First draft prepared by Dr. D. Arnold
Federal Institute for Health Protection of Consumers and Veterinary Medicine
Berlin, Germany
IDENTITY
Chemical names:
International non-proprietary name (INN): CYFLUTHRINInternational Union of Pure and Applied Chemistry (IUPAC) name: (RS;SR)-alpha-Cyano-4-fluoro-3-phenoxybenzyl(1RS,3RS;1RS,3SR)-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylate
Chemical Abstract Service (CAS) name: Cyano(4-fluoro-3-phenoxyphenyl)methyl-3-(2,2-dichloroethenyl)-2,2-dimethylcyclopropanecarboxylate
CAS number: 68359-37-5
Synonyms: Baythroid, Cyfoxylate, Bay FCR 1272, FCR-1272, BAY VL 1704
Structural formula:
Cyfluthrin (* Chiral centres)
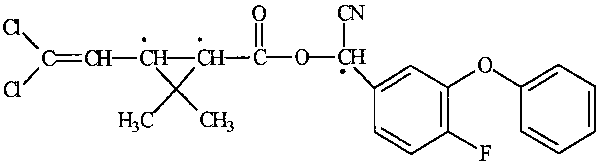
The technical active ingredient consists of 4 diastereomeric enantiomer pairs
|
Enantiomer pair no |
Stereochemical description |
Specified content |
|
I |
1RS, 3RS, a RS (1,3-cis) |
23-27% |
|
II |
1RS, 3RS, a SR(1,3-cis) |
17-21% |
|
III |
1RS, 3SR, a RS (1,3-trans) |
32-36% |
|
IV |
1RS, 3SR, a SR (1,3-trans) |
21-25% |
Molecular formula: C22H18Cl2FNO3
Molecular mass: 434.4 g/mole
OTHER INFORMATION ON IDENTITY AND PROPERTIES
Pure active ingredient:
Specification of purity ³ 92% (sum of isomers). Enantiomer pairs II and IV are regarded as the active components. The inactive enantiomer pairs I and III are not regarded as impurities. The sum of the maximal contents of all unknown by-products is 1%. The following impurities have been identified:
Qualitative and Quantitative Composition of Identified Impurities
|
Chemical name (CA) |
Molecular formula |
Molecular mass |
Max % |
|
4-Fluoro-3-phenoxybenzaldehyde |
C13H9FO2 |
216.2 |
0.5 |
|
(cis+trans)-3-(2,2-Dichloroethenyl)-2,2-dimethylcyclopropane carboxylic acid |
C8H10Cl2O2 |
209.1 |
0.5 |
|
Methyl (cis+trans)-3-(2,2-dichloro-ethenyl)-2,2-dimethylcyclopropane carboxylate |
C9H12Cl2O2 |
223.1 |
1.0 |
|
Tetramethylbutanedinitrile |
C8H12N2 |
136.2 |
1.0 |
|
Cyano(4-fluoro-3-phenoxyphenyl)methyl-3-(2-chloroethinyl)-2,2-dimethylcyclopropane carboxylate |
C22H17ClFNO3 |
397.8 |
2.0 |
|
Cyano(3-phenoxyphenyl)methyl-3-(2,2-chloroethenyl)-2,2-dimethylcyclopropane carboxylate (Cypermethrin) |
C22H19Cl2NO3 |
416.3 |
0.5 |
|
Methylbenzene (solvent) |
C7H8 |
92.1 |
2.0 |
Appearance:
Yellow-brown mass of oily to paste-like consistency, above 60 °C a clear yellow-brown oil.
Melting point:
|
Enantiomer pair |
I |
II |
III |
IV |
|
Melting point (°C) |
64 |
81 |
65 |
106 |
Solubility (g/1000 mL) at 20°C:
|
Solvent/Enantiomer pair |
I |
II |
III |
IV |
|
Water |
2 x 10-6 |
2 x 10-6 |
2 x 10-6 |
2 x 10-6 |
|
Dichloromethane |
>200 |
>200 |
>200 |
>200 |
|
n-Hexane |
10-20 |
10-20 |
10-20 |
1-2 |
|
2-Propanol |
20-50 |
5-10 |
10-20 |
2-5 |
|
Toluene |
>200 |
>200 |
>200 |
100-200 |
Partition coefficient (n-octanol/water):
|
Enantiomer pair |
I |
II |
III |
IV |
|
logPow |
6.00 |
5.94 |
6.04 |
5.91 |
Stability: The shelf-life of the therapeutically used mineral oil formulation is 3.5 years.
RESIDUES IN FOOD AND THEIR EVALUATION
Conditions of use
General
Cyfluthrin is a broad spectrum synthetic type II pyrethroid insecticide and acaricide. It is present on the market in a variety of formulations under the trade name Baythroid as wettable powder, emulsifiable concentrates, oil-in-water emulsions, concentrates and dusts. Major world-wide uses are for pest control, industrial hygiene and vector control. The veterinary medicinal product consists of a 1% ready-to-use mineral oil formulation with the trade name BAYOFLY Pour-on. Indications for its use include infestations by flies and tabanids of cattle of all ages on pasture including lactating dairy cows. Bayofly should not be used in calves. Withdrawal periods are 0 days for edible tissues and milk of cattle.
Dosages
The standard dose volume (10 mL per animal, corresponding to 100 mg of cyfluthrin) is topically applied to the midline of the back of the animal. The resulting calculated dose ranges between 0.2 and 0.5 mg/kg of bw, depending on the body weight of the treated animals. The application may be repeated after 4-6 weeks depending on level of re-infestation.
