
Animal health

23 May 2024, 08:30 hours; Rome
Overview
This update covers avian influenza viruses (AIV) with zoonotic potential occurring worldwide, i.e. H5Nx, H7Nx high pathogenicity avian influenza (HPAI) viruses and H3N8, H5Nx, H6N1, H7Nx, H9N2, H10Nx and H11 low pathogenicity avian influenza (LPAI).
Specific information is available for Avian Influenza A(H7N9) virus viruses and Sub-Saharan Africa HPAI in related FAO Avian Influenza situation updates.
HPAI outbreaks in animals officially reported since last update (25 April 2024): in total, 91 outbreaks/events have been reported in five geographic regions caused by H5Nx (5), H5N1 (79), H5N5 (4), H7N3 (1)* and H7N9 (1)*. (see Table 1 for details)
LPAI events in animals officially reported since the last update (25 April 2024): 0 new event was reported.
Number of human cases officially reported since last update (25 April 2024): 3 new events were reported.1,2,3
* corrected on 24 May 2024.
1 https://www.chp.gov.hk/files/pdf/2024_avian_influenza_report_vol20_wk15.pdf
2 https://www.health.vic.gov.au/health-advisories/human-case-of-avian-influenza-bird-flu-detected-in-returned-traveller-to-victoria
3 https://www.cdc.gov/media/releases/2024/s0522-human-case-h5.html
Map 1. Global distribution of AIV with zoonotic potential* observed since 1 October 2023 (i.e. current wave)
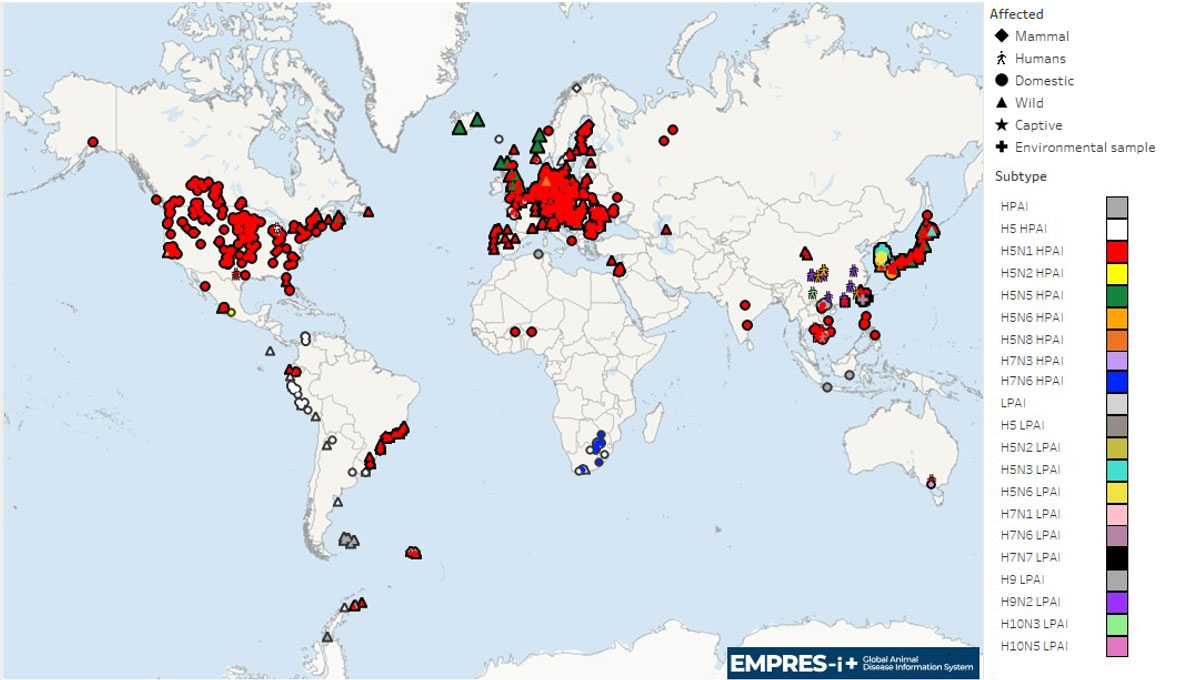
Note: Symbols may overlap for events in similar geographic locations.
