by
Norman Stanley
FMC Corporation, Marine Colloids Division
5 Maple Street, Rockland
Maine 04841, USA
CARRAGEENANS
INTRODUCTION
Carrageenans are commercially important hydrophilic colloids (water-soluble gums) which occur as matrix material in numerous species of red seaweeds (Rhodophyta) wherein they serve a structural function analogous to that of cellulose in land plants. Chemically they are highly sulfated galactans. Due to their half-ester sulfate moieties they are strongly anionic polymers. In this respect they differ from agars and alginates, the other two classes of commercially exploited seaweed hydrocolloids. Agars, though also galactans, have little half-ester sulfate and may be considered to be nonionic for most practical purposes. Alginates, though anionic, are polymers of mannuronic and guluronic acids and as such owe their ionic character to carboxyl rather than sulfate groups. In this respect alginates are more akin to pectins, found in land plants, than to the other seaweed hydrocolloids.
Applications of carrageenans make use of both their hydrophilic and anionic properties, the latter influencing the former.
SOURCES
In the West the algae Chondrus crispus and Gigartina stellata, as the sun-bleached whole plants, have been used for centuries for making jellies and milk puddings (blanc mange). An old recipe for blanc mange (Smith, 1905) is as follows:
"Soak half a cup of dry moss in cold water for five minutes, tie in a cheesecloth bag, place in a double boiler with a quart of milk and cook for half an hour; add half a teaspoonful of salt or less, according to taste, strain, flavor with a teaspoonful of lemon or vanilla extract as desired, and pour into a mold or small cups, which have been wet with cold water; after hardening, eat with sugar and cream."
"Moss" here refers to Irish moss, a common name for Chondrus crispus. Irish moss has also been known as carrageen from the Irish word, carraigeen, meaning "rock moss."
Stanford (1862) coined the name "carrageenin" for the gelatinous material extracted by water from Chondrus crispus. The present spelling, "carrageenan", has become accepted within the past 25 years, this being consonant with the use of the - an suffix for the names of polysaccharides.
In the Far East marine algae of the genus Eucheuma in the Solieriaceae family have a long history of use as articles of food, for their supposed medicinal properties, and in trades such as bookbinding wherein their mucilage is used as an adhesive (Eisses 1953; Zaneveld 1955, 1959). Although the Malayan word, "agar-agar" refers to Eucheuma species it is now known that these yield carrageenans rather than agar-type polysaccharides.
Due to the importance of sea plants in the economy of Indonesia and the general area of the Malay peninsular sporadic attempts were made to set up programs for the comprehensive investigation of the marine algae of the area and the products obtainable from them. Unfortunately, due to the underdeveloped economies and unsettled political conditions in these countries, none of the proposed programs was ever carried out to the extent comtemplated. The principal results of these investigations have been reported by Eisses (1952, 1953) and Zaneveld (1955, 1959). They describe a number of types of Eucheuma which have been harvested in this area, listing them by their botanical and native names. It was not until the Eucheumas were recognized as valuable carrageenophytes by the western carrageenan industry that the large-scale export of these species became established. The introduction of Eucheuma farming in the Philippines in 1971 greatly promoted this industry.
From the original identification of Chrondrus crispus as a carrageenophyte this classification is now known to cover numerous species from seven different families: Solieriaceae, Rhabdoniaceae, Hypneaceae, Phyllophoraceae, Gigartinaceae, Furcellariaceae, and most recently, Rhodophyllidaceae (Deslandes et al., 1985). While not all of these have been, or are perhaps likely to be, exploited commercially, present-day sources of carrageenans go well beyond the original Irish moss. Seaweeds which have been used for carrageenan production include Chondrus crispus, C. ocellatus, Gigartina stellata, G. acicularis, G. pistillata, G. canaliculata, G. chamissoi, G. radula (also identified in the literature as Iridea species), G. skottsbergii, Gymnogongrus furcellatus, Eucheuma cottonii, E. spinosum, E. gelatinae, Furcellaria fastigiata, Hypnea musciformis, and H. spicifera. Utilization of these not only has greatly extended the base, and the geographical area, from which the industry can draw raw materials, but also has extended the range of properties of their extractives, as different species yield carrageenans of differing structure and properties.
Chondrus crispus is largely harvested in the Maritime Provinces of Canada with smaller quantities collected along the coasts of Maine and Massachusetts in the United States. The difference in volume is due not so much to lack of abundance as to differences in the local economies. The carrageenan from C. crispus, which comprises a mixture of kappa- and lambda-carrageenan, is much valued as the preferred type for applications such as chocolate milk stabilization.
It is now known that kappa- and lambda-carrageenan do not occur together in the same plant, but are elaborated at different stages of the reproductive cycle. Kappa-carrageenan occurs in the haploid gametophytic plants and lambda in the diploid tetrasporophytes (Chen et al., 1973). Since these occur and are harvested together a mixture of kappa and lambda is obtained on processing. This is true not only of Chondrus but of other species of Gigartinaceae and Phyllophoraceae as well (McCandless, West and Guiry, 1982, 1983). It has not been found to hold true for carrageenophytes in the Solieriaceae family (Doty and Santos, 1978) or, as far as is known, in those of other families.
Gigartina acicularis and G. pistillata occur and are harvested together along the coasts of southern France, Spain, Portugal, and Morocco. The latter two species are unique in that they yield a nongelling, predominantly lambda or xi type carrageenan.
Gigartina radula is harvested in Chile and comprises a major resource for carrageenan production. The taxonomy of Chilean G. radula is a subject of controversy, with some algologists holding to the earlier classification of this seaweed as one or more Iridea species.
Eucheuma cottonii and E. spinosum are now heavily used by carrageenan producers and are harvested in large quantities in Indonesia and the Philippines. Farming of these Eucheuma species is now practiced on a large scale in the Philippines, and this has done much to increase and stabilize the supply of these important carrageenophytes. E. cottonii and E. spinosum are remarkable in that these two species, once thought to be varieties of a single species, yield quite different types of carrageenans. E. cottonii, which actually may comprise two very similar species, E. cottonii and E. striatum (Doty, 1973), yields nearly ideal kappa-carrageenan, while E. spinosum yields nearly ideal iota-carrageenan.
Gymnogongrus furcellatus, harvested in Peru, has been used as a source of iota-type carrageenan. Supplies of this weed, however, are small.
Furcellaria fastigiata yields furcellaran ("Danish agar"), often treated in the literature as a polysaccharide distinct from carrageenan but now considered, on the basis of chemical evidence (Lawson, et al., 1973), to be a member of the carrageenan family of polysaccharides. Although F. fastigiata is found along many coasts of the North Atlantic and its adjacent seas major quantities are harvested only in Denmark and the Maritime Provinces of Canada. It grows both in an attached and unattached form, the latter reproducing only vegetatively. A large body of unattached Furcellaria in the Kattegat, near the coast of Jutland, was for many years the sole source of raw material for furcellaran production (Lund and Bjerre-Petersen, 1952, 1961). In some Canadian locations Furcellaria grows alone, but most commonly it grows in mixed beds with Chondrus crispus. It is collected principally on the coasts of Prince Edward Island and Nova Scotia after being cast up on the beaches. The unattached form of Furcellaria has not been found in Canada (Bjerre-Petersen, Christensen and Hemmingsen, 1973).