PHARMACOKINETICS AND METABOLISM
Pharmacokinetics
The pharmacokinetic behaviour of cyfluthrin is largely dependent upon the formulation used. A quantitative comparison of pharmacokinetic parameters is particularly not normally possible, if the information on the vehicle used is incomplete.
Pharmacokinetics in laboratory animals
Summaries of studies in which unlabeled cyfluthrin was orally administered either as a single dose (formulation not given) or over 7 days at different levels in feed to mice and rats were presented. The maximum concentration in sera of rats (average of three animals) of the parent compound was reached 60 minutes after a single dose of 1 mg/kg of bw (Heukamp and Maasfeld 1990). When cyfluthrin was added to the feed of mice (200 and 800 ppm) and rats (150 and 300 ppm), respectively, the concentrations of the parent compound measured in the sera at days 1, 3 and 7 of the feeding experiment were approximately linearly related to the dose in both species. They were highest on day one to three and declined thereafter (Heukamp 1990).
In a large kinetic study [U-14C] fluorobenzene-ring-labeled cyfluthrin (specific activity 26.9 mCi/mmole, radiochemical purity 98%, cis/trans ratio 42/58) was administered to Sprague Dawley rats in a vehicle of 5% CremophorR EL in physiological saline. The time course of the changes of the radioactive counts in body fluids, tissues and excreta as a function of sex, dose, route of administration, and pre-treatment was investigated. Typically 5 animals of each sex were included in each treatment group. Treatment groups alternatively received as single doses either a "low dose" of 0.5 mg/kg of bw (intravenous and oral route), or a "high dose" of 10 mg/kg of bw (oral route), or a "low dose" of 0.5 mg/kg of bw following a 14-day pre-treatment with single daily oral doses of 0.5 mg/kg of bw of the unlabeled active ingredient. The kinetics following a single intraduodenal "low dose" was studied in male rats with bile fistulae. The results of the entire study refer to the sum of the parent compound and its metabolites.
Absorption following oral administration could be described by a lag time of approximately 0.2 hours and half lives of the order of 0.5 hours for both sexes and irrespective of pre-treatment and of the dose applied. The extent of absorption was calculated as about 100% and 90% in males and females, respectively, with the lowest extent of absorption observed in the "high dose" females (86%). The estimate for the male animals was confirmed in the experiment with the animals with bile duct fistulae.
The radioactivity was slowly distributed after application of an intravenous bolus (half life 1.6 hours in males and 2.6 hours in females, respectively).
After oral administration the maximum concentrations in plasma were reached after approximately 2 hours, in most experiments the elimination from plasma could best be described by the sum of two exponential terms with half lives of 2.7 hours for the first term and 12 hours (males) and 9 hours (females) for the second term.
The excretion of 14CO2 was insignificant over a period of 48 hours within which the cumulative excretion via urine and faeces had already reached over 93% and up to 99% of the total recovered radioactivity. The ratio of cumulative urinary/faecal excretion was 2.2 to 3 in male animals and was 1.1 to 1.8 times higher compared to the same ratio in females. It was estimated that approximately one third of the recovered radioactivity was excreted via the bile fluid within the first 48 hours post application of the radioactive dose.
Radioactivity of the tissues, skeletal muscle, liver, kidney and renal fat at the time of sacrifice of 48 hours post administration was highest in renal fat, followed by liver/kidney and skeletal muscle (Klein et al. 1983).
The effects on the absorption of the active ingredient of two vehicles was investigated in a further study with male Wistar rats. The animals received a single oral dose of 10 mg/kg of b.w. either as a 0.1% emulsion in CremophorR EL/distilled water or as a 0.2% solution in polyethylene glycol 400 (PEG 400). The cis/trans ratio of the isomers was 0.84 (45.7% cis-isomers, 54.3% trans-isomers). Two animals of each treatment group were sacrificed 0.5, 1, 2, 4, 6, 16, and 24 hours after dosing. The individual concentrations of all four isomers were separately determined in samples of whole blood and stomach content.
The maxima of the concentrations in blood (sum of all isomers) were reached 1 hour after administration of the CremophorR EL/water emulsion. They were about 4-7 times higher than the maxima obtained with the solution of the active ingredient in PEG 400 which were reached as late as after 6 hours post administration.
In the blood of the CremophorR EL group the cis/trans ratio of the isomers increased to about 2.3 over the first four hours and subsequently dropped down to at or below 1. This ratio, however, never exceeded a value of about 1.2 in the PEG 400 group. In the gastric contents of both groups the isomeric ratio remained slightly above 1 over the whole time where measurable concentrations of the isomers were present. The active ingredient initially disappeared much faster from the stomachs of the CremophorR EL group than from those of the PEG 400 group. However, the concentrations remaining in the stomachs of the CremophorR EL group were still relatively high between 1 and 6 hours post treatment, the period within which the concentrations of the active ingredient dropped down to about 1/10 of the peak concentration (Eben et al. 1982)
Pharmacokinetics in the target animal species
No data were made available for this review.
