Risk assessment, along with risk management and risk communication, is one of the components of risk analysis, which can be defined as an overall strategy for addressing risk. The importance of an overlap between these three elements (risk assessment, risk management and risk communication) is well recognized, but some functional separation is also necessary. In relation to risk assessment, such separation ensures that issues are addressed in a transparent manner with a scientific basis.
The FAO/WHO Codex Alimentarius Commission (CAC) defines risk assessment as a scientifically based process consisting of four steps:
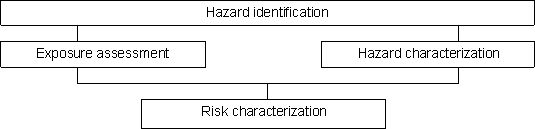
1. Hazard identification, which is the identification of the biological agent that may be present in a particular food or group of foods and capable of causing adverse health effects.
2. Hazard characterization, which is the qualitative or quantitative, or both, evaluation of the nature of the adverse health effects associated with the biological agents that may be present in food, and in such cases a dose-response assessment should be performed if the data are obtainable.
3. Exposure assessment, which is the qualitative or quantitative, or both, evaluation of the likely intake of the biological agent through food, as well as through exposure from other sources, if relevant.
4. Risk characterization, namely the qualitative or quantitative, or both, estimation, including attendant uncertainties, of the probability of occurrence and severity of known or potential adverse health effects in a given population based on hazard identification, exposure assessment and exposure assessment.
These steps are illustrated schematically in Figure 1.1. The risk assessment process is a means of providing an estimate of the probability and severity of illness attributable to a particular pathogen-commodity combination. The four-step process enables this to be carried out in a systematic manner, but the level of detail in which each step is addressed will depend on the scope of the risk assessment. This should be defined clearly by the risk manager through ongoing dialogue with the risk assessor.
Figure 1.1. Schematic representation of the risk assessment process.
Undertaking a risk assessment is recognized as a resource-intensive task requiring a multidisciplinary approach. While MRA is becoming an important tool for assessing the risks to human health from foodborne pathogens, and can be used in the elaboration of standards for food in international trade, it is not within the capacity of many, perhaps even most, countries to carry out a complete quantitative MRA. Yet foodborne illness is among the most widespread public health problems and creates social and economic burdens as well as human suffering, making it a concern that all countries need to address, and risk assessment is a tool that can be used in the management of the risks posed by foodborne pathogens. At the same time, risk assessment can also be used to justify the introduction of more stringent standards for imported foods. A knowledge of MRA is therefore also important for trade purposes, and there is a need to provide countries with the tools for understanding and, if possible, carrying out MRA.
Risk analysis has evolved over the last decade within CAC. Since the Uruguay Round Trade Agreement on the Application of Sanitary and Phytosanitary Measures (SPS) entered into force in 1995, the importance of risk analysis has increased. Risk analysis is now considered to be an integral part of the decision-making process of Codex. CAC has adopted definitions of risk analysis terminology related to food safety, and statements of principle relating to the role of food safety risk assessment. Furthermore, in 1999, it adopted the Principles and Guidelines for the Conduct of Microbiological Risk Assessment (CAC, 1999a), which were developed by the Codex Committee on Food Hygiene (CCFH).
In addition to these developments in risk assessment, the 22nd Session of CAC requested FAO and WHO to convene an international advisory body on the microbiological aspects of food safety in order to address MRA in particular (CAC, 1997). In response to this, and as follow-up to previous activities in the area of risk analysis, FAO and WHO convened an expert consultation in March 1999 to examine in an international forum the issue of MRA. The main outcome of this expert consultation was an outline strategy and mechanism for addressing MRA at the international level (WHO, 1999). Subsequently, at its 32nd Session, in November 1999, CCFH recognized that there are significant public health problems related to microbiological hazards in foods (CAC, 1999b). It identified 21 pathogen-commodity combinations of concern, and prioritized these according to criteria such as the significance of the public health problem, the extent of the problem in relation to geographical distribution and international trade, and the availability of data and other information with which to conduct a risk assessment. CCFH suggested that FAO and WHO convene ad hoc expert consultations to provide advice on MRA, and also recommended that these consultations be conducted according to the format outlined at the 1999 expert consultation (WHO, 1999).