Hypnea musciformis has been harvested along the south-eastern coast of the United States, in Brazil, and in Senegal. It yields a kappa or furcellaran type carrageenan. It is no longer used by the major carrageenan manufacturers due to difficulty in processing and low yield.
Table 1 lists annual world production of carrageenophytes by producing area over the past 15 years. The large increase over this period is principally due to the growth of Eucheuma farming in the Philippines. In most other areas production has remained relatively static.
Table 1 Production of carrageenan seaweed world basis in metric tons
|
Countries |
1971 |
1979 |
1984 |
|
Canada |
6 000 |
5 700 |
5 000 |
|
Philippines |
500 |
14 000 |
25 000 |
|
Chile |
4 000 |
6 000 |
6 000 |
|
Indonesia |
4 000 |
3 500 |
3 000 |
|
Others |
5 500 |
4 500 |
4 500 |
|
Total |
20 000 |
33 700 |
43 500 |
CHEMICAL COMPOSITION
Carrageenans have the common feature of being linear polysaccharides with a repeating structure of alternating 1,3-linked b -D-galactophyranosyl arid 1,4-linked a -D-galactophyranosyl units (Figure 1). The 3-linked units occur as the 2- and 4-sulfate, or unsulfated, while the 4-linked units occur as the 2-sulfate, the 2,6-disulfate, the 3,6-anhydride, and the 3,6-anhydride-2-sulfate. Sulfation at C3 apparently never occurs. Pyruvate has been reported present in the carrageenans from some Gigartina species; these carrageenans have been termed "pi-earrageenan" (Hirase and Watanabe, 1972; DiNinno, McCandless and Bell, 1979; McCandless and Gretz, 1984). Methoxyl groups occur in sulfated polysaccharides from the Grateloupiaceae family (Hirase, Araki and Watanabe, 1967; Nunn and Parolis, 1968; Allsobrook, Nunn and Parolis, 1971). There is some question, though, as to whether these have the alternating structure characteristic of carrageenans (Parolis, 1981).
In their original work on fractionation of carrageenan from Chondrus crispus with potassium chloride Smith and Cook (1953) isolated two fractions which they named kappa- and lambda-carrageenan. Kappa was defined as that fraction which was precipitated by potassium chloride, while lambda was the fraction which remained in solution. Chemical studies on these fractions revealed that nearly half of the sugar units in kappa were 3,6-anhydro-D-galactose, a sugar not previously known to occur in nature, while lambda contained little or none of this sugar (Smith, Cook and Neal, 1954).
Due largely to the investigations by Rees and his co-workers (Rees 1963; Dolan and Rees, 1965; Anderson, Dolan and Rees, 1968, 1973; Anderson et al., 1968, 1968a; Lawson and Rees, 1968; Lawson et al., 1973; Penman and Rees, 1973, 1973a, 1973b;) carrageenans are now defined in terms of chemical structure. While it is true that more or less of a continuous spectrum of carrageenans exists (Pernas et al., 1967), it is nevertheless possible to distinguish a small number of ideal or limit polysaccharides. The names mu, kappa, nu, oita, lambda, theta, and xi are presently applied to these limit carrageenans. Figure 2 shows the repeating units of these polysaccharides. Mu and nu are believed to be precursors in the biosynthesis of kappa and iota respectively (Anderson et al., 1968b; Stancioff and Stanley, 1969), the transformation's being accomplished in the alga by an enzyme, "dekinkaase" (Lawson and Rees, 1970), or, in industrial processing, by the base-catalyzed Sn2 elimination of 6-sulfate (Stanley, 1963). Lambda likewise can be at least partially converted to theta-carrageenan by this reaction, but theta has yet to be identified as occurring naturally.
Xi-carrageenan, which constitutes the KCl-soluble fraction of some Gigartina species (G. chamissoi and G. canaliculata), has not been completely characterized but seems to differ from lambda in that the 1,3-linked units are unsubstituted at C6 (Penman and Rees, 1973a). Sugar units lacking sulfate at this position cannot be converted to the anhydride.
A new family of carrageenans for which the 3-linked units are unsulfated has recently been reported as the polysaccharide of Eucheuma gelatinae. This family consists of beta-carrageenan, analogous to kappa but lacking sulfate on C4 of the 1,3-linked units, and its precursor, gamma-carrageenan, analogous to mu (Greer and Yaphe, 1984).
Furcellaran, the polysaccharide from Furcellaria fastigiata, is very much like kappa-carrageenan, differing mainly in the amount of half-ester sulfate present. Furcellaran contains one sulfate group per three or four sugar units, as compared with one sulfate group per two sugar units for kappa-carrageenan. D-galactose 2-sulfate, D-galactose 4-sulfate, D-galactose 6-sulfate, and 3,6-anhydro-D-galactose 2-sulfate have been identified as components of furcellaran (Painter, 1966). The distribution of sulfate along the molecular chain is still not completely known.
Native furcellaran, like carrageenan, can be modified by treatment with hot alkali to cleave 6-sulfate, with formation of 3,6-anhydro-D-galactose units. As with kappa-carrageenan this results in an increase in water and milk gel strengths.
Precipitation of furcellaran with potassium chloride occurs at much lower concentrations than does precipitation of carrageenans (Smidsrød et al., 1967). This is due to furcellaran's having fewer charged groups and therefore being less hydrophilic than other carrageenans.
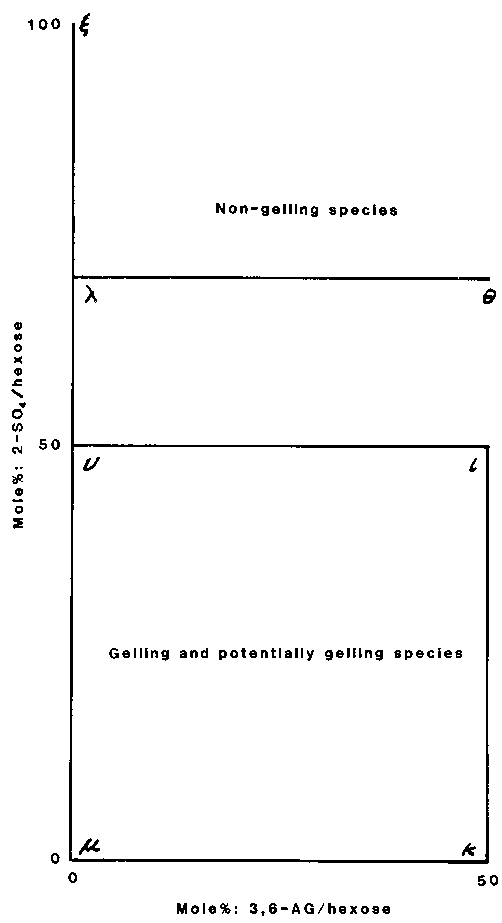
Native carrageenans from different algae may be regarded as varying mixtures of the limit polysaccharides and intermediate hybrids ranging in degree of anhydridation and 2-sulfation of the 1,4-linked units. This is shown graphically in Figure 3. This diagram divides the carrageenans into two general groups. One consists of mu, nu, kappa, iota, and their hybrids. Carrageenans in this group gel with potassium ions, or can be made to gel by treatment with alkali; they are characterized by having their 1,3-linked units either unsulfated or sulfated only at C4. The other group consists of lambda, xi, theta, and their hybrids; these do not gel either before or after alkali treatment and characteristically have both their 1,4- and 1,3-linked units sulfated at C2, though occasionally the latter are unsulfated (Stancioff and Stanley, 1969).