Metabolism
Metabolism of the permethrinic acid part of the molecule
Studies on the metabolism of cyfluthrin in the rat and the cow were confined to the 3-phenoxy-4-fluoro-benzyl alcohol moiety of the molecule. The metabolism of the remaining permethrinic acid fraction of the molecule is already known from studies with chemically related pyrethroids. One common step of the metabolism of all of these substances is the cleavage of the parent compound at the ester bond yielding permethrinic acid and an individual compound, depending on the structure of the pyrethroid. Ester cleavage of the cyano-pyrethroids is likely to be hydrolytic for the trans-isomers but it may be partly oxidative for the cis-isomers. Both, oxidases and esterases are important in the detoxification of these substances. Hydroxylation of the trans methyl group may take place before ester cleavage. The CN-group of the alpha-cyano pyrethroids is partly excreted as thiocyanate in the urine. Postulated intermediates are the unstable cyanohydrins which are rapidly oxidised to form an aldehyde and to release HCN (Eben and Thyssen 1981).
In male Swiss-Webster mice which were treated with each of cis- and trans-([14C]-cyclopropyl)cypermethrin, metabolism occurred mainly by ester cleavage and elimination of the cis- and trans-3-(2,2-dichlorovinyl)-2,2-dimethylcyclopropanecarboxylic acid moieties (Cl2CA) as glucuronide conjugates. The free acid as well as conjugates with glycine and taurine were also found in the urine of the treated animals. In urine of animals treated with the trans-isomer 3-(2,2-dichlorovinyl)-trans-2-hydroxymethyl-2-methylcyclopropanecarboxylic acid (t-HO-Cl2CA) was also identified. The lactone of the cis-isomer of this compound (c-HO-Cl2CA-lactone) and its glucuronide were tentatively identified (Hutson et al. 1981). The metabolic fate of cypermethrin in young Wistar rats of both sexes was similar (Crawford et al. 1981). The proportions of the major urinary metabolites of cypermethrin were independent on sex or dose in Wistar-derived rats (Rhodes et al. 1984).
Decamethrin labelled in the dibromovinyl carbon atoms and orally administered to male albino mice yielded metabolites which were mostly Br2CA, t-HO-Br2CA, and their glucuronide conjugates. Smaller amounts of Br2CA-glycine and t-HO-Br2CA-sulfate were also detected (Ruzo et al. 1979). Similar results were obtained with rats (Ruzo et al. 1978).
After oral administration of permethrin labelled with 14C in the acid moiety of the molecule the acids (cis- and trans-Cl2CA) and some hydroxylated derivatives of these acids, were in part excreted without conjugation. Hydroxylated metabolites were more extensively formed with cis-permethrin than with trans-permethrin (Gaughan et al. 1977).
Jersey cows were treated with either trans- or cis-permethrin labelled with 14C at the carboxyl group of the acid moiety, c-HO-Cl2CA-lactones appeared in both faeces and urine. The remaining metabolites Cl2CA, c- and t-HO-Cl2CA and various conjugates (including glutamic acid conjugates) appeared only in the urine (Gaughan et al. 1978).
Metabolism of the 3-phenoxy-4-fluorobenzyl alcohol moiety of the molecule in the rat
In a preliminary study a single oral dose of 10 mg/kg of bw of [U-14C]-fluorobenzene labelled cyfluthrin was given to male Sprague Dawley rats ("as apparent solution in physiological saline containing detergent"). More than 65% of the radioactivity were excreted in urine within the first 24 hours post treatment. The metabolites found in the urine collected during the first eight hours were tentatively identified as conjugates of 4'-hydroxy-3-phenoxy-4-fluorobenzoic acid (33% of administered radioactivity) and 3-phenoxy-4-fluorobenzoic acid (27% of administered radioactivity), respectively (Ecker 1982).
In a further study with Sprague Dawley rats of both sexes the treatment scheme of the above mentioned (Klein et al. 1983) kinetic study was followed except that the intraduodenal treatment was omitted. By separating and quantitatively determining the radioactivity associated with the parent drug, free and conjugated 4'-hydroxy-3-phenoxy-4-fluorobenzoic acid and free and conjugated 3-phenoxy-4-fluorobenzoic acid, 65 to 82% of the recovered radioactivity could be assigned to identified compounds isolated from urine and/or faeces. Metabolism was slightly dependent on the dose, in so far as higher concentrations of the parent drug were found in faeces of animals given a single "high dose" (10 mg/kg of bw) or which were pre-treated with 14 daily oral doses of 0.5 mg/kg bw of unlabeled drug.
After intraperitoneal administration of 1, 5, 10 and 15 mg cyfluthrin/kg of bw in 0.02-0.04% polyethylene glycol 400 to Wistar rats, 24 to 42% of the alpha-cyano group of the active ingredient was excreted through the kidneys as thiocyanate (Eben and Thyssen 1981).
Metabolism of the 3-phenoxy-4-fluorobenzyl alcohol moiety of the molecule in the cow
[Phenyl-UL-14C]cyfluthrin, 98.5% radiochemically pure, was orally administered to one lactating Holstein dairy cow on five consecutive days at a dose level of 0.5 mg/kg bw. The substance was formulated in a gelatine capsule with 5 g of alpha-lactose. Milk samples were taken each day after the morning and evening milking. The cow was slaughtered on day 6 after the morning milking.
Nearly all of the radioactive residue (fat >93%, muscle >98%, kidney 99%, liver 100%) was extractable with organic solvents. Tissues had radioactive residues of 622 m g/kg in liver, 120 m g/kg in subcutaneous fat to 230 m g/kg in renal and omental fat, 188 m g/kg in kidney, 40 m g/kg in heart and 21 m g/kg in shoulder and 28 m.g/kg in loin muscle. More than 93% of the radioactivity in the tissues was identified. Kidney, heart, liver, fat and muscle contained unmetabolized cyfluthrin in amounts of 56%, 71%, 86%, >93% and 98%, respectively. The only metabolites found were 4-fluoro-3-phenoxybenzaldehyde (14% in liver) and 4-fluoro-3-phenoxybenzyl alcohol (29% in heart and 43% in liver, respectively).