The need by member countries for advice on risk assessment in order to reduce the risk to consumers of becoming ill from food, and to meet their obligations imposed under World Trade Organization (WTO) agreements, combined with the requests of CAC and CCFH for scientific advice on MRA, led FAO and WHO to undertake a programme of activities to address at international level the issue of MRA. The aim of the joint programme is to provide a transparent review of scientific opinion on the state of the art of MRA, and to develop the means to achieve sound quantitative risk assessments of specific pathogen-commodity combinations. It also aims to create awareness of the risk assessment process, provide information and advice, and develop tools that could be used by countries in undertaking MRA.
This programme of MRA activities aims to serve two user groups: CAC; and FAO and WHO member countries. CAC requires sound scientific advice as a basis for the development of standards, guidelines and related texts for the management of risks posed by microbiological hazards in foods. Member countries, in contrast, need adaptable risk assessment tools to use in conducting their own assessments and, if possible, some modules directly applicable to their national situation.
Taking these needs into account, FAO and WHO initiated work on three of the pathogen-commodity combinations identified as priority issues: Salmonella Enteritidis in eggs; Salmonella in broiler chickens; and Listeria monocytogenes in ready-to-eat foods. In order to facilitate communication with risk managers, a two-year process of work was introduced (Figures 1.2a, b). Problems such as the lack of a clear-cut risk management question at the outset, and limitations in the usefulness of a global risk estimate, were recognized and addressed to the extent possible in the course of the work.
Figure 1.2a. Year 1 of the FAO/WHO process for undertaking microbiological risk assessment.
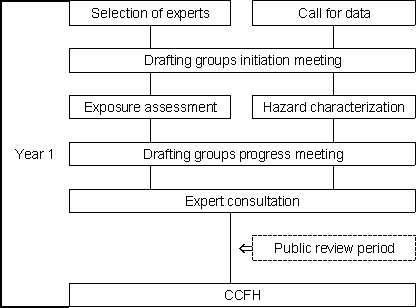
The risk assessments of Salmonella in eggs and broiler chickens began in January 2000. Risk assessment work on L. monocytogenes in ready-to-eat foods was undertaken concurrently. In the first year of the process, drafting groups were established to examine available information and prepare technical documentation on the hazard identification, exposure assessment and hazard characterization components of the risk assessment. These documents were then reviewed and evaluated by a joint expert consultation in July 2000 (FAO, 2000). That expert consultation made recommendations for the improvement of the preliminary documents, identified knowledge gaps and information requirements needed to complete the risk assessment work, and developed a list of issues to be brought to the attention of CCFH. The report of that consultation was presented to the 33rd Session of CCFH in order to inform risk managers regarding the progress of the risk assessment and to seek more precise guidance on the needs of risk managers. A number of specific risk management questions were identified by the Committee (CAC, 2000) and these issues were subsequently addressed in the completion of the risk assessment. The documentation was also made available for public comment as a means of reviewing the preliminary work.
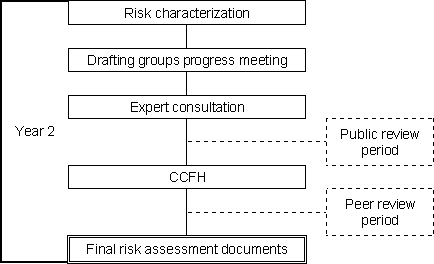
The second year of the process focused on the completion of the risk assessment by undertaking the risk characterization step. Again, the risk characterization document that was developed was critically reviewed by an expert consultation convened in April-May 2001 in Rome (FAO, 2001). The report of this consultation, which included preliminary answers to the questions posed by CCFH, was presented to the 34th Session of the Committee.
Figure 1.2b. Year 2 of the FAO/WHO process for undertaking MRA.
The risk assessment initially set out to understand how the incidence of human salmonellosis is influenced by various factors, from the agricultural phase of chicken meat and egg production, through marketing, processing, distribution, retail storage, consumer storage and meal preparation, to final consumption. Such models are appealing because they permit the study of the broadest range of intervention strategies. However, as the work progressed it became evident that the quantity and quality of information available from all sources was not sufficient to allow the construction of a full and expansive model. Thus the final scope of the Salmonella risk assessment, and the components of the food production and consumption continuum that were considered, became:
1. Salmonella enterica serotype Enteritidis in eggs. This risk characterization estimates the probability of human illness due to Salmonella Enteritidis (SE) following the ingestion of a single food serving of internally contaminated shell eggs, either consumed as whole eggs, egg meals or as ingredients in more complex food (e.g. cake). This work addressed selected aspects of egg production on farms, further processing of eggs into egg products, retail and consumer egg handling, and meal preparation practices. Risk reductions for specific intervention strategies were also estimated.