The complex fine structure of carrageenans is still an active field of research. Enzymic, immunological, and 13C NMR techniques have proved to be powerful tools for these investigations.
Figure 3 Composition of carrageenans. (Reproduced with permission from Handbook of water-soluble gums, edited by R.L. Davidson. New York, McGraw-Hill, 1980)
EXTRACTION PROCESSES
Specific details of extraction processes are closely guarded as trade secrets by the several manufacturers of carrageenans, but broadly these follow a similar pattern. Weed, usually dried and baled, is received at the processing location from the harvesting location. The shipment may be sampled and the sample subjected to a test extraction to evaluate the quality of the extractive. Other need quality factors such as contents of moisture, sand and salt, and non-carrageenophytes are evaluated at this stage. Obtaining a representative sample from a weed shipment is not a trivial exercise, as weed quality may vary widely not only from one shipment to the next but also within a shipment, due to factors over which the processor may have but limited control. Sampling protocols used generally represent what is feasible rather than what a statistician might regard as adequate.
Prior to plant-scale extraction the weed may be washed to remove adhering salts, sand, stones, and marine organisms. Washed, or unwashed, weed, usually as a blend selected to achieve the desired properties in the extractive, is then digested with hoc water under alkaline conditions to exhaustively extract the carrageenan. The alkali, usually calcium or sodium hydroxide, performs two functions: firstly it promotes swelling and maceration of the weed to aid in bringing the carrageenan into solution, while, secondly, when employed at sufficiently high concentrations, it effects cleavage of 6-sulfate groups from the carrageenan to generate 3,6-anhydro-D-galactose residues in the polysaccharide chain. These function to enhance the water gel strength and milk reactivity of the carrageenan. Maceration is promoted by agitation of the resultant paste. Conversion of 6-sulfated moieties to the 3,6-anhydride continues during resting of the paste at temperatures near 100°C.
When the desired degree of conversion has been achieved the solution of carrageenan is separated from weed solids by filtration, or by centrifugation followed by filtration. Concentration of the filtrate by evaporation, and adjustment of pH, are done prior to the recovery of the carrageenan from solution.
The foregoing processing operations inevitably involve some degradation of the polysaccharide, due to the rigors (e.g., heat, alkalinity) of processing. Although carrageenans are reasonably stable under the conditions of alkalinity encountered in processing a drop in pH can occur from the consumption of alkali for the neutralization of sulfuric acid formed by cleavage of half-ester sulfate groups. Saccharinic acids may also be formed through alkali-catalyzed "peeling" reactions.
Several methods have been used to recover the carrageenan from solution. Direct drying of the concentrated filtrate on steam-heated rolls has been used extensively. Products of much higher quality are obtained by precipitation of the carrageenan from solution by 2-propanol or other alcohols. An interesting historical note is that perhaps the earliest process described for recovering carrageenan from Irish moss employed alcohol precipitation (Bourgade, 1871).
Precipitation is followed by further alcohol washes to dehydrate the coagulum. Vibrating screens or basket centrifuges may be used to separate the coagulum from the alcohol following precipitation and each wash. Following the final wash the coagulum is dried under conditions permitting recovery of the residual alcohol.
The fibrous carrageenan from the dryer is ground and sifted to specified particle sizes which may range from 80 mesh to 270 mesh. This basic product, segregated into batches, is sampled and tested for compositional and functional properties (e.g., moisture, viscosity, gel strength).
Another process presently in use for the recovery of carrageenan from solution was originally developed for furcellaran production but is also employed for kappa-carrageenan. This cakes advantage of several properties common to furcellaran and kappa-carrageenan. First, solutions of these polysaccharides form gels in the presence of potassium ions. Second, these gels exude water by syneresis on standing, the more so when squeezed in a press. Third, much water separates from the gel when it is frozen and then allowed to thaw. The latter phenomenon is the same as that used for the production of agar.
In the case of Furcellaria, the weed may be treated in the cold with an alkaline solution for one or more weeks. This alkaline treatment removes colouring matter and some proteins and makes the gum more easily extractable. Some alkaline elimination of 6-sulfate may also occur during this treatment. Extraction follows a procedure generally similar to that described above for the alcohol process. Following concentration by evaporation the filtrate is extruded through spinnerets into a cold 1-1.5% solution of potassium chloride. The resulting gelled threads are further dewatered by subsequent potassium chloride washes followed by pressing. The gel is then frozen, thawed, chopped, again washed with fresh KCl solution, and air-dried.
A limitation of the freeze-thaw process as applied to carrageenans is that it is applicable only to furcellaran and kappa-carrageenan, which are the only types whose gels with potassium ions exhibit marked syneresis. Moreover the requirement that potassium be present precludes making products wherein sodium is the major counterion.
The "gel-press" process, used by some minor producers of carrageenans and agar, likewise relies on pressure to dewater the gel, but omits the freeze-thaw cycle.
The economics of extraction processes are strongly affected by the cost of the energy required to bring the carrageenan into solution and subsequently to recover it in dry form. This includes the heat necessary to digest and cook the seaweed, to concentrate the filtrate in evaporators, to dry the coagulum, and, in the case of alcohol precipitation, to recover the spent alcohol by distillation. Steam, generated by oil-fired boilers, is the usual source of process heat. Given the volatility of oil prices in the present-day market it will be appreciated that the cost of energy from this source has changed drastically in the past and can be expected to do so in the future. The impact of energy cost can of course be less in any locality where local low-cost fuels can be exploited. As an example, one major manufacturer of carrageenans uses locally-mined peat as a source of process heat.
High energy costs can be countered by employing cogeneration to supply the not inconsiderable electrical power requirements for plant operation. Alternatives to evaporation (e.g., ultrafiltration) have been investigated. These capital-intensive measures are not presently economical, but may be expected to become so in the likely event of rising energy costs.
An adequate supply of cheap, good quality, fresh water is an obvious prerequisite for the economical operation of an extraction process. In at least one instance a carrageenan factory was forced to relocate to another area when it was found chat its expanding demands exceeded the capacity of the local water supply.
Filtration must be employed if refined, completely water-soluble, products are to be produced. Owing to the viscosity of the extract and the swollen, gelatinous nature of the residual solids pressure filters are a necessity for efficient throughput and a filter aid must be added to the feed to prevent clogging ("blinding") of the filter medium and to afford a porous filter cake that will drain well and can be washed in the filter press to recover retained carrageenan. The filter aids most commonly employed are calcined diatomaceous earth and expanded volcanic glass. Since large amounts of aid are required the choice may depend on the location of the carrageenan processing plant with respect to the source of supply of aid.
Diatomaceous filter aids are available in a range of grades of retentiveness. A retentive aid may be used for a secondary "polish" filtration of the effluent from the primary filtration. This may be preceded by treatment of the primary filtrate with activated carbon to decolorize it. However this practice is now uncommon as the expense of these added steps usually cannot be recouped as added value of the product. Moreover the tendency of carbon to peptize and pass through even the most retentive filter can result in the product's having an undesirable grayish color.
