.0 |
1 For converting Fahrenheit to Centigrade, use the following formula:
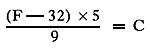
or the reverse, Centigrade to Fahrenheit:
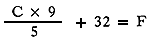
| Anus, or vent | The posterior opening of the alimentary canal through which the excreta are ejected |
| Brackish water | Mainly marine water with reduced salinity caused by the mingling of salty water with fresh water |
| Browsers | Fishes living on leaves and shoots |
| Calcium | A chemical element (Ca) Calcium carbonate (CaCo3) is known as lime |
| Concave | A hollow shape |
| Conservation dam | see Dam |
| Contour | A line joining up points of equal height |
| Convex | A curved or rounded shape |
| Crustacea | Animals including shrimps, crabs, water fleas, etc., mostly aquatic |
| Dam | Artificially built storage for water |
| Dambo | see Vlei |
| Detritus | Organic debris from decomposing plants and animals |
| Diet, balanced | The way of feeding that includes all the essential foodstuff |
| Encysted | Surrounded by a cyst or shell |
| Endemic | Fishes occurring in a specified region |
| Environment | Conditions or influences under which any organism lives |
| Erosion | Wearing away of land by rain, floods, etc. |
| Exotic fish | Fish species introduced from other areas and not indigenous to a given region |
| Factor | A circumstance, fact, or influence which produces a result, e.g., physical, chemical, and biological factors |
| Flood plain | Plain regularly overflooded by flood water |
| Food chain | A chain of organisms where one of the organisms feeds on another from the chain below, and is eaten in turn by an organism from the chain above A short food chain is present when, e.g., a plant is eaten by a fish. If the same fish is eaten in turn by a carnivorous fish, a longer food chain will be present |
| Herbivorous fish | Fish feeding on plants |
| Hullings, maize | Maize waste |
| Hybrid fish | The offspring of fishes of two different species |
| Hypolimnion | That part of the water mass in a lake which is deep and removed from surface influences and generally very low in oxygen |
| Indigenous | Native to a region or a country |
| Larvae | The preadult form, which in the case of some animals hatches from the egg; e.g., tadpole of frog, larvae of mosquitoes, larvae of flies, etc. |
| Laterite | Red porous rock containing iron |
| Lymphatic system | A system for the secretion and conveyance of lymph |
| Mercury | A chemical element commonly known as quicksilver (Hg), used in thermometers and for other purposes |
| Nematode | A group of small, unsegmented roundworms (threadworms) |
| Nitrogen | A chemical element (N), contained in all nitrogenous fertilizers |
| Oligochaete | A class of Annelida comprising the earthworms and several families of small mud-living or aquatic species |
| Ovaries | Organs which produce eggs |
| Oxygen | A chemical element (O) essential for the respiration of most forms of life |
| Parasite | An organism living in or on another organism and drawing its food directly from it |
| Photosynthesis | The conversion by a plant of the carbon dioxide and water of the air into carbohydrates brought about by exposure to sunlight |
| Plankton feeder | A fish that feeds on plankton |
| Progeny | A stock or line descended from a common ancestor |
| Prolific breeding | A method of breeding that produces the maximum amount of offspring |
| Proteins | A complex organic compound composed of numerous amino acids |
| Proteins, animal | Proteins of animal origin |
| Reservoir | see Dam |
| Reservoir culture | Artificial breeding of fish in reservoirs |
| Schist | A crystalline rock, the component minerals of which are arranged in a more or less parallel manner |
| Silt, siltation | Sediment deposited by water |
| Spawning | Breeding, the release of eggs and sperm resulting in fertilized eggs |
| Swamp | A piece of wet, spongy ground which never dries up |
| Vector | An animal which transmits parasites |
| Vitamins | Occur in minute quantities in many foodstuffs and are regarded as essential to normal growth and development |
| Vlei | A piece of low-lying, wet ground sometimes covered with water during the rainy season |
Acacia 106
Algae 128
Alkaline reserve 129
Alkalinity 148
Animals, predatory 41
Aquatic plants 99, 128
Aquiculture 4
Artificial food 130
Barbus 90, 102
Basic slag 57
Baskets 90
Bass 5, 88
Bauhinia 106
Benthos 128, 129
Bilharzia 59, 83, 99, 143
Biological production 123 et seq.