Milk contained a maximum of 79 m g/kg total radioactive residue (24 hours after the 3rd dose), of which 98% was unmetabolized cyfluthrin (Straw II et al. 1983).
TISSUE RESIDUE DEPLETION STUDIES
Residue depletion studies with radiolabelled cyfluthrin were not available for the present review. All studies discussed here were conducted with unlabelled active ingredient using a variety of formulations.
In three residue depletion studies (I. Woodley 1985 (1), 1985 (2), 1986 (1)) cyfluthrin was dermally applied to female Hereford cattle, poured along dorsal midline at dose levels of 400 mg/animal and 800 mg/animal, respectively, in each study. The range of the animal body weights was from 345 to 445 kg in these studies. The formulation applied was prepared from 10% wettable powder which was dispersed in tap water. Groups of three animals per dose level were sacrificed 1, 3, 7 days (I. Woodley 1985 (1)), 10, 14 days (I. Woodley 1985 (2)), and 42 days (I. Woodley 1986 (1)) post dose, respectively. None of the samples of liver, kidney and muscle contained detectable residues of the parent compound. The limit of detection was 10 m g/kg (GLC with electron capture detection). Visceral fat from the bellies was the only tissue with quantifiable residues of the parent compound. Maximum concentrations of approximately 90 m g/kg were found in fat of the high-dose group 14 days after treatment.
In another study (Woodley 1986 (2)), a pour-on formulation (Bay 1704, 1% w/v Pour on 034-V) was used. 200 mg per animal were applied to 21 Hereford cattle (17 females, 4 castrated males) in one spot between the shoulder blades. Three animals were slaughtered at each of seven different time points post dose (1, 3, 7, 10, 14, 21, and 28 days). Again, fat was the only tissue with quantifiable residues. The highest concentrations, in the order of 50 m g/kg, were found on days 7 to 14 after treatment.
Waggoner and Kohlenberg reported two studies where cattle were topically dosed along the backline with cyfluthrin 1% pour-on formulations. Following sacrifice the concentration of parent compound was determined in fat, liver, kidney and muscle. In the first study (Waggoner and Kohlenberg 1990 (1)) two groups of three female Holstein cattle were treated. One group received 0.63 mg/kg of bw on day one and a second dose of 0.9 mg/kg of bw 13 days later. The animals were sacrificed two days after the second treatment. Residues of the parent compound were found in fat (average 90 m g/kg), kidney (average 12 m g/kg) and liver (average 8 m g/kg). No residues were detected in muscle. The other group was treated five times with 0.9 mg/kg of bw (three doses at one-day intervals on the first three days, 2 further doses at intervals of 12 days). The animals were sacrificed two days after the fifth treatment. Again no residue was detected in any sample of muscle meat. Residues in the other tissues, however, were more variable and (on the average) higher than in the first treatment group. The values found (3 animals) were 170, 86, and 240 m g/kg in fat, 17, 12, and 23 m g/kg in kidney, and 22, 9, and 8 m g/kg in liver.
These studies showed that under the described conditions of significant overdosing, residue levels in fat were about 7.5 to 9.5 times higher than in any other of the standard edible tissues.
In the second study (Waggoner and Kohlenberg 1990 (1)) the same authors treated 3 groups each of 3 yearling cattle (Hereford, Angus and mixed breed) with either one, two or three doses of 0.44 mg/kg of bw of Mobay's cyfluthrin 1% pour-on formulation. The dose interval was 21 days. Animals were slaughtered 3 days after the last treatment. Fat was the only tissue were quantifiable residues were found. The average concentrations increased approximately linearly with the number of repeated doses and were 31, 86 and 122 m g/kg after one, two and three doses, respectively.
In a further study with Friesian Lowland cattle Bayofly pour on, 10 ml of the product per 300 kg of animal weight equivalent to about 0.22-0.25 mg/kg of bw, were poured along the animal withers. Two animals were killed on each of days 1, 4, 7, 14, 21 and 28 post application. No detectable residues were found in liver, kidney and muscle. Concentrations of residues in fat increased from day 1 (14 m g/kg) to day 14 (34 m g/kg) and then decreased to reach 16 m g/kg on day 28.
In a study (Shaw II and Ayers 1983) designed to simulate the continuous uptake by dairy cattle of cyfluthrin residues of feed originating from plants treated with plant protection products, the animals received the drug orally once per day over 29 days in a capsule containing 9 g of alpha-lactose. The doses were adjusted to simulate concentrations in feed of 5, 15 and 50 mg/kg, respectively. Residues were monitored in selected milk samples and all three animals of each dose group were slaughtered on day 29, shortly after the administration of the last dose. Fat, liver, kidney and muscle tissues were analysed for the presence of the parent drug. Liver and kidney were also assayed for 'acid'-Baythroid (cyanogroup transformed to carboxyl group) and the sum of 'FPB acid' (4-fluoro-3-phenoxybenzoic acid), 'FPB aid' (4-fluoro-3-phenoxybenzaldehyde) and 'FPB ale' (4-fluoro-3-phenoxybenzyl alcohol). The analyses of liver were repeated two years later (Murphy 1985) when it became evident that the contract laboratory of the first analysis had not complied with the foreseen analytical procedure. Concentrations of the parent compounds in liver and kidney samples were, therefore, initially underestimated. The maximum residue concentration in milk (parent compounds) was reached at day 14 with a range from 160 to 260 m g/kg in the high dose group. The corresponding values were 100 to 170 m g/kg on day 28. The concentrations of the parent drug were lower, in a dose dependent manner, in milk from the low-dose and intermediate-dose animals on day 28.