2. Salmonella enterica (multiple serotypes) in broiler chickens. This risk characterization estimates the probability of acute gastroenteritis per person per serving and per year, due to the ingestion of Salmonella enterica on fresh whole broiler chicken carcasses with the skin intact and which are cooked in the domestic kitchen for immediate consumption. This work commences at the conclusion of slaughterhouse processing, and considers in-home handling and cooking practices, including cross-contamination events. The effects of pre-slaughter interventions and the slaughter process are not currently included in this model. However, for any intervention strategy, whether at farm or during processing, that reduces the prevalence or numbers, or both, of Salmonella on poultry or carcasses by a measurable quantity, the amount of risk reduction can be calculated from the risk model, and examples are provided.
Risk estimates for S. Enteritidis in eggs and S. enterica in broiler chickens used a common dose-response model. Within the hazard-characterization step, the objectives were to produce one or more curves describing the probability that an individual would become ill versus the dose of Salmonella ingested within food.
Human-host-adapted, predominantly invasive Salmonella serotypes (e.g. S. Typhi, S. Paratyphi) were not considered in developing the dose-response model. As noted, the outcome of interest was defined as acute gastroenteritis. Hence, disease outcomes that may occur beyond the diagnosis of gastroenteritis were not included in the risk estimations, but are described in hazard identification. Similarly, severity of disease outcomes attributable to multiresistant strains of Salmonella were not estimated, nor for the more highly invasive Salmonella serotypes that are not commonly associated with poultry, i.e. S. Dublin and S. Cholerasuis. Cost-benefit analysis of risk reduction interventions was not included in the risk management charge to the risk assessors, and thus is beyond the scope of this work.
In practice, one would start with the model and then look for information. In fact, during the course of drafting meetings, the team considered the model at an earlier stage. However, considering the purpose and the task of international risk assessment, the team needed to look at data at the same time. In this document, data used with modelling structures is first explained, followed by the mathematical modelling of inputs.
The writing format differs between the Exposure assessments and the Risk characterizations of S. Enteritidis in eggs and S. enterica in broiler chickens. This difference is derived from different approaches taken for these commodities. In the egg exposure assessment, previously reported risk assessments are critically reviewed and compared. Therefore one could look back into the original reports to see the details of input parameters to the model. In contrast, the broiler exposure assessment is written to describe a desirable structure of exposure assessment for this pathogen-commodity combination, without any referable assessment. The models and their parameters are described in Section 6.4.
CAC [Codex Alimentarius Commission]. 1997. Report of the 22nd Session of the Joint FAO/WHO Codex Alimentarius Commission. Geneva, 23-28 June 1997.
CAC. 1999a. Principles and Guidelines for the Conduct of Microbiological Risk Assessment. CAC/GL-30.
CAC. 1999b. Report of the 32nd Session of the Codex Committee on Food Hygiene. Washington, DC, 29 November-4 December 1999.
CAC. 2000. Report of the 33rd Session of the Codex Committee on Food Hygiene. Washington, DC, 23-28 October 2000.
FAO [Food and Agriculture Organization of the United Nations]. 2000. [Report of the] Joint FAO/WHO Expert Consultation on Risk Assessment of Microbiological Hazards in Foods. FAO headquarters, Rome, 17-21 July 2000. FAO Food and Nutrition Paper, 71.
FAO. 2001. Joint FAO/WHO Expert Consultation on Risk Assessment of Microbiological Hazards in Foods. Risk characterization of Salmonella spp. in eggs and broiler chickens and Listeria monocytogenes in ready-to-eat foods. FAO Food and Nutrition Paper, 72.
WHO [World Health Organization]. 1999. Risk assessment of microbiological hazards in foods. Report of a Joint FAO/WHO Expert Consultation. Geneva, 15-19 March 1999.