Map 2. Global distribution of AIV with zoonotic potential* observed in the period 1 October 2022 to 30 September 2023 (i.e. previous wave)
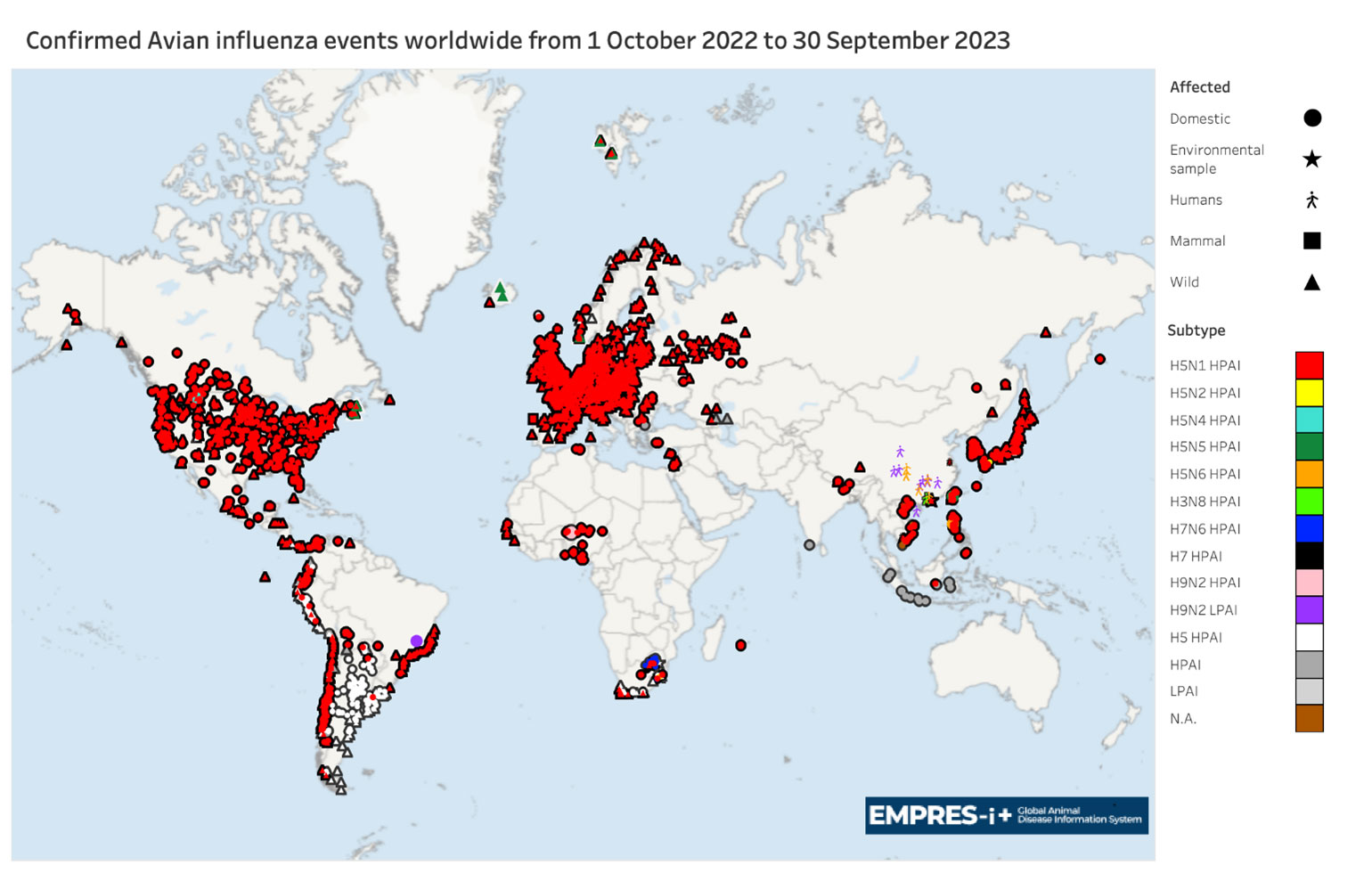
Note: Symbols may overlap for events in similar geographic locations.