Biological productivity of waters 129
Bluegill 5
Breeding habits, 138
Haplochromis 77
T. andersonii 75
T. macrochir 73
T. melanopleura 70
T. mossambica 72
T. sparrmanii 71
Serranochromis 77
Calcium 124
Carp 5, 7, 15, 61
Catfish 123, 133, 135
Chara 128
Checks daily, 45
weekly 45, 46
monthly 46
Cichlid (family) 5, 17, 24, 62, 63, 83, 89, 134
Clarias 83, 123
Core (or key) trench 34
Cropping See under ponds and dams
Dambo 11, 12, 16, 21, 33
Dams (conservation) 79
clearing 81
cropping 89, 104
stocking 83, 88
stumping 81, 83, 84–85
Demonstration 12
Diseases 105, 106
Draining (ponds) 45
Ducks 59
Eichhornia crassipes (water hyacinth) 99
Epilimnion 125
Equipment for pond construction 42
Eutrophic 129
Extension 12
Fence 41, 43
Fertilizers organic, 48
inorganic 48
Filling (of ponds) 44
Fish, anatomy of 131 et seq.
biology of 131 et seq.
carnivorous 137
culture 3, 7–8, 11, 43, 108
drying of 114–118
farming 58
grass-eating 128
growth of 71–76, 138
herbivorous 137
identification of 62–63, 134
loss of 98
mortalities 106–107
omnivorous 137
predatory 58, 105, 137
production 3, 97, 98
size of 98
spawning 138, 145
spoilage and preservation 142, 112–114
storage of 118
Fish, structure of 131 et seq.
Fish culture economics of, 108
Furrow 19
Gill netting 90, 93, 94
Gnathonemus macrolepidotus 90
Grain foods 50
Grass planting 39, 41
Haplochromis 17, 61, 62, 63, 69, 77–78, 83, 88, 103, 144
Hypolimnion 125
Individual pond record 140
Insect larvae 128
Instruction 12
Key trench See core trench
Labeo spp. 90
Liming 47, 57
Lines 90
Littoral zone 126
Liver fluke 83, 99
Lungfish 123
Malaria 99, 143, 144
Manure (duck, pig) 59–60
Mesotrophic 129
Metalimnion 125
Micropterus salmoides 58
Mixed method culture 57
Monosex culture 58
Mormyrus spp. 133
Mosquitoes 83, 103, 144
Nematodes 128
Nitella 128
Nitrogen 123
Nutrient salts 124, 129
Offal 118–119
Oligochaets 128
Oligotrophic 129
Operculum(a) 134
Otters 22, 41, 105
Oxygen 123, 129, 135, 137, 145–147
Ponds 79
barrage 14, 15, 18, 27, 34
construction 11 et seq.