The concentrations of the parent drug found in fat were the highest of all standard edible tissues. The average values were dose-dependent (250 m g/kg of residue at feed level of 5 mg/kg, 700 m g/kg of residue at feed level of 15 mg/kg, and 2600 m g/kg of residue at feed level of 50 mg/kg). In muscle no parent drug was detectable in the tissues of the animals of the low-dose group and in two of the animals of the intermediate-dose group. The respective concentration in muscle tissues of the high-dose group was 30 m g/kg. The concentrations in liver and kidney of the parent drug were 130 and 170 m g/kg, respectively, in tissues of high dose animals. The sum of all metabolites was <30 m g/kg in liver and kidney, respectively, in samples of the same animals.
P. Hedemann (1985 (1), 1985 (2), 1986) conducted three studies where cyfluthrin was topically applied to either Friesian or Illawara Shorthorn dairy cattle. In the first two studies Bay 1704 1% pour on was applied to the dorsal midline (two groups dosed with 100 and 200 mg/animal, respectively). In the third study a 0.2% formulation was prepared from 10% wettable powder and water and was sprayed on the animals along the backline. In this study 400 and 800 mg/animal, respectively, were applied. None of the milk samples taken up to 56 hours post administration contained detectable quantities of the parent drug (limit of detection 10 m g/kg). However, in a study which was apparently similarly designed 10 m g/kg of parent drug were found in one sample taken 3 days post administration and in three samples taken 5 days post dosing in the group dosed with 800 mg/animal (P. Gyr 1987).
Two groups of three Holstein dairy cattle each were treated with cyfluthrin with a 1% pour on formulation. The animals of the first group received a single dose equivalent to 0.63 mg/kg of bw. In the second group the animals were treated three times at daily intervals with 0.9 mg/kg of bw. The maximum concentrations of the parent drug in whole milk of the animals treated only once, were found 2-4 days post administration. They were in the order of 6-26 m g/kg. In milk samples of the animals treated three times with the higher dose maximum concentrations ranged from 23 to 54 m g/kg and were reached 1-2 days after the last administration (Waggoner and Kohlenberg 1990 (2)).
Bayofly was dermally applied to 6 Friesian dairy cows at doses equivalent to 200 mg/animal. Residues of the parent drug were analysed in milk samples taken 9, 24, and 72 hours post dosing, respectively. The results of this study were expressed on the basis of the fat content of the milk. The highest concentrations were measured in samples taken 1 day after treatment and were ranging from 50 to 210 m g/kg (Alexander and Booth 1989).
METHODS OF ANALYSIS FOR RESIDUES IN TISSUES
The analytical parts of a number of residue studies refer to a method described in Mobay Report 85883 (Straw II et al. 1983 (1)). The report fully describes a gas chromatographic method for measuring the intact residues of BAYTHROID in bovine and poultry tissues, milk and eggs. Baythroid is removed from the sample matrix by solvent extraction followed by partitionning steps and purification on silica gel (column or Florisil Sep-Pak cartridge). The purified extract is then subjected to GLC with electron capture detection without prior derivatization. A standard column and a second confirmatory column is proposed. The isomers are not separated. Several chromatograms and some recovery data are included in the report. The limit of detection is in the order of 10 m g/kg for all tissues. The method was, e.g., used with minor modifications in residue studies reported by Waggoner and Kohlenberg (1990 (1) and (2)). Since in these studies all samples were analysed in duplicate, the % RSD for incurred tissues could be estimated. It was slightly concentration dependent. Most estimates were ranging between 0 and 10% with some outliers up to about 35%.
A routine method for the determination of residues of BAYOFLY in bovine tissues and milk has been developed more recently (Maasfeld 1989). This method is to be considered as the proposed regulatory method. The fully described method uses acetonitril for extraction. Lipids are removed with hexane. The dry residue remaining after solvent evaporation is further cleaned on a silica column or on Sep-Pak-C18 cartridge. Following solvent evaporation of the fraction containing cyfluthrin the dry residue is dissolved in ethyl acetate and subjected to capillary GLC with electron capture detection. The four isomers are sufficiently separated to allow their separate determination.
Recovery experiments were conducted with fortified tissues at two concentrations (0.01 and 0.05 mg/kg for fat, kidney, liver and muscle; 0.005 and 0.05 mg/kg for milk) performing five replicate analyses of each tissue/concentration pair. Depending on the matrix and the analyte concentration the recoveries were between 72 and 89% with relative standard deviations of 4-12%.
Three control samples of each matrix were analyzed in order to estimate the limit of detection (LOD) which was defined as average noise of the baseline of the chromatographic trace plus three times the standard deviation of this figure. The reported LOD's are 0.001 mg/kg for milk, 0.002 mg/kg for muscle and kidney, and 0.003 mg/kg for liver and fat.
For the definition of the limit of quantification (LOQ) the requirements were as follows: recovery >70% and RSD <0.2. With these requirements the LOQ was 0.005 mg/kg for milk and 0.01 mg/kg for liver, kidney, muscle and fat.