Table 1. High pathogenicity avian influenza viruses with zoonotic potential reported since the last update
| Virus | Country/Area | Last observed outbreak | # events reported since the last update | Total # events reported since 1 October 2023 | Species affected during the reporting preiod |
|---|---|---|---|---|---|
H5 |
China |
17/05/2024‡ |
2 |
2 |
Pallas's gull |
South Africa |
29/02/2024 |
1 |
31 |
Poultry |
|
United States of America3 |
23/04/2024 |
2 |
101 |
Rock pigeon |
|
H5N1 |
Brazil |
23/04/2024 |
4 |
58 |
Cabot's tern, Common Tern |
Bulgaria |
10/05/2024 |
2 |
16 |
Poultry |
|
Canada1 |
08/04/2024 |
26 |
98 |
American Black Duck, American Crow, American goshawk, Anatidae, Black-billed Magpie, Blue-winged Teal, Brent Goose, Canada Goose, Great Horned Owl, Horned Grebe, Mallard, Peregrine Falcon, Red-tailed Hawk, Rough-legged Buzzard, Snow Goose, Tundra Swan, Turkey Vulture, Velvet Scoter; Red Fox, Striped skunk |
|
Finland |
15/12/2023 |
1 |
71 |
Arctic fox |
|
Germany |
18/04/2024 |
1 |
186 (incl. |
Anatidae |
|
Hungary |
26/04/2024 |
1 |
135 |
Poultry |
|
Japan |
28/04/2024 |
5 |
138 (incl. |
Chicken; Large-billed crow, Mallard, Mountain hawk-eagle, Steller’s sea eagle |
|
Slovenia |
19/04/2024 |
1 |
14 |
Mute Swan |
|
United States of America3 |
16/05/2024‡ |
38 |
2 283 |
Turkey, Poultry, WOAH Non-Poultry, Live Bird Market; Bald eagle, Black-billed magpie, Common grackle, Hawk, Horned grebe, Red-tailed hawk, Rock dove, Rock pigeon; Bobcat, Cat, Cattle, Red fox, Virginia opossum |
|
H5N5 |
Canada |
08/04/2024 |
3 |
7 |
American Crow, Striped Skunk |
Japan |
30/04/2024 |
1 |
31 |
Large-billed crow |
|
H7N3 |
Australia4 |
23/05/2024‡ |
1* |
1* |
Poultry |
H7N6 |
South Africa |
18/12/2023 |
1 |
81 |
Poultry |
H7N9* |
Australia4 |
24/05/2024‡ |
1 |
1 |
Poultry |
Data was retrieved from WOAH WAHIS portal, government websites. Data cut off time: reported on 22 February 2024 8:30 CEST. $:estimate. ‡: date of confirmation.
The full list of bird and mammalian species affected by H5Nx HPAI are available HERE.
Notes: Only those reporting events in animals since 24 August 2023 are listed in the table, those reporting for the first time since 1 October 2023 in orange. Codes: D:domestic, C:captivity, W:wild birds, F: Feral, E:Environment, M: mammalian species other than humans, example: W123 indicates 123 wild birds affected. EA: Fully Eurasian; rEN: Reassortment Eurasian and North American.
1 the total includes events with sample collection date since 1 October 2023 data issued from the Canada Food and Inspection Agency dashboard [link]
2 a detailed list of wild bird species affected, consult weekly findings report on avian influenza in wild birds from Animal and Plant Health Agency (APHA) [link]
3 for more information, consult dedicated webpage of the USDA Animal and Plant Health Inspection Service (USDA/APHIS) [link]
4 for more information, consult dedicated webpage of the Department of Energy, Environment and Climate Action, Victoria, Australia [link]
§: British Antarctic Survey (BAS) [link]
Recent publications
WILD BIRDS
Azat, C., Alvarado-Rybak, M., Aguilera, J.F. & Benavides, J.A. 2024. Spatio-temporal dynamics and drivers of highly pathogenic avian influenza H5N1 in Chile. Front Vet Sci, 11:1387040. reference
Muñoz, G., Ulloa, M., Alegría, R., Quezada, B., Bennett, B., Enciso, N., Atavales, J., et al. 2024. Stranding and mass mortality in humboldt penguins (Spheniscus humboldti), associated to HPAIV H5N1 outbreak in Chile. Prev Vet Med, 227:106206. reference
Hall, V., Cardona, C., Mendoza, K., Torchetti, M., Lantz, K., Bueno, I. & Franzen-Klein, D. 2024. Surveillance for highly pathogenic avian influenza A (H5N1) in a raptor rehabilitation center-2022. PloS One, 19(4):e0299330. reference