construction - labor costs 152
construction - walls 22–23, 44
Ponds contour 13, 15, 18, 33
paddy 14–15, 16, 27, 34
sites for 17, 21, 22
stocking of 49, 57, 60
water level in 44
Paddy rice 60
Periphyton 128
pH 129, 147
Pharyngeal teeth 134
Phosphorus 124
Phytoplankton 127, 129
Pisciculture 4
Plankton 127, 129, 137, 148
Plants as food 50
Potash 124
Potomogeton 128
Production (of ponds) 56
Profundal zone 126
Protein for consumption 3, 11, 143
Quantities of food 50
Schilbe mystus 90
Salvinia auriculata (Kariba weed) 99
Seine netting 89, 91, 92
Separate age method culture 57–58
Serranochromis 58, 61–64, 67, 68–69, 83
Snails 59, 103, 128, 144
Strigeidae 106
Superphosphate 48, 57
Temperature 124, 125, 137, 145
Tench 5
Tilapia 7, 17, 58, 61, 62, 63, 83, 88, 103, 106
Tilapia andersonii 60, 63, 74–75, 76
Tilapia macrochir 60, 63, 73, 76, 88
Tilapia melanopleura 62, 63, 70, 71, 76, 83, 99, 134
Tilapia mossambica 60, 63, 72, 88, 138
Tilapia sparrmanii 63, 70, 71
Tilapia zillii 63
Transport 109, 112
Traps 90
Trout 5, 61, 62, 88
Water level 44
lilies 128
quality of 19, 98, 124–126
sources of 18, 19
supply of 17–19
Weed control 46, 47
Weirs 79
Zooplankton 127, 129
FAO SALES AGENTS AND BOOKSELLERS
| Algeria | Société nationale d'édition et de diffusion (SNED), Algiers. |
| Argentina | Librería de las Naciones, Cooperativa Ltda., Alsina 500, Buenos Aires. |
| Australia | Hunter Publications, 58A Gipps Street, Collingwood, Vic. 3066; The Assistant Director, Sales and Distribution, Australian Government Publishing Service, P.O. Box 84, Canberra, A.C.T. 2600, and Australian Government Publications and Inquiry Centres in Canberra City, Melbourne, Sydney, Perth, Adelaide and Hobart. |
| Austria | Wilhelm Frick Buchhandlung, Graben 27, Vienna 1. |
| Bangladesh | Shilpa Niketan, 29 D.I.T. Super Market, Mymensingh Road, Dacca-2. |
| Belgium | Agence et Messaggeries de la Presse, 1 rue de la Petite-Ile, Brussels 7. |
| Bolivia | Librería y Editorial “Juventud,” Plaza Murillo 519, La Paz; Librería Alfonso Tejerina, Comercio 1073, La Paz. |
| Brazil | Livraria Mestre Jou, Rua Guaipá 518, São Paulo 10; Rua Senador Dantas 19-S205/206, Rio de Janeiro. |
| Bulgaria | Hèmus, 11 place Slaveïkov, Sofia. |
| Canada | Information Canada, Ottawa. |
| Chile | Biblioteca, FAO Oficina Regional para América Latina, Av. Providencia 871, Casilla 10095, Santiago; Editorial y Distribuidora Orbe Ltda., Galería Imperio 256, Santiago; Cámara Latinoamericana del Libro, Casilla Postal 14502, Correo 21, Santiago. |
| China | China National Publications Import Corporation, P.O. Box 88, Peking. |
| Costa Rica | Imprenta y Librería Trejos S.A., Apartado 1313, San José. |
| Cuba | Instituto del Libro, Calle 19 y 10 No 1002, Vedado. |
| Cyprus | MAM, P.O. Box 1722, Nicosia. |
| Denmark | Ejnar Munksgaard, Norregade 6, Copenhagen S. |
| Dominican Republic | Librería Dominicana, Calle Mercedes 45-47-49, Santo Domingo. |
| Ecuador | Librería Universitaria, García Moreno 739, Quito; Su Librería, Plaza de Independencia, Quito. |
| Egypt | Al Ahram, El Galaa St., Cairo. |
| El Salvador | Librería Cultural Salvadoreña S.A., 6a Calle Oriente 118, Edificio San Martin, San Salvador. |