The specificity of the method was confirmed with permethrin and cypermethrin.
The method was applied to incurred tissues obtained in a residue depletion study with cattle (Dom and Maasfeld 1989 (1)). In three of 12 fat samples analysed in duplicate the variability of the analytical results was higher than expected, if compared with the results obtained with fortified control samples.
RESIDUES IN EDIBLE ANIMAL TISSUES RESULTING FROM THE USE OF CYFLUTHRIN IN CROP PROTECTION
The sponsor has provided a copy of relevant parts of the dossier submitted to European Community authorities in accordance with the provisions of Directive 91/414/EEC. From the data which cannot be fully discussed in this monograph, it appears, however, that the use of cyfluthrin for spray treatment in agriculture and horticulture does not lead to a quantifiable transfer of residues from animal feed to liver, kidney and muscle tissues. Residues in fatty tissues and milk of animals which are transferred from feed as a result of the use of cyfluthrin in crop protection should be significantly lower than those found after authorized uses of the veterinary product.
APPRAISAL
The Committee reviewed numerous data including both proprietary information and data from the open literature. A comprehensive expert report covering the use of cyfluthrin in veterinary medicine and an additional report dealing with the residues of the compound that are likely to result from non-veterinary uses had also been provided.
Residues in tissues
Residue depletion studies with radiolabelled cyfluthrin were not available for the present review. All studies presented were conducted with unlabelled active ingredient in a variety of formulations.
In three similarly designed residue depletion studies, a formulation of cyfluthrin was topically applied to cattle at dose levels of 400 mg/animal (0.9-1.1 mg/kg of bw) and 800 mg/animal (1.8-2.3 mg/kg of bw), respectively, in each study. The formulation applied was prepared from 10% wettable powder dispersion in water. Groups of three animals per dose level were sacrificed 1, 3 and 7 days or 10 and 14 days or 42 days post-dose, respectively. None of the samples of muscle, liver and kidney contained detectable residues of the parent compound. The limit of detection was 10 m g/kg. Maximum concentrations of 70-100 m g/kg were found in visceral fat of the high-dose group 14 days after treatment.
In four other studies cattle were dosed with cyfluthrin 1% pour-on formulations. In the first of these four studies, 200 mg/animal (0.5-0.93 mg/kg of bw) were applied to 21 cattle. Three animals were slaughtered at each of seven different time-points post-dose (1, 3, 7, 10, 14, 21 and 28 days). Fat was the only tissue with quantifiable residues. The highest concentrations, approximately 50 m g/kg, were found on days 7 to 14 after treatment.
In the second study the cattle were treated with 0.33 mg/kg of bw. Two animals were sacrificed on each of days 1, 4, 7, 14, 21 and 28 post application of the dose. No detectable residues were present in muscle, liver and kidney. Concentrations of residues in fat increased from day 1 (14 and 15 m g/kg) to a maximum of 32 and 36 m g/kg on day 14 and then decreased to reach 14 and 18 m g/kg on day 28 post-dose.
In the third study, two groups of three cattle each were treated. One group received 0.63 mg/kg of bw on day one and a second dose of 0.9 mg/kg of bw 13 days later. The animals were sacrificed two days after the second treatment. No residues were detected in muscle. Residues of the parent compound were found in liver (6-9 m g/kg), kidney (11-13 m g/kg) and fat (72-110 m g/kg). The second group was treated five times with 0.9 mg/kg of bw (one daily dose during the first three days and 2 further doses at 12-day intervals). The animals were sacrificed two days after the fifth treatment. The concentrations of residues were 8-22 m g/kg in liver, 12-23 m g/kg in kidney, and 86-240 m g/kg in fat. This study showed that under the described conditions of significant overdosing, residue levels in fat were about 7.5 times higher than in kidney and 9.5 times higher than in liver.
In the fourth study, three groups each of three yearling cattle were treated with either one, two or three doses of 0.44 mg/kg of bw. The dose interval was 21 days. Animals were slaughtered 3 days after the last treatment. Fat was the only tissue where quantifiable residues were found. The concentrations increased with the number of repeated doses and were 21-42 m g/kg after one, 77-102 m g/kg after two, and 104-151 m g/kg after three doses.
The four studies indicate that residues in fat increase with increasing dosage and only in the highest treatment study (significant overdosing) were residues of liver and kidney quantifiable. No residues were detected in muscle tissue.
Residues in Milk
Two groups of three dairy cattle each were treated with cyfluthrin as a 1% pour-on formulation. The animals of the first group received a single dose equivalent to 0.63 mg/kg of bw. In the second group the animals were treated three times at daily intervals with 0.9 mg/kg of bw. The maximum concentrations of the parent drug in whole milk of the animals treated only once were found 2-4 days after treatment. They were 6-26 m g/kg. At 7 days after treatment the concentrations of residues were 2-5 m g/kg. In milk samples of the animals treated three times with the higher dose, maximum concentrations ranged from 23 to 54 m g/kg and were reached 1-2 days after the last administration.
Method of Analysis
A routine method for the determination of the marker residue of cyfluthrin in bovine tissues and milk was proposed. The residues are extracted with organic solvent and solid phase chromatography and subjected to capillary gas liquid chromatography with electron capture detection. The four diastereoisomers are sufficiently resolved to allow their separate determination.
Recovery experiments were conducted with fortified tissues at two concentrations (10 and 50 m g/kg for muscle, liver, kidney and fat; 5 and 50 m g/kg for milk). Depending on the matrix and the analyte concentration, the recoveries were between 72 and 89% with relative standard deviations of 4-12%.