Provencher, J.F., Brown, M.G.C., Hargan, K., Lang, A.S., Lapen, D., Lewis, H., Mallory, M.L., et al. 2024. Pathogen Surveillance in Swallows (family Hirundinidae): Investigation into Role as Avian Influenza Vector in Eastern Canada Agricultural Landscapes. J Wildl Dis, 2024 Apr 26. reference
Ahrens, A.K., Jónsson, S.R., Svansson, V., Brugger, B., Beer, M., Harder, T.C. & Pohlmann, A. 2024. Iceland: an underestimated hub for the spread of high-pathogenicity avian influenza viruses in the North Atlantic. J Gen Virol, 105(5). reference
Cho, A.Y., Si, Y.J., Lee, D.Y., Kim, D.J., Kim, D., Jeong, H., Song, C.S. & Lee, D.H. 2024. Index case of H5N1 clade 2.3.4.4b highly pathogenic avian influenza virus in wild birds, South Korea, November 2023. Front Vet Sci, 11:1366082. reference
DOMESTIC
Barnes, A.P., Sparks, N., Helgesen, I.S. & Soliman, T. 2024. Financial impacts of a housing order on commercial free range egg layers in response to highly pathogenic avian influenza. Prev Vet Med, 228:106209. reference
Ramsubeik, S., Stoute, S., Crossley, B., Rejmanek, D., Jerry, C., Jackson, W., Bland, M. & Ochoa, J. 2024. Natural Infection with H5N1 Highly Pathogenic Influenza (HPAI) Virus in 5- and 10-Day-Old Commercial Pekin Ducklings (Anas platyrhynchos domesticus). Avian Dis, 68(1):65-71. reference
Wolters, W.J., Vernooij, J.C.M., Spliethof, T.M., Wiegel, J., Elbers, A.R.W., Spierenburg, M.A.H., Stegeman, J.A. & Velkers, F.C. 2024. Comparison of the Clinical Manifestation of HPAI H5Nx in Different Poultry Types in the Netherlands, 2014-2022. Pathogens, 13(4):280. reference
Qui, N.H., Thu, N.T.A. & Linh, N.T. 2024. Factors affecting highly pathogenic avian influenza vaccination practices at poultry farms in Tra Vinh, Vietnam. Open Vet J, 14(3):794-804. reference
MAMMALS
[Preprint] Nguyen, T.Q., Hutter, C., Markin, A., Thomas, M., Lantz, K., Killian, M.L., Janzen, et al. 2024. Emergence and interstate spread of highly pathogenic avian influenza A(H5N1) in dairy cattle. bioRxiv, 2024.05.01:591751. reference
Burrough, E.R., Magstadt, D.R., Petersen, B., Timmermans, S.J., Gauger, P.C., Zhang, J., Siepker, C., et al. 2024. Highly Pathogenic Avian Influenza A(H5N1) Clade 2.3.4.4b Virus Infection in Domestic Dairy Cattle and Cats, United States, 2024. Emerg Infect Dis, 2024 Apr 29;30(7). reference
Uyeki, T.M., Milton, S., Abdul Hamid, C., Reinoso Webb, C., Presley, S.M., Shetty, V., Rollo, S.N., et al. 2024. Highly Pathogenic Avian Influenza A(H5N1) Virus Infection in a Dairy Farm Worker. N Engl J Med, 2024 May 3. reference
Meade, P.S., Bandawane, P., Bushfield, K., Hoxie, I., Azcona, K.R., Burgos, D., Choudhury, S., et al. 2024. Detection of clade 2.3.4.4b highly pathogenic H5N1 influenza virus in New York City. J Virol, 2024 May 15:e0062624. reference
Graziosi, G., Lupini, C., Catelli, E. & Carnaccini, S. 2024. Highly Pathogenic Avian Influenza (HPAI) H5 Clade 2.3.4.4b Virus Infection in Birds and Mammals. Animals (Basel), 14(9):1372. reference
Kwon, T., Trujillo, J.D., Carossino, M., Lyoo, E.L., McDowell, C.D., Cool, K., Matias-Ferreyra, F.S., et al. 2024. Pigs are highly susceptible to but do not transmit mink-derived highly pathogenic avian influenza virus H5N1 clade 2.3.4.4b. Emerg Microbes Infect, 2024 May 7:2353292. reference
Szaluś-Jordanow, O., Golke, A., Dzieciątkowski, T., Czopowicz, M., Kardas, M., Mickiewicz, M., Moroz-Fik, A., Łobaczewski, A., Markowska-Daniel, I. & Frymus, T. 2024. Upper Respiratory Tract Disease in a Dog Infected by a Highly Pathogenic Avian A/H5N1 Virus. Microorganisms, 12(4):689. reference
Tomás, G., Marandino, A., Panzera, Y., Rodríguez, S., Wallau, G.L., Dezordi, F.Z., Pérez, R., et al. 2024. Highly pathogenic avian influenza H5N1 virus infections in pinnipeds and seabirds in Uruguay: Implications for bird-mammal transmission in South America. Virus Evol, 10(1):veae031. reference