It has been estimated that for a carrageenan extraction plant to be economical it should have a capacity of at least 450-750 metric Cons of product annually. This would require processing 1 400-2 300 MT of dried seaweed. To allow for variations in the harvest, available seaweed sources should exceed these tonnages by 40-50%. Initial investment for procurement, production, and marketing has been estimated to be $ 4 500 000 - $ 6 750 000. While these may be taken as ballpark estimates, obviously many factors other than scale of production determine whether a given operation will prove profitable. It has been recommended that any move toward production should be preceded by a three- to four-year pilot program of harvesting and selling dried seaweeds to other producers. This will serve to develop information required for starting an extraction plant (Moss, 1978).
A new process, and product, is semi-refined carrageenan. This process, which is distinguished by its low energy input, uses Eucheuma cottonii as a raw material.
Semi-processed E. cottonii is prepared by a method which superficially resembles that for French fried potatoes. A basket of seaweed fronds is immersed and cooked in hot aqueous potassium hydroxide and then soaked in fresh water to extract most of the residual alkali. The product is dried and ground to produce a flour having many of the properties of conventional extracted carrageenans.
The economic advantage lies in not extracting the carrageenan from the seaweed but rather performing the reaction which maximizes gel strength upon the polymer within the plant structure. By doing this the ratio of process water to product is minimized, thereby reducing the cost of isolating the dry product.
The rationale for the process is as follows: the kappa-carrageenan in E. cottonii does not dissolve in hot water if the concentration of gel-inducing cations (e.g., K+, Ca++) is maintained at a level corresponding to a melting temperature for the in situ carrageenan gel which is greater than the processing temperature. If this is done the carrageenan will imbibe water to form a gel structure like that in the living plant. The reagent (OH- ions) is able to diffuse into this structure and produce the chemical modification desired.
PROPERTIES
The chemical reactivity of carrageenans is primarily due to their half-ester sulfate groups which are strongly anionic, being comparable to inorganic sulfate in this respect. The free acid is unstable, and commercial carrageenans are available as stable sodium potassium and calcium salts or, most commonly, as a mixture of these. The associated cations together with the conformation of the sugar units in the polymer chain determine the physical properties of the carrageenans. For example, kappa- and iota-carrageenans form gels in the presence of potassium or calcium ions whereas lambda-carrageenan does not.
Reactivity with proteins is exhibited by both gelling and nongelling carrageenans, although regularity of the polymer is an important factor. In most, if not all, cases ion-ion interactions between the sulfate groups of the carrageenan and the charged groups of the protein are involved. Reaction depends on protein/carrageenan net charge ratio, and thus is a function of the isoelectric point of the protein, the pH of the system, and the weight ratio of carrageenan to protein (MacMullan and Eirich, 1963). At pH levels below the isoelectric point the protein has a net positive charge and thus can undergo direct electrostatic interaction with the negatively-charged carrageenan. The commercially important reaction of carrageenan with kappa-casein in milk is specific for this protein and unique in that it can occur at pH levels above the isoelectric point of the casein. It appears to be due to a region of positively charged amino acid residues in the kappa-casein molecule which can interact electrostatically with the sulfate groups of the carrageenan even though the net charge of the casein is negative (Snoeren et al., 1975).
The functionality of carrageenans in various applications depends largely on their rheological properties. Carrageenans, as linear, water-soluble, polymers, typically form highly viscous aqueous solutions. This is due to their unbranched, linear macromolecular structure and polyelectrolytic nature. The mutual repulsion of the many negatively charged half-ester sulfate groups along the polymer chain causes the molecule to be highly extended, while their hydrophilic nature causes it to be surrounded by a sheath of immobilized water molecules. Both of these factors contribute to resistance to flow.
Viscosity depends on concentration, temperature, the presence of other solutes, and the type of carrageenan and its molecular weight. Viscosity increases nearly exponentially with concentration. This behaviour is typical of linear polymers carrying charged groups and is a result of the increase with concentration of interaction between polymer chains. Salts lower the viscosity of carrageenan solutions by reducing the electrostatic repulsion among the sulfate groups. This behaviour is likewise normal for ionic macromolecules. At low temperature and high enough salt concentration, however, carrageenan solutions may gel, with an increase in apparent viscosity. This is particularly true for the strongly gel-inducing cations, K+ and Ca++. At high temperatures, however, Ca++ lowers viscosity to a greater extent than does Na+ or K+.
Viscosity decreases with temperature. Again, the change is exponential. It is reversible provided that heating is done at or near the stability optimum at pH 9, and is not prolonged to the point where significant thermal degradation occurs. Both gelling (kappa-, iota-) and nongelling (lambda-) carrageenans behave in this manner at temperatures above the gelling point of the carrageenan. On cooling, however, the gelling types will abruptly increase in apparent viscosity when the gelling point is reached, provided that the counter-ions (K+ and Ca++) promotive of gelation are present.
Viscosity increases with molecular weight in accordance with the Mark-Houwink equation:
[h] = KMa
where [h] is intrinsic viscosity, M is an average molecular weight (since carrageenans are polydisperse) and K and a are constants. Intrinsic viscosities correlate well with practical viscosity measurements taken at 1.5% concentration and 75°C.
Commercial carrageenans are available in viscosities ranging from about 5 mPa.s to 800 mPa.s when measured at 1.5% concentration and 75°C. Carrageenan solutions having viscosities less than 100 mPa.s have flow properties very close to Newtonian. At higher viscosities the solutions exhibit shear-thinning behaviour and it becomes necessary to specify the shear rate at which the measurement is taken. Where non-Newtonian behaviour is expected viscosity measurements should be made at a shear rate comparable to that encountered in the application considered.
Carrageenans specifically tailored for water-thickening applications are usually lambda types or the sodium salt of mixed lambda and kappa. They dissolve in either cold or hot water to form viscous solutions. There high water viscosities are desirable, and the high molecular weight and hydrophilicity of lambda contribute to this. For gelling applications a low viscosity in hot solution is usually desirable for ease in handling, and, fortunately, high gel strength carrageenans (mixed calcium and potassium salts of kappa or iota) fulfill this requirement because of their lesser hydrophilicity and the effect of the calcium ions.
Kappa- and iota-carrageenans and furcellaran form gels on cooling of their hot solutions in the presence of certain cations, notably K+ and Ca++. Heating is required to bring them into solution under these conditions. According to Rees (1972) carrageenans which form aqueous gels do so because of double helix formation. At temperatures above the melting point of the gel thermal agitation overcomes the tendency to form helices and the polymer exists in solution as random coils. On cooling the polymer chains become interlinked through double helix formation to form "domains" (Morris, Rees and Robinson, 1980). This occurs regardless of the counterions present and does not directly lead to gelation. Only when potassium or other gel-promoting cations are present will the domains aggregate to form a three-dimensional network. An alternative model of carrageenan gelation, based on cation-induced aggregation of single helices has also been proposed (Paoletti, Smidsrød and Grasdalen, 1984).