| Finland | Akateeminen Kirjakauppa, 2 Keskuskatu, Helsinki. |
| France | Editions A. Pedone, 13 rue Soufflot, 75005 Paris. |
| Germany, F.R. | Alexander Horn Internationale Buchhandlung, Spiegelgasse 9, Postfach 2163, Wiesbaden. |
| Ghana | Ghana Publishing Corporation, P.O. Box 3632, Accra. |
| Greece | “Eleftheroudakis,” 4 Nikis Street, Athens. |
| Guatemala | Agencia de Representaciones “Orbes,” Apartado Postal 753, Guatemala. |
| Haiti | Max Bouchereau, Librairie “A la Caravelle,” B.P. 111B, Port-au-Prince. |
| Hong Kong | Swindon Book Co., 13–15 Lock Road, Kowloon. |
| Iceland | Snaebjörn Jónsson and Co. h.f., Hafnarstraeti 9, P.O. Box 1131, Reykjavik. |
| India | Oxford Book and Stationery Co., Scindia House, New Delhi; 17 Park Street, Calcutta. |
| Indonesia | P.T. Gunung Agung, 6 Kwitang, Djakarta. |
| Iran | Economist Tehran, 99 Sevom Esfand Avenue, Tehran. |
| Iraq | Mackenzie's Bookshop, Baghdad. |
| Ireland | The Controller, Stationery Office, Dublin. |
| Israel | Emanuel Brown, P.O. Box 4101, 35 Allenby Road, and Nachlat Benyamin Street, Tel Aviv; 9 Sblomzion Hamlka Street, Jerusalem. |
| Italy | Libreria Internazionale Rizzoli, Largo Chigi, 00187 Rome; Libreria Scientifica Dott. L. De Biasio “Aeiou,” Via Meravigli 16, 20123 Milan; Libreria Commissionaria Sansoni, S.p.A., Via Lamarmora 45, 50121 Florence; Libreria Macchiaroli, Via Carducci 55/59, 80121 Naples. |
| Japan | Maruzen Company Ltd., P.O. Box 5050, Tokyo Central 100-31. |
| Kenya | The E.S.A. Bookshop, P.O. Box 30167, Nairobi; University Bookshop, University College, P.O. Box 30197, Nairobi. |
| Korea | The Eul-Yoo Publishing Co. Ltd., 5 2-Ka, Chong-ro, Seoul. |
| Kuwait | All Prints Distributors and Publishers, P.O. Box 1719, Kuwait. |
| Lebanon | Dar Al-Maaref Liban S.A.L., place Riad El-Solh, B.P. 2320, Beirut. |
| Malaysia | Caxton Stationers Ltd., 13–15 Leboh Pasar, Kuala Lumpur; Parrys Book Centre, 25 Lobby Arcade, K.L. Hilton, IIn. Treacher, Kuala Lumpur. |
| Mauritius | Nalanda Company Limited, 30 Bourbon Street, Port Louis. |
| Mexico | Sucesión de Manuel Gómez Pezuela y M., Donceles 12, México, D.F.; Editorial Iztaccihuatl, S.A., Miguel Schultz 21, México 4, D.F.; Av. Morelos Ote 437, Monterrey, N.L.; Colón 175, Guadalajara, Jal.; Librería Casarrubias, S.A., Apartado Postal 1–992, Guadalajara, Jal.; Librería Agropecuaria, Av. La Paz 1522, Guadalajara, Jal. |
| Morocco | Librairie “Aux Belles Images,” 281 avenue Mohammed V, Rabat. |
| Netherlands | N.V. Martinus Nijhoff, Lange Voorhout 9, The Hague. |
| New Zealand | Government Printing Office: Government Bookshop at Rutland Street, P.O. Box 5344, Auckland; Mulgrave Street, Private Bag, Wellington; 130 Oxford Terrace, P.O. Box 1721, Christchurch; Princes Street, P.O. Box 1104, Dunedin; Alma Street, P.O. Box 857, Hamilton. |
| Nicaragua | Librería Universal, 15 de Septiembre 301, Managua. |
| Nigeria | University Bookshop Nigeria Ltd., University College, Ibadan. |
| Norway | Johan Grundt Tanum Forlag, Karl Johansgt. 43, Oslo. |
| Pakistan | Mirza Book Agency, 65 The Mall, Lahore 3. |