AI VIRUSES
Fusaro, A., Zecchin, B., Giussani, E., Palumbo, E., Agüero-García, M., Bachofen, C., Bálint, Á., et al. 2024. High pathogenic avian influenza A(H5) viruses of clade 2.3.4.4b in Europe-Why trends of virus evolution are more difficult to predict. Virus Evol, 10(1):veae027. reference
Ospina-Jimenez, A.F., Gomez, A.P., Osorio-Zambrano, W.F., Alvarez-Munoz, S. & Ramirez-Nieto, G.C. 2024. Sequence-based epitope mapping of high pathogenicity avian influenza H5 clade 2.3.4.4b in Latin America. Front Vet Sci, 11:1347509. reference
Lin, S., Chen, J., Li, K., Liu, Y., Fu, S., Xie, S., Zha, A., et al. 2024. Evolutionary dynamics and comparative pathogenicity of clade 2.3.4.4b H5 subtype avian influenza viruses, China, 2021-2022. Virol Sin, 2024 Apr 26:S1995-820X(24)00060-9. reference
Tare, D.S., Keng, S.S., Walimbe, A.M. & Pawar, S.D. 2024. Phylogeography and gene pool analysis of highly pathogenic avian influenza H5N1 viruses reported in India from 2006 to 2021. Arch Virol, 169(5):111. reference
Heo, G.B., Kang, Y.M., An, S.H., Kim, Y., Cha, R.M., Jang, Y., Lee, E.K., Lee, Y.J. & Lee, K.N. 2024. Concurrent Infection with Clade 2.3.4.4b Highly Pathogenic Avian Influenza H5N6 and H5N1 Viruses, South Korea, 2023. Emerg Infect Dis, 30(6). reference
Cho, A.Y., Si, Y.J., Kim, D.J., Seo, Y.R., Lee, D.Y., Kim, D., Lee, D., et al. 2024. Novel Avian Influenza A(H5N6) in Wild Birds, South Korea, 2023. Emerg Infect Dis, 30(6). reference
Reid, S.M., Byrne, A.M.P., Lean, F.Z.X., Ross, C.S., Pascu, A., Hepple, R., Dominguez, M., et al. 2024. A multi-species, multi-pathogen avian viral disease outbreak event: Investigating potential for virus transmission at the wild bird – poultry interface. Emerg Microbes Infect, 2024 Apr 30:2348521. reference
Lee, S.H., Jeong, S., Cho, A.Y., Kim, T.H., Choi, Y.J., Lee, H., Song, C.S., Nahm, S.S., Swayne, D.E. & Lee, D.H. 2024. Caught Right on the Spot: Isolation and Characterization of Clade 2.3.4.4b H5N8 High Pathogenicity Avian Influenza Virus from a Common Pochard (Aythya ferina) Being Attacked by a Peregrine Falcon (Falco peregrinus). Avian Dis, 68(1):72-79. reference
Souci, L. & Denesvre, C. 2024. Interactions between avian viruses and skin in farm birds. Vet Res, 55(1):54. reference
Murashkina, T., Sharshov, K., Gadzhiev, A., Petherbridge, G., Derko, A., Sobolev, I., Dubovitskiy, N., et al. 2024. Avian Influenza Virus and Avian Paramyxoviruses in Wild Waterfowl of the Western Coast of the Caspian Sea (2017-2020). Viruses, 16(4):598. reference
Thomazelli, L.M., Pinho, J.R.R., Dorlass, E.G., Ometto, T., Meneguin, C., Paludo, D., Frias, R.T., et al. 2024. Evidence of reassortment of avian influenza A (H2) viruses in Brazilian shorebirds. PloS One, 19(5):e0300862. reference
Wang, Y., Li, X., Lv, X., Li, Y., An, Q., Xiu, Y., Lv, X., et al. 2024. H6N2 reassortant avian influenza virus isolate in wild birds in Jiangxi Province, China. Virus Genes, 2024 May 9. reference
Amin, F., Mukhtar, N., Ali, M., Shehzad, R., Ayub, S., Aslam, A., Sheikh, A.A., et al. 2024. Mapping Genetic Markers Associated with Antigenicity and Host Range in H9N2 Influenza A Viruses Infecting Poultry in Pakistan. Avian Dis, 68(1):43-51. reference
Lai, C.C. & Hsueh, P.R. 2024. Human infection caused by avian influenza A (H10N5) virus. J Microbiol Immunol Infect, 2024 Apr 22:S1684-1182(24)00074-4. reference
Feoktistova, S., Sayganova, M., Trutneva, K., Glazova, O., Blagodatski, A.S., Shevkova, L., Navoikova, A., et al. 2024. Abundant Intra-Subtype Reassortment Revealed in H13N8 Influenza Viruses. Viruses, 16(4):568. reference
ASSAY