Regardless of the mechanism it appears that the occurrence of 1,4-linked 6-sulfated residues in the polymer chain of either kappa- or iota-carrageenan detracts from the strength of their gels. This is ascribed to kinks, produced by these residues, in the chain which inhibit the formation of double helices (Mueller and Rees, 1968). Alkali modification of the carrageenan during processing increases the gel strength of the product by removal of these kinks through conversion of 6-sulfated residues to the 3,6-anhydride. Increased hydrophobicity from the added anhydride residues may also contribute to gelation.
Kappa-carrageenan and furcellaran gels are relatively rigid and are subject to syneresis. Incorporation of locust bean galactomannan along with kappa or furcellaran yields a more compliant gel. "Smooth" regions of the mannan chain (i.e., regions carrying no galactose side groups) are believed to bind to the double helices of the kappa to reduce their tendency to aggregate (Rees, 1972). Iota-carrageenan by itself yields compliant gels with very little tendency to undergo syneresis. Here the 2-sulfate groups on the 3,6-anhydride residues act as wedging groups to prevent the tightly-packed aggregation believed responsible for the rigidity of kappa gels. Whereas potassium is more effective than calcium in inducing gelation of kappa the reverse is true for iota-carrageenan.
All carrageenans have the ability to form gels by cooling a solution of the carrageenan in hot milk. Even lambda-carrageenan, which does not gel in water regardless of the cations present, will form a gel at levels of 0.2% or more by weight of the milk. This gelation is ascribed to the formation of carrageenan-casein bonds, as previous described.
With kappa- and iota-carrageenan as well as furcellaran there is, in addition to the carrageenan-casein interaction, a water-gelling effect from the cations present in the carrageenan as well as the Ca++ and K+ present in the milk. These cations appear to be required for milk gel formation as milk which has been ion-exchanged to remove Ca++ and K+ does not gel with the sodium salts of lambda, kappa, or iota. On the other hand the strength of milk gels is enhanced by the addition of soluble calcium and potassium salts in a manner quite similar to that in water gels.
The presence of fat influences the behaviour of carrageenans in milk. Strongly gelling kappa-carrageenans can be used in high-fat systems but are not well tolerated in low-fat systems wherein they may exert a destabilizing action resulting in whey separation. For the latter, weak milk-gelling kappa-carrageenans with high ester sulfate and moderate to high 3,6-anhydride are more suitable. The reason that strongly-gelling types can be employed in high-fat but not low-fat systems is due in part to the disperse fat phase. This apparently tempers the carrageenan-casein complex, serving to interrupt aggregation to some extent. Interaction may also occur between carrageenan and the phospholipid which is present as a monomolecular layer covering the disperse globules of butterfat in the milk. Since the phospholipid contains basic amino groups with which the ester sulfate groups of the carrageenan can react it is very likely that electrostatic bonds are formed between the carrageenan and phospholipid. This may account for the extraordinary effectiveness with which very low levels (ca. 50 ppm) of carrageenan stabilize evaporated milk against fat separation (Moirano, 1977).
Carrageenans are susceptible to depolymerization through acid-catalyzed hydrolysis. At high temperatures and low pH this may rapidly lead to complete loss of functionality. They can be used in acid systems, however, if not subjected to prolonged heating. The rate of hydrolysis at a given pH and temperature is markedly lower if the carrageenan is in the gel rather than the sol state. This can be achieved by ensuring that gel-promoting cations are present at sufficient concentration to raise the gel melting temperature above the temperature to which the carrageenan will be subjected.
Carrageenan is listed by the U.S. Food and Drug Administration (FDA) as Generally Recognized as Safe (GRAS) (U.S. Food and Drug Administration, 1979). Following reports of cecal and colonic ulceration in guinea pigs and rabbits induced by a highly degraded carrageenan provided, ironically, for the symptomatic relief and cure of peptic and duodenal ulcers in man, intensive investigations into the safety of carrageenans were carried out by the FDA and other groups sponsored by the carrageenan industry. By lace 1976 food grade carrageenan, defined as having a water viscosity of no less than 5 mPa.s at 1.5% concentration and 75°C (U.S. Food and Nutrition Board, 1981) had been demonstrated to be safe.
Carrageenans have been shown not to be teratogenic (Collins, Black and Prew, 1977, 1977a, 1979). A study conducted on rats and hamsters at the Eppley Institute for Cancer Research demonstrated chat carrageenans are not carcinogenic. A review of the physiological effects of carrageenans has been published (Stancioff and Renn, 1975).
CURRENT USES
Carrageenans are used to gel, thicken, or suspend; therefore they are used in emulsion stabilization, for syneresis control, and for bodying, binding and dispersion. Major uses are in foods, particularly dairy applications. Tables 2 and 3 list dairy and water-based applications respectively (Guiseley, Stanley and Whitehouse, 1980).
Furcellaran generally finds applications similar to those for kappa-carrageenan. Historically, furcellaran has dominated two major European application fields: Tarte or cake glaze powders and flan powders. Today the special properties of excellent gel texture and flavour release make furcellaran a preferred product for use in milk pudding powders.
Carrageenan is unique in its ability at very low concentration (ca. 300 ppm) to suspend cocoa in chocolate milk; no other gum has been found to match it. A very delicate milk gel structure, undetectable on pouring or drinking the milk, is believed to hold the cocoa in suspension. A substantial differential ("spread") between the concentration at which settling of cocoa occurs and that at which visible gelation is evident is required for practical stabilization. This is achieved by careful selection of weed type and quality.
The use of iota-carrageenan in dessert gel formulations affords gels which have textures very similar to those of gelatin gels. They have an advantage over gelatin gels in that their melting point is higher, so that they find a ready market in tropical climates or where refrigeration is not available. This is offset to some extent by the different mouth-feel, since these gels do not "melt in the mouth", as does gelatin. A further advantage is that iota gels retain their tender structure on aging, whereas gelatin tends to toughen. This is important for ready-to-eat desserts, an item popular in Europe.
Kappa-carrageenan or furcellaran by itself is unsatisfactory for dessert gel applications due to the "short", brittle structure of its gel. This can be ameliorated by the incorporation of locust bean galactomannan into the formulation, and kappa-locust bean or iota- kappa-locust bean blends are also offered for this application. To achieve sparkling-clear gels it is necessary to use a locust bean gum which has been clarified by filtration. The clarified gum is produced for this purpose by several of the major carrageenan manufacturers.
In toothpastes carrageenans function as a "binder" to impart the desired rheological properties to the paste and to provide the cosmetic quality of "sheen". Toothpastes consist of ingredients which interact in complex and poorly understood ways and the carrageenan often must be carefully tailored to achieve satisfactory performance in a particular formulation. Carrageenan suffers severe competition in the U.S. domestic market from sodium carboxymethylcellulose, a much cheaper gum. Despite this, business has been retained - and regained - due to the superior quality and appearance carrageenan imparts to a toothpaste. Outside the United States carrageenan has maintained a strong position in this application, due, among other factors, to its immunity to degradation by enzymes which attack cellulose gums.