The reported LOD's were 1 m g/kg for milk, 2 m g/kg for muscle and kidney, and 3 m g/kg for liver and fat. The LOQ was 5 m g/kg for milk and 10 m g/kg for muscle, liver, kidney and fat. The specificity of the method was confirmed in the presence of permethrin and cypermethrin.
Maximum Residue Limits
The Committee confirmed the ADI of 0-20 m g/kg of bw previously allocated by JMPR. This would result in a maximum acceptable daily intake of 1200 m g for a 60-kg person. In recommending MRLs, the Committee took also into account the following facts:
· Fat was the only suitable target tissue.· Residue concentrations in liver and kidney are about 10 times lower than those in fat.
· The parent compound was the only marker residue and the only residue of concern.
· There was no residue study available which had been conducted exactly at the upper limit of the recommended dose range (0.5 mg/kg of bw) and with repeated dosing at 28 day intervals, the shortest recommended time interval for repetition of the treatment. The numerous available data showed that
- estimates of the maximum concentrations resulting from a given single dose could be obtained by linear interpolation of the data obtained with other similar doses,- only studies with identical formulations of cyfluthrin could be quantitatively compared,
- the kinetics of repeated treatment could be described by superposition and addition of the data obtained after a single dose with the same formulation,
- the results obtained after identical treatment could vary highly from animal to animal.
The maximum residue concentration in milk, expressed as the geometric mean of the data, could be interpolated to approximately 13 m g/l. The expected maximum residue level in fat at the dose of 0.5 mg/kg of bw, representing the upper limit of the recommended dose range, could be estimated by interpolation from the results of the available studies to be 40 m g/kg.
The maximum residue concentrations in fat were reached between 10 and 14 days after a single dose. The residue levels decreased to 50% around 28 days post-dose and to 30% of the maximum concentration around 40 days post-dose. The expected numerical value of the maximum after one repetition of the treatment would be approximately 1.3-times higher than the maximum value obtained after a single dose. With more than one repetition at 28 day intervals, the maximum concentration would probably not exceed 1.5 times the maximum obtained after a single dose. It could therefore be estimated that the highest concentration, expressed as the geometric mean of the obtained values, to be observed after repeated treatments at the recommended dose interval and the upper limit of the recommended dose range would be in the order of 1.5 x 40 m g/kg = 60 m g/kg. This value could serve as a basis for establishing an MRL in fat.
It was, however, necessary to also consider the significant biological variability of the residue concentrations in fat of individual animals. It was observed that the geometric mean plus three times its standard deviation would cover this variability.
Taking all these factors into account, an MRL for fat of 200 m g/kg was recommended. This is the only MRL which can be used to monitor residues of cyfluthrin in edible tissues of treated cattle. The recommended MRL for liver and kidney in both tissues was 20 m g/kg. The recommended MRL for muscle was 20 m g/kg on the basis of twice the limit of quantification of the analytical method. This MRL, however, far exceeds the estimated concentrations of the residues in muscle resulting from the use of the drug following the label instructions and can, therefore, not be used to monitor residues in the other tissues. Liver and kidney tissues are not suited as target tissues for the control of residues of cyfluthrin in cattle.
The maximum residue concentration in milk, expressed as the geometric mean of the data could be interpolated to approximately 13 m g/kg. Taking into account the standard deviation of that figure, an MRL for milk was recommended at 40 m g/kg.
The present Committee noted that the MRLs for apple, cotton seed, rape seed, maize, sweet peppers and tomato recommended by the JMPR in 1987 resulted in a TMDI of 4% of the ADI (Middle Eastern diet), 0.7% (Far Eastern diet), 1.2% (African diet), 1.5% (Latin American diet) and 4.6% (European diet), respectively. The MRLs for muscle, liver, kidney and fat recommended by the present Committee would result in an additional TMDI of 79 m g/person or 6.6% of the ADI.
It was also noted that the JMPR had recommended an MRL of 10 m g/kg for milk suggesting that it was covering the veterinary uses of cyfluthrin. The present Committee, however, noted that important studies of the residues in milk had not been available to the JMPR for evaluation. It was, therefore, necessary to propose a higher MRL for milk, in order to cover the authorized uses of cyfluthrin in veterinary medicine.
REFERENCES
Akhtar, M.H., Hamilton, R.M.G., and Trenholm, H.L. (1985), Metabolism, Distribution, and Excretion of Deltamethrin by Leghorn Hens, J. Agoc. Food Chem., 33, 610-617.
Alexander, A.M. and Booth, D.H. (1989), An estimation of residue levels of cyfluthrin in milk fat (dairy cows), Massey University, Palmerston North, New Zealand.
Crawford, M.J., Croucher, A. and Hutson, D.H. (1981), The Metabolism of the Pyrethroid Insecticide Cypermethrin in Rats; Excreted Metabolites, Pestic. Sci., 12, 399-411.
Dom H. and Maasfeld, W. (1989) (1), Residue investigations of tissue from cattle after application of Bayofly pour on, Report No. RA-654/89.
Dom, H. and Maasfeld, W. (1989) (2), Residue investigations of milk from cows after application of Bayofly pour on, Report No. RA-655/89.
Eben, A., Heimann, K.G., and Machemer, L. (1982), Comparative study of rats on absorption of FCR 1272 after single oral administration in polyethylene glycol 400 or Cremophor EL/water as formulation vehicle, Report No. 10715.