Bordes, L., Gonzales, J.L., Vreman, S., Venema, S., Portier, N., Germeraad, E.A., van der Poel, W.H.M. & Beerens, N. 2024. In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks. Viruses, 16(4):563. reference
SURVEILLANCE
Shemmings-Payne, W., De Silva, D., Warren, C.J., Thomas, S., Slomka, M.J., Reid, S.M., James, J., Banyard, A.C., Brown, I.H. & Ward, A.I. 2024. Repeatability and reproducibility of hunter-harvest sampling for avian influenza virus surveillance in Great Britain. Res Vet Sci, 173:105279. reference
Pinotti, F., Kohnle, L., Lourenço, J., Gupta, S., Hoque, M.A., Mahmud, R., Biswas, P., Pfeiffer, D. & Fournié, G. 2024. Modelling the transmission dynamics of H9N2 avian influenza viruses in a live bird market. Nat Commun, 15(1):3494. reference
Fujita, R., Tachi, T., Hino, M., Nagata, K., Saiki, M., Inumaru, M., Higa, Y., et al. 2024. Blowflies are potential vector for avian influenza virus at enzootic area in Japan. Sci Rep, 14(1):10285. reference
Increase surveillance efforts for the early detection of influenza viruses in poultry and dead wild species including certain mammals For full recommendations including non-avian species please see [link].
FAO's support to countries
Global level
- OFFLU is organising a webinar on the avian influenza vaccine matching (AIM) for poultry in July, to provide a technical overview, engage stakeholders and create a network and feedback forum for stakeholders.
- OFFLU has published an updated statement on the current situation of HPAI in dairy cows [link] and diagnostic guidance for HPAI in dairy cattle [link]
- Joint FAO/IAEA Centre (Nuclear Techniques in Food and Agriculture) (CJN) organized a webinar entitled “Highly Pathogenic Avian Influenza – UPDATE” on 9 May 2024.
- WOAH updated its High Pathogenicity Avian Influenza in Cattle webpage on 8 May [link]
- A three-day workshop of the Wildlife Health Intelligence Network (WHIN) was held in FAO Rome from 6 to 8 May. The network is working towards developing a global community of practice around wildlife surveillance which will be relevant to zoonotic influenza.
- FAO/WHO/WOAH released the Joint FAO/WHO/WOAH preliminary assessment of recent influenza A(H5N1) viruses on 26 April. [link]
- The UNESCO/CMS/FAO/Ramsar/IUCN webinar on AI and wildlife - How to protect wildlife from avian flu in UNESCO World Heritage sites, Biosphere Reserves and Ramsar sites – the 1st, 2nd, 3rd sessions were held in April – May. [link]
- OFFLU (WOAH/FAO network of expertise on animal influenza) has launched a webpage for collection of relevant links on HPAI Detections in Livestock. [link]
- The FAO Virtual Learning Centers have launched a new self-paced virtual learning course on avian influenza (AI). The course aims to raise awareness of AI and to develop capacity on its detection and prevention. This introductory course is intended to be useful for veterinarians, veterinary paraprofessionals and others working in the poultry industries. There are six short modules (15-20 minutes each), can be used as a ready-reference resource as needed. It supports multiple platforms including smartphones and tablets. [link] The course is now available also in Spanish and Portuguese.
Regional/country level
- Americas
- GF-TADs for the Americas held an informative electronic meeting of the Standing Group of Experts on Avian Influenza (SGE-AI) entitled ‘Detection of HPAI in Ruminants and Humans in the USA’ to share findings, ongoing research and future actions, following the detection and notification of Avian Influenza cases in dairy cattle and goats in the United States of America on 4 April. [link, recording] The second virtual meeting on H5N1 infection in dairy cows in the United States was organized by the GF-TADs for the Americas on 22 May.