Table 2 Typical milk (dairy) applications of carrageenan
|
Use |
Function |
Product |
Approx. use level, % |
|
|
Frozen desserts: |
||||
|
|
Ice cream, ice milk |
Whey prevention |
|
|
|
Control meltdown |
Kappa- |
0.010-0.030 |
||
|
Pasteurized milk products: |
||||
|
|
Chocolate, eggnog, fruit-flavored |
Suspension, bodying |
Kappa- |
0.025-0.035 |
|
Fluid skim milk |
Bodying |
Kappa-, iota-, |
0.025-0.035 |
|
|
Filled milk |
Emulsion stabilization bodying |
Kappa-, iota- |
0.025-0.035 |
|
|
Creaming mixture for cottage cheese |
Cling |
Kappa- |
0.020-0.035 |
|
|
Sterilized milk products: |
||||
|
|
Chocolate, etc. |
Suspension, bodying |
Kappa- |
0.010-0.035 |
|
Controlled calorie |
Suspension, bodying |
Kappa- |
0.010-0.035 |
|
|
Evaporated |
Emulsion stabilization |
Kappa- |
0.005-0.015 |
|
|
Infant formulations |
Fat and protein stabilization |
Kappa- |
0.020-0.040 |
|
|
Milk gels: |
||||
|
|
Cooked flans or custards |
Gelation |
Kappa-, Kappa- + iota- |
0.20-0.30 |
|
Cold prepared custards (with added TSPP) |
Thickening, gelation |
Kappa-, iota- lambda- |
0.20-0.50 |
|
|
Pudding and pie fillings (starch base) Dry mix cooked with milk |
Level search gelatinization |
Kappa- |
0.10-0.20 |
|
|
Ready-to-eat |
Syneresis control, bodying |
Iota- |
0.10-0.20 |
|
|
Whipped products: |
||||
|
|
Whipped cream |
Stabilize overrun |
Lambda- |
0.05-0.15 |
|
Aerosol whipped cream |
Stabilize overrun stabilize emulsion |
Kappa- |
0.02-0.05 |
|
|
Cold prepared milks: |
||||
|
|
Instant breakfast |
Suspension, bodying |
Lambda- |
0.10-0.20 |
|
Shakes |
Suspension, bodying, stabilize overrun |
Lambda- |
0.10-0.20 |
|
|
Acidified milks: |
||||
|
|
Yogurt |
Bodying, fruit suspension |
Kappa- + locust bean gum |
0.20-0.50 |
(Reproduced with permission from Food colloids, edited by H.D. Graham. Westport, Connecticut, AVI Publishing Co., Inc. (1977), and Handbook of water-soluble gums, edited by R.L. Davidson. New York McGraw-Hill Book Co. (1980))
Table 3 Typical water applications of carrageenan
|
Use |
Function |
Carrageenan type |
Approx. use level, % |
|
Dessert gels |
Gelation |
Kappa- + iota- |
0.5-1.0 |
|
Kappa- + iota- + locust bean gum |
|||
|
Low-calorie Jellies |
Gelation |
Kappa- + iota- |
0.5-1.0 |
|
Kappa- + galactomannans |
|||
|
Pet-foods (canned) |
Fat stabilization, thickening, suspending gelation |
Kappa- + locust bean gum |
0.2-1.0 |
|
Fish gels |
Gelation |
Kappa- + locust bean gum |
0.5-1.0 |
|
Kappa- + iota |
|||
|
Syrups |
Suspension, bodying |
Kappa- + lambda- |
0.3-0.5 |
|
Fruit drink powders and frozen concentrates |
Bodying |
Sodium kappa-, lambda- |
0.1-0.2 |
|
Pulping effects |
Potassium calcium kappa- |
0.1-0.2 |
|
|
Relishes, pizza, barbecue sauces |
Bodying |
Kappa- |
0.2-0.5 |
|
Imitation milk |
Bodying, fat stabilization |
Iota-, lambda- |
0.03-0.06 |
|
Imitation coffee creams |
Emulsion stabilization |
Lambda- |
0.1-0.2 |
Whipped toppings (artificial) |
Stabilize emulsion, overrun |
Kappa-, iota- |
0.1-0.3 |
|
Puddings (nondairy) |
Emulsion stabilization |
Kappa- |
0.1-0.3 |
|
Air-treatment gels |
Gelation |
Kappa- + iota- |
2.0-3.5 |
|
Kappa- + galactomannans |
|||
|
Toothpastes |
Binder |
Sodium kappa-, lambda-, iota- |
0.8-1.2 |
|
Lotions |
Bodying, emollient |
Sodium kappa-, lambda-, iota- |
0.2-1.0 |
|
Suspensions (industrial) |
Suspension |
Iota- |
0.2-1.0 |
|
Dispersions |
Suspension, dispersion |
Hydrolyzed kappa-, lambda-, iota- |
0.2-0.5 |
|
Water-based paints |
Suspension, flow control emulsion stabilization |
kappa-, + galactomannans, Iota- |
0.15-0.5 |
(Reproduced with permission from Food colloids, edited by H.D. Graham. Westport, Connecticut, AVI Publishing Co., Inc. (1977), and Handbook of water-soluble gums, edited by R.L. Davidson, New York, McGraw-Hill Book Co. (1980))
Carrageenan is used in combination with locust bean and guar galactomannans as a gelling agent in pet foods. However extracted carrageenans have now been almost entirely replaced by semi-refined carrageenan from Eucheuma cottonii (Table 4).
Another major application of the gelling properties of carrageenans is in air freshener gels. The demand for carrageenan in this market peaked in the 1970s and has undergone some erosion since then, but still persists at a fairly stable level.
PRODUCTS
Basically there are three types of carrageenan of commercial importance. These are kappa-, iota-, and lambda-carrageenan. Furcellaran may be considered to be an extreme kappa type. These commercial extractives approximate to the limit polysaccharides, their criteria being functionality rather than strict chemical characterization.
Batches of the basic carrageenans are tested for their functional properties and then blended to produce standardized products. Diluents, usually sucrose or glucose, may be added for standardization. Food grade salts, such as potassium chloride or citrate, and other gums, particularly locust bean gum, may be incorporated in the blend to achieve desired functional properties. In all, more than two hundred different carrageenan and furcellaran blends, tailored to meet specific applications, are presently offered by the several manufacturers, as well as blending houses, under their various trade names.
MARKETING
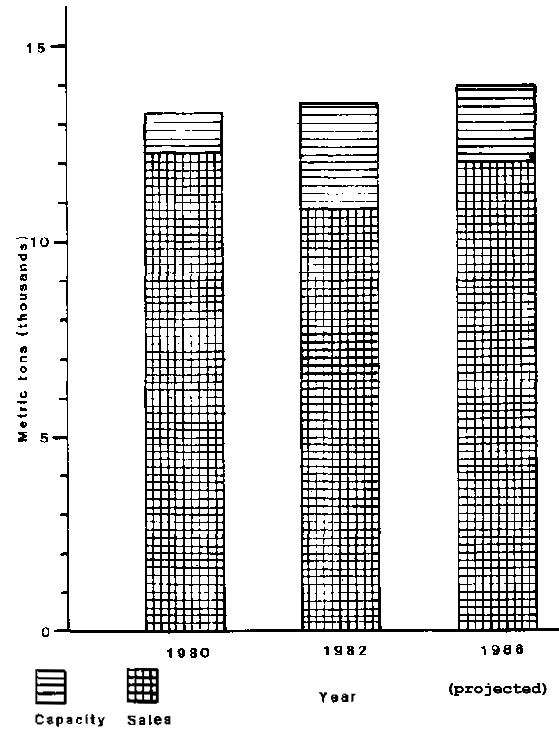
Sales of carrageenans in millions of kilograms and dollars are shown in Table 4. Figures are shown for both extractive and sime-refined carrageenan. The latter is used almost exclusively for the huge pet food market, and its explosive growth in the 1980-82 period reflects the replacement of extractive by semi-refined in pet food formulations. This changeover has now been accomplished and steady, but unexciting, growth is now projected for both types. The growth rate reflects the maturity of the food processing industry which is the staple outlet for carrageenans. Although the number of new food products introduced annually doubled from 1 026 in 1970 to 2 200 in 1985 volume demand does not increase accordingly. Table 5 shows the distribution of extractive sales by end use and by geographical region. Figure 4 shows that a moderate excess of production capacity over sales exists.