Eben, A. and Thyssen, J. (1981), Thiocyanate Excretion in Rats Urine after Intraperitoneal Administration of FCR 1272 and Decamethrin in Comparable Doses and after Exposure to Defined FCR 1272 Concentrations in the Inhalation Air, Bayer Report No. 10130.
Ecker, W. (1982), Biotransformation of [fluorobenzene ring-U-14C]Cyfluthrin; Characterisation and provisional identification of metabolites. Report No. 10575.
Ecker, W. (1983), [Fluorobenzene-UL-14C]FCR 1272; [fluorobenzene-UL-14C]Cyfluthrin: Metabolism part of the general metabolism studies in the rat, Report No. 2059.
Gaughan, L.C., Unai, T., and Casida, J.E. (1977), Permethrin Metabolism in Rats, J. Agric. Food Chem., 25, 9-17.
Gaughan, L.C., Ackerman, M.E., Unai, T., and Casida, J.E. (1978), Distribution and Metabolism of trans- and cis-Permethrin in Lactating Jersey Cows, J. Agric. Food Chem., 26, 13-618.
Gyr, P. (1987), To determine the Bay 1704 residues in the milk of cattle at various times after treatment, Bayer Report No. 87/12856.
Hedemann, P. (1985)(1), To assess the residual concentration of BAY 1704 in the milk of treated cattle, Bayer Report No. 85/11963.
Hedemann, P. (1985)(2), To assess the residual concentration of BAY 1704 in the milk of treated cattle, Bayer Report No. 85/12081.
Hedemann P. (1986), To assess the residual concentration of BAY 1704 in the milk of treated cattle, Bayer Report No. 85/12357.
Heukamp, U. and Maasfeld, W. (1990), Determination of the residue of BayoflyR (FCR 1272) in rat serum following oral administration, Bayer Report No. RA 848-90.
Heukamp, U. (1990), Determination of the residue of BayoflyR (FCR 1272) in mouse and rat serum following oral administration, Bayer Report No. RA 849-90.
Hutson, D.H., Gaughan, L.C. and Casida, J.E. (1981), Metabolism of the cis- and trans-isomers of Cypermethrin in mice, Pestic. Sci., 12, 385-398.
Klein, O., Weber, H., and Suwelack, D. (1983), [U-14C] Cyfluthrin ([U-14C] FCR 1272; Fluorobenzene label): Biokinetic part of the general metabolism studies in the rat, Report No. 11872.
Maasfeld, W. (1989), Method for the gas chromatographic determination of residues of Bayofly in bovine tissues and milk, Report No. RA-653.
Murnhy, J.J. (1985), Baythroid: Identity of major components in cow liver, Addendum to Mobay Report No. 86045, Mobay Report No. 88970.
Rhodes, C., Jones, B.K., Croucher, A., Hutson, D.H., Logan, C.J., Hopkins, R., Hall, B.E., and Vickers, J.A. (1984), Pestic. Sci., 15, 471-480.
Ruzo, L.O., Engel, J.L., and Casida, J.E. (1979), Decamethrin Metabolites from Oxidative, Hydrolytic, and Conjugative Reactions in Mice, J. Agric. Food Chem., 27, 725-731.
Ruzo, L.O., Unai, T., and Casida, J.E. (1978), Decamethrin Metabolism in Rats, J. Agric. Food Chem., 26, 918-925.
Shaw, H.R. and Ayers, J.E. (1983), Baythroid 28 day bovine feeding study, Mobay Report No. 86045.
Shaw II, H.R. et al. (1983) (1), An analytical method for Baythroidä in bovine and poultry tissues, milk and eggs, Mobay Report No. 85883.
Shaw II, H.R. et al. (1983) (2), An analytical method for quantitating Baythroidä metabolite residue in animal tissues, Mobay Report No. 86217.
Shawn, H.R., Ayers, J.E., and McCann, S.A. (1983), Metabolism of Baythroidä in a dairy cow, Report No. 86043.
Stockham, M.A. (1994), Cyfluthrin - Report on residues for MRL application, International Research Consultants, Ely, UK.
Waggoner, T.B. and Kohlenberg, M.L. (1990) (1), Cyfluthrin - Magnitude of the residue; Dermal application to beef cattle, Mobay Report No. 74051.
Waggoner, T.B. and Kohlenberg, M.L. (1990) (2), Cyfluthrin - Magnitude of the residue; Dermal application to lactating dairy cattle, Mobay Report No. 74050.
Weber, H. and Suwelack, D. (1983), Fluorophenyl-UL 14C-Cyfluthrin (FCR 1272); Biokinetic study on rats, Report No. 11575.
Werner, G. (1994), Cyfluthrin - Evaluation of residues resulting from other than veterinary uses and estimation of the overall potential cyfluthrin exposure through diet of plant and animal origin. Addendum to the expert report on the residue file prepared in accordance with Council Regulation (EEC) 2377/90 by M.A. Stockham
Woodley, I. (1985)(1), To measure the residues of BAY 1704 in tissues of cattle at various times after treatment, Report No. 85/11999.
Woodley, I. (1985)(2), To measure the residues of BAY 1704 in tissues of cattle at various times after treatment, Report No. 85/12007.
Woodley, I. (1986) (1), To measure the residues of BAY 1704 in fat samples collected from cattle 42 days after treatment, Report No. 86/12222.
Woodley, I. (1986) (2), To measure the residues of BAY 1704 in tissue collected from cattle at various times after treatment, Report No. 86/12215.