- Risk communication materials e.g. flyers and posters were sent to the countries under the regional TCP project. While the regional emergency technical cooperation programme (TCP) project is closing by June 2024, a follow-up regional project is needed to support countries sustaining the achievement for long-term, FAO is seeking partners to mobilize resources.
- FAO Outbreak Costing Tool (OutCosT) has been implemented in Bolivia and Panama to estimate the economic impact of HPAI outbreaks.
- FAO Emergency Center for Transboundary Animal Disease Control (ECTAD) at regional and country (Colombia, El Salvador, Guatemala, Honduras and Peru) levels are supporting activities to respond to HPAI outbreaks e.g. simulations exercises, HPAI diagnostic trainings, information materials.
- Currently, Bolivia, Dominican Republic (for prophylaxis; no outbreak reported), Ecuador, Guatemala, Mexico, Peru and Uruguay are using vaccines to prevent and/or control HPAI, while some other countries are evaluating implementation.
- Africa
- FAO ECTAD regional offices in Eastern and Southern Africa (ESA) is supporting countries in the region to enhance biosecurity in poultry farms which contributes to minimising HPAI incursion. Poultry value chain risk assessment along the entire poultry value chain is planned in 10 countries namely Ethiopia, Kenya, Madagascar, Malawi, Mozambique, Rwanda, South Sudan, the United Republic of Tanzania, Uganda and Zambia as a part of ECTAD's ongoing support for HPAI risk mitigation/management.
- A regional training courses on infectious substance shipment for Eastern and Southern Africa will be held in Nairobi in June 2024.
- In Ethiopia, the Ministry of Agriculture has developed a comprehensive National Biosecurity Management Guideline for Poultry Farms.
- FAO ECTAD regional office in Western and Central Africa (WCA) held a Regional Training of Surveillance Evaluation Tool (SET) Evaluators in West and Central Africa in Abidjan (Cote D’Ivoire) from 25 to 29 March 2024.
- FAO ECTAD regional office in WCA organized a regional training workshop on good sampling practices at the Veterinary School in Dakar (EISMV) from 23 to 26 January with special attention to biosafety and biosecurity. A total of 18 participants from 10 countries in West, Central and East Africa received theoretical and practical training in collecting diagnostic specimens from poultry.
- The VLC in West Africa is providing a four-week Avian Influenza tutored course in English and French.
- A regional training courses on infectious substance shipment for Western and Central Africa will be held in Dakar (Fr) and Accra (En) in June - July.
- FAO ECTAD Benin supported epidemiological investigation of suspected avian influenza outbreak in Parakou. [link]; the General Directorate of Veterinary Services conducted active surveillance around the site.
- Burkina Faso released an alert for poultry farmers regarding HPAI outbreak in Ouagadougou on 6 March 2024. FAO has supported the veterinary services to conduct field investigation and implement response measures (Stamping out, carcass disposal, disinfection, quarantine and awareness creation). Support for laboratory reagents, consumables, personal protective equipment (PPE), as well as transport of samples to the reference laboratory (IZSVe) for confirmation and sequencing. Under the FAO TCP project on HPAI, eight HPAI specific tools has been developed, namely: Case Definition, Investigation Form, Notification Form, Outbreak Information/Management Form, Standard Operating Procedures (SOP) for sampling and diagnosis, communication during an HPAI crisis, donning/doffing PPE, biosecurity measures in HPAI suspected poultry farms.
- In Guinea, FAO ECTAD has supported suspected HPAI outbreak investigation in Labé, setting up active surveillance in a high-risk area and raising community awareness [link].
- Niger confirmed an outbreak of H5N1 HPAI by the Niamey Central Laboratory (LABOCEL) on 27 January 2024 on a family farm in Niamey, which has been the only confirmed case to date; a case of low-pathogenic avian influenza was also confirmed in a backyard guinea fowl farm in Niamey. FAO ECTAD provided with some PCR reagents and rapid test kits from the ECTAD stockpile project. Biosafety equipment (PPE), disinfectants, bags for biohazard waste), which FAO ECTAD prepared at the beginning of current influenza season under a HPAI prevention and control project, supported General Directorate of Veterinary Services in conducting rapid response.
- In Nigeria, H5N1 HPAI was confirmed in peacock farm in Kebbi State and in geese in Plateau State. FAO ECTAD supported the Federal Ministry of Agriculture and Food Security and other One Health stakeholders in conducting follow-up outbreak investigations and ensuring disease containment.