Table 4 World market for refined and semi-refined carrageenan
|
Carrageenan |
Market size 1982 |
Volume growth, % |
Compound volume growth, %/yeara/ |
|
|
kg MM |
$MM |
80-82 |
83-88 |
|
|
Refined |
10.8 |
88.3 |
-6.1 |
1.8 |
|
Semi-refined |
2.4 |
10.1 |
600 |
4.0 |
|
Both |
13.2 |
98.4 |
3.6 |
2. 2 |
a/ Projected
Table 5 Distribution of carrageenan sales
|
By end use |
Percent |
By region |
Percent |
|
Dairy |
52 |
Europe |
45 |
|
Water gel |
16 |
North America |
23 |
|
Other food |
10 |
Latin America |
12 |
|
Non-food |
22 |
Far East |
20 |
Figure 4 Carrageenan extract capacity vs. sales
Speciality gums, such as carrageenans, are sold on the basis of their functionality in specific applications and not as commodities. For this reason the major carrageenan manufacturers devote substantial portions of their budgets to maintaining active applications and technical marketing groups to serve the ever-changing needs of their customers. There is no room for complacency in the carrageenan industry.
FUTURE PROSPECTS
After nearly fifty years of development the carrageenan industry can now be said to have come of age and to be a mature industry. As previously mentioned its close ties to the food processing industry, likewise in its maturity, presages that future growth should be steady, if unspectacular. New products and applications can be expected, but these may be slow in coming. An insight into the time scale may be gained from the observation that the last "new" application for carrageenan of any great commercial significance was air freshener gels in the early 1970s. Opportunities exist wherever the functionality of carrageenans can confer advantages not possessed by cheaper competitive gums.
A favorable factor has been the stabilization of seaweed supplies and prices due to the advent of Eucheuma farming. Barring political upheavals in the harvesting regions the industry can remain assured of adequate supplies of good quality weed at reasonable prices.
Future progress now appears to lie in the areas of achieving cost reduction in processing and developing more versatile and better quality-controlled products.
REFERENCES
Allsobrook, A.J.R., J.R. Nunn and H. Parolis, 1971. Sulphated polysaccharides of the Grateloupiaceae family. Part 5. A polysaccharide from Aeodes ulvoidea. Carbohydr.Res., 16:71-8
Anderson, N.S., T.C.S. Dolan and D.A. Rees, 1968. Carrageenans. Part 3. Oxidative hydrolysis of methylated kappa-carrageenan and evidence for a masked repeating structure. J.Chem.Soc.(C), 1968:596-601
Anderson, N.S., T.C.S. Dolan and D.A. Rees, 1973. Carrageenans. Part 7. Polysaccharides from Eucheuma spinosum and Eucheuma cottonii. The covalent structure of iota-carrageenan. J.Chem.Soc.Lond.Perkins Trans.I, 1973:2173-6
Anderson, U.S., et al., 1968. Carrageenans. Part 4. Variations in the structure and gel properties of kappa-carrageenan, and the characterization of sulphate esters by infrared spectroscopy. J.Chem.Soc.(C), 1968:602-6
Anderson, U.S., et al., 1968a. Carrageenans. Part 5. The masked repeating structures of lambda- and mu-carrageenans. Carbohyd.Res., 7:468-73
Bjerre-Petersen, E., J. Christensen and P. Hemmingsen, 1973. Furcellaran. In Industrial gums, edited by R.L. Whistler and J.N. BeMiller. New York, Academic Press, pp.123-36
Bourgade, G., 1871. Improvement in treating marine plants to obtain gelatine and c. U.S. Patent 112,535.
Chen, L. C-M., et al., 1973. The ratio of kappa- to lambda-carrageenan in nuclear phases of the rhodophycean algae, Chondrus crispus and Gigartina stellata. J.Mar. Biol.Assoc.U.K., 53:11-6
Collins, T.F.X., T.N. Black and J.H. Prew, 1977. Long term effects of calcium carrageenan in rats. 1. Effects on reproduction. Food Cosmet.Toxicol., 15:533-8
Collins, T.F.X., T.N. Black and J.H. Prew, 1977a. Long term effects of calcium carrageenan in rats. 2. Effects on fetal development. Food Cosmet. Toxicol., 15:539-45
Collins, T.F.X., T.N. Black and J.H. Prew, 1979. Effects of calcium and sodium Carrageenans and iota-carrageenan on hamster foetal development. Food Cosmet.Toxicol., 17:443-9
Deslandes, E., et al., 1985. Evidence for lambda-carrageenans in Solieria chordalis (Solieriaceae) and Callibepharis jubata, C. ciliata and Cystoclonium purpureum (Rhodophyllidaceae). Bot.Mar., 28:317-8
DiNinno, V., E.L. McCandless and R.A. Bell, 1979. Pyruvic acid derivative of a carrageenan from a marine red alga (Petrocelis species). Carbohydr.Res., 71:C1-4
Dolan, T.C.S. and D.A. Rees, 1965. The Carrageenans. Part 2. The positions of the glycosidic linkages and sulphate esters in lambda-carrageenan. J.Chem.Soc. 1965:3534-9
Doty, M.S., 1973. Farming the red seaweed Eucheuma for Carrageenans. Micronesica, 9:59-73
Doty, M.S. and G.A. Santos, 1978. Carrageenans from tetrasporic and cystocarpic Eucheuma species. Aquat.Bot., 4:143-9
Eisses, J., 1952. The research of gelatinous substances in Indonesian seaweeds at the Laboratory for Chemical Research, Bogor. J.Sci.Res.Indon., 1:44-9
Eisses, J., 1953. Seaweeds in the Indonesian trade. Indon.J.Nat. Sci., 1(2-3):41-56
Greer, C.W. and W. Yaphe, 1984. Characterization of hybrid (beta-kappa-gamma) carrageenan from Eucheuma gelatinae J. Agardh (Rhodanhyta, Solieriaceae) using carrageenases, infrared and 13C-nuclear magnetic resonance spectroscopy. Bot.Mar., 27:473-8
Guiseley, K.B., N.F. Stanley and P.A. Whitehouse, 1980. Carrageenan. In Handbook of water-soluble gums, edited by R.L. Davidson. New York, McGraw-Hill Book Co., pp.5-1 to 5-30
Hirase, S. and K. Watanabe, 1972. The presence of pyruvate residues in lambda-carrageenan and a similar polysaccharide. Bull. Inst.Chem.Res.Kyoto Univ., (50):332-6
Hirase, S., C. Araki and K. Watanabe, 1967. Component sugars of the polysaccharide of the red seaweed Grateloupia elliptica. Bull.Chem.Soc.Japan, 40:1445-8