| Panama | Agencia Internacional de Publicaciones J. Menéndez, Apartado 2052, Panama. |
| Peru | Librería La Universidad, Av. Nicolás de Piérola 639, Lima; Librería Studium, Amargura 939, Lima; Distribuidora Inca, Emilio Althaus 470, Lince, Lima. |
| Philippines | The Modern Book Company, 928 Rizal Avenue, Manila. |
| Poland | Ars Polona-Ruch, Krakowskie Przedmiescie 7, Warsaw. |
| Portugal | Livraria Bertrand, S.A.R.L., Apartado 37, Amadora; Galeria Itau, Rua de Entrecampos 66-A, Lisbon 5. |
| Romania | Rompresfilatelia, P.O. Box 2001, Bucharest (periodicals only); Romlibri, Str. Biserica Amzei 3–5, Bucharest (nonperiodical publications). |
| Saudi Arabia | Khazindar Establishment, King Faysal Street, Riyadh. |
| Singapore | Parrys Book Centre, 25 Lobby Arcade, K.L. Hilton, IIn. Treacher, Kuala Lumpur (Malaysia). |
| Somalia | “Samater's,” P.O. Box 936, Mogadiscio. |
| Spain | Librería Mundi-Prensa, Castelló 37, Madrid; Librería Agrícola, Fernando VI 2, Madrid 4; José Bosch, Librero, Ronda Universidad 11, Barcelona; “Adlha,” Av. General Mitre 100, Barcelona. |
| Sri Lanka | M.D. Gunasena and Co. Ltd., 217 Norris Road, Colombo 11. |
| Sweden | C.E. Fritze, Fredsgatan 2, 103 27 Stockholm 16; Universitetsbokhandel, Sveavägen 166, Stockholm Va.; Gumperts A.B., Göteborg. |
| Switzerland | Librairie Payot S.A., Lausanne and Geneva; Hans Raunhardt, Kirchgasse 17, Zurich 1. |
| Syrian Arab Rep. | Librairie Internationale, B.P. 2456, Damascus. |
| Tanzania | Dar es Salaam Bookshop, P.O. Box 9030, Dar es Salaam. |
| Thailand | FAO Regional Office for Asia and the Far East, Maliwan Mansion, Bangkok; Suksapan Panit, Mansion 9, Rajadamnern Avenue, Bangkok. |
| Togo | Librairie du Bon Pasteur, B.P. 1164, Lomé. |
| Turkey | Librairie Hachette, 469 Istiklal Caddesi, Beyoglu, Istanbul. |
| Uganda | The E.S.A. Bookshop, P.O. Box 2615, Kampala. |
| United Kingdom | Her Majesty's Stationery Office, 49 High Holborn, London, W.C.1; P.O. Box 569, London, S.E. 1 (trade and London area mail orders); 13a Castle Street, Edinburgh EH2 3AR; 109 St. Mary Street, Cardiff CF1 1JW; 7 Linenhall Street, Belfast BT2 8AY; Brazennose Street, Manchester M60 8AS; 258 Broad Street, Birmingham 1; 50 Fairfax Street, Bristol BS1 3DE. |
| United States of America | UNIPUB, Inc., 650 First Avenue, P.O. Box 433, Murray Hill Station, New York, N.Y. 10016. |
| Uruguay | Barreiro y Ramos, 25 de Mayo esq. J.C. Gómez, Montevideo; Librería Albe, Soc. Com., Cerrito 566, Montevideo. |
| Venezuela | Librería Politécnica, Apartado 50738, Sabana Grande, Caracas; Librería del Este, Pericás S.A., Av. Fco. de Miranda 52, Edificio Galipán, Caracas. |
| Yugoslavia | Jugoslovenska Knjiga, Terazije 27/11, Belgrade; Prosveta Export-Import Agency, Terazije 16, Belgrade; Cankarjeva Zalozba, P.O. Box 201 - IV, Ljubljana. |
| Other countries | Requests from countries where sales agents have not yet been appointed may be sent to: Distribution and Sales Section, Food and Agriculture Organization of the United Nations, Via delle Terme di Caracalla, 00100 Rome, Italy. FAO publications are priced in U.S. dollars and pounds sterling. Payment to FAO sales agents may be made in local currencies. |