Lawson, C.J. and D.A. Rees, 1968. Carrageenans. Part 6. Reinvestigation of acetolysis products of lambda-carrageenan. Revision of the structure of a -1,3-galactotriose, and a further example of the reverse specificities of glycoside hydrolysis and acetolysis. J.Chem.Soc.(C), 1968:1301-4
Lawson, C.J. and D.A. Rees, 1970. An enzyme for the metabolic control of polysaccharides conformation and function. Nature, Lond., 227:390-3
Lawson, C.J., et al., 1973. Carrageenans. Part 8. Repeating structures of galactan sulphates from Furcellaria fastigiata, Gigartina canaliculata, Gigartina atropurpurea, Ahnfeltia durvillaei, Gymnogongrus furcellatus, Eucheuma cottonii, Eucheuma spinosum, Eucheuma isiforme, Eucheuma uncinatum, Agardhiella tenera, Pachymenia hymantophora and Gloiopeltis cervicornis. J.Chem.Soc.Lond.Perkins Trans.1, 1973:2177-82
Lund, S. and E. Bjerre-Petersen, 1952. Industrial utilization of Danish seaweeds. Proc.Int.Seaweed Symp., 1:85-7
Lund, S. and E. Bjerre-Petersen, 1961. Collection and utilization of Danish Furcellaria, 1946-60. Proc.Int.Seaweed Symp., 4:410-1
MacMullan, E.A. and F. Eirich, 1963. The precipitation reaction of carrageenan with gelatin. J.Colloid Sci., 18:526-37
McCandless, E.L. and M.R. Gretz, 1984. Biochemical and immunochemical analysis of carrageenans of the Gigartinaceae and Phyllophoraceae. Hydrobiologia, 116/117:175-8
McCandless, E.L., J.A. West and M.D. Guiry, 1982. Carrageenan patterns in the Phyllophoraceae. Biochem.System.Biol., 10:275-84
McCandless, E.L., J.A. West and M.D. Guiry, 1983. Carrageenan patterns in the Gigartinaceae. Biochem.System.Ecol., 11:175-82
Moirano, A., 1977. Sulfated seaweed polysaccharides. In Food Colloids, edited by H.D. Graham, Westport, Connecticut, AVI Publishing Co., pp.347-81
Morris, E.R., D.A. Rees and G. Robinson, 1980. Cation-specific aggregation of carrageenan helices: domain model of polymer gel structure. J.Mol.Biol., 138:349-62
Moss, J.A., 1978. Essential considerations for establishing seaweed extraction factories. In The marine plant biomass of the Pacific Northwest coast, edited by R. Krauss. Corvallis, Oregon State University Press, pp. 301-14
Mueller, G.P. and D.A. Rees, 1968. Current structural views of red seaweed polysaccharides. In Drugs from the sea, edited by H.D. Freundenthal. Washington, D.C., Marine Technology Society, pp.241-55
Nunn, J.R. and H. Parolis, 1968. A polysaccharide from Aeodes orbitosa. Carbohydr.Res., 6:1-11
Painter, T.J., 1966. The location of the sulphate half-ester groups in furcellaran and kappa-carrageenan. Proc.Int.Seaweed Symp., 5:305-13
Paoletti, S., O. Smidsrød and H. Grasdalen, 1984. Thermodynamic stability of the ordered conformation of carrageenan polyelectrolytes. Biopolymers, 23:1771-94
Parolis, H., 1981. The polysaccharides of Phyllymenia hieroglyphica (=P. belangeri) and Pachymenia hymantophora. Carbohydr. Res., 93:261-7
Penman, A. and D.A. Rees, 1973. Carrageenans. Part 9. Methylation analysis of galactan sulphates from Furcellaria fastigiata, Gigartina canaliculata, Gigartina chamissoi, Gigartina atropurpurea, Ahnfeltia durvillaei, Gymnogongrus furcellatus, Eucheuma isiforme, Eucheuma uncinatum, Aghardhiella tenera, Pachymenia hymantophora and Gloiopeltis cervicornis. Structure of xi-carrageenan. J.Chem.Soc.Lond.Perkins Trans.1, 1973:2182-7
Penman, A. and D.A. Rees, 1973a. Carrageenans. Part 10. Synthesis of 3,6-di-0-methyl-D-galactose, a new sugar from the methylation analysis of polysaccharides related to xi-carrageenan. J.Chem.Soc.Lond.Perkins Trans.1, 1973: 2188-91
Penman, A. and D.A. Rees, 1973b. Carrageenans. Part 11. Mild oxidative hydrolysis of kappa- and lambda-carrageenans and the characterization of oligosaccharide sulphates. J.Chem.Soc.Lond.Perkins Trans.1, 2191-6
Pernas, A.J., et al., 1967. Chemical heterogeneity of Carrageenans as shown by fractional precipitation with potassium chloride. Acta Chem.Scand., 21:98-110
Rees, D.A., 1963. The carrageenan system of polysaccharides. Part 1. The relation between the kappa- and lambda-components. J.Chem.Soc., 1963:1821-32
Rees, D.A., 1972. Mechanism of gelation in polysaccharide systems. In Gelation and gelling agents. London, British Food Manufacturing Research Association, Symposium proceedings, 13:7-12
Smidsrød, O., et al., 1967. The effect of alkali treatment on the chemical heterogeneity and physical properties of some Carrageenans. Acta Chem.Scand., 21:2585-98
Smith, D.B. and W.H. Cook, 1953. Fractionation of carrageenin. Arch.Biochem.Biophys., 45:232-3
Smith, D.B., W.H. Cook and J.L. Neal, 1954. Physical studies on carrageenin and carrageenin fractions. Arch.Biochem.Biophys., 53:192-204
Smith, H.M., 1905. The utilization of seaweeds in the United States. Bull.U.S.Bur.Fish., 24:169-71
Snoeren, T.H.M., et al., 1975. Electrostatic interaction between kappa-carrageenan and kappa-casein. Milchwissenschaft, 30:393-6
Stancioff, D.J. and D.W. Renn, 1975. Physiological effects of carrageenan. In Physiological effects of food carbohydrates, edited by A. Jeanes and J. Hodge. Washington, D.C., American Chemical Society, pp.282-95
Stancioff, D.J. and N.F. Stanley, 1969. Infrared and chemical studies on algal polysaccharides. Proc.Int.Seaweed Symp., 6:595-609
U.S. Food and Drug Administration, 1979. Carrageenan, salts of carrageenan, and Chondrus extract (carrageenin); withdrawal of proposal and termination of rulemaking procedure. Fed.Reg., 44:40343-5
U.S. Food and Nutrition Board, 1981. Committee on Codex Specification, Carrageenan. In Food chemicals codex. Washington, D.C., National Academy Press, pp. 74-5 3rd.ed.
Zaneveld, J.S., 1955. Economic marine algae of tropical south and east Asia and their utilization. Spec.Publ.IPFC, (3):55 p.
Zaneveld, J.S., 1959. The utilization of marine algae in tropical south and east Asia. Econ.Bot., 13(2):89-131