- In Senegal, a total of 350 swab samples of fresh droppings were collected from Djoudj National Bird Sanctuary, the Langue de Barbarie National Park, the Gueumbeul Wildlife Reserve, the Saloum Delta National Park, the Abéné Marine Protected Area, and the Palmarin Community Reserve in the regions of Saint-Louis, Fatick, and Ziguinchor. All samples tested negative for avian influenza. In addition to active surveillance, passive surveillance and vigilance in the main risk area continue to be implemented.
- North Africa and Middle East
- FAO ECTAD Egypt in the Regional office for Near East and North Africa (RNE) is supporting the implementation of the targeted risk-based surveillance plan for 2024 to understand the prevalence of Avian Influenza sub-types currently circulating in Egypt and to detect incursion of new subtypes as early as possible.
- FAO ECTAD Egypt supported the development of National One Health strategic framework which has been endorsed by the Government.
- Asia
- An H5N1 infection in human was confirmed in a child in Australia who travelled and presumed to have acquired the infection in India in March 2024 [link]. FAO ECTAD RAP is in contact with FAO country team to obtain more information on poultry in relation to this case.
- In Bangladesh, active surveillance at live bird markets was conducted in Dhaka and Chattogram.
- Since October 2023, Cambodia confirmed H5N1 HPAI in birds in seven provinces, while nine human cases reported. FAO Cambodia is collaborating National Animal Health and Production Research Institute (NAHPRI)/the General Directorate of Animal Health and Production (GDAHP) of the Ministry of Agriculture, Forestry and Fisheries, the Institut Pasteur du Cambodge (IPC), WHO and other partners, participated in joint risk assessment, and providing technical assistance, awareness materials for distribution, and some PPE. [link1, link2]
- FAO ECTAD Indonesia facilitated meetings with animal health expert commission and Directorate of Animal Health to update Avian Influenza clade 2.3.4.4b situation in mammals in Indonesia as a follow up of detected AI clade 2.3.4.b in dairy cattle in the United States; and a meeting involving Indonesian Veterinary Drug Commission and Directorate of Animal Health to recommend challenge strain for AIV H5N1 clade 2.3.4.4b to accommodate Indonesian private companies which produce vaccine for AI 2.3.4.4b.
- FAO ECTAD Myanmar in cooperation with Myanmar Livestock Federation organized two workshops entitled ‘Biosecurity, good animal husbandry practice (GAHP) and antimicrobial resistance (AMR) for farmers, feed sellers, and egg collectors’ in Ayeyarwady Region on 19 March and 9 April 2024. Similar workshops was also conducted in Yangon Region in January. FAO ECTAD Myanmar plans to organize a workshop for private veterinarians to encourage reporting practices especially the notifiable animal diseases in Myanmar under USAID funded project ‘Global Health Security Program: Immediate technical assistance for animal health systems to address emerging and priority zoonotic diseases and health threats in Myanmar’.
- FAO ECTAD Nepal have printed 10 000 copies of brochure on avian influenza to distribute in different parts of the country to increase awareness among practitioners and poultry farmers. Training on biosecurity measures for farmers is being conducted as well. Awareness training of animal health professionals on prioritized zoonotic diseases including avian influenza was held in Tanahun and Kailali districts.
- FAO ECTAD Papua New Guinea participated in an Avian Disease Workshop (including AIV with zoonotic potential) organized by the National Agriculture Quarantine and Inspection Authority (NAQIA - PNG Biosecurity) and the Australian Department of Agriculture, Fisheries and Forestry (DAFF) from 12 - 15 May in Port Moresby. The aim of the workshop was to strengthen field and diagnostic capacity to detect, diagnose and respond rapidly to such disease events.
- In the Philippines, laboratory capacity review was conducted in March using FAO Laboratory Mapping Tool (LMT) showed laboratory capacity of the government can be supported by regional/university/private laboratories when simultaneous outbreaks occur, and if equipment, manpower and technical supports are provided. Tabletop Simulation Exercise on AI and ASF was also conducted. FAO ECTAD, in collaboration with the Bureau of Animal Industry, organized a Coordination Meeting and Simulation Exercise for Animal Disease Emergencies (Avian Influenza and foot-and-mouth disease for Visayas on 12-16 February and South Luzon on 18-22 March. The meeting specifically discussed the Philippine’s Avian Influenza (AI) Vaccination Guidelines to seek inputs and agreement of local veterinarians and animal health workers. To date, the same activity has been conducted in 13 regions covering 60 provinces across the country and will be conducted in North Luzon clusters in April to cover the remaining 22 provinces in 4 regions.
- In Viet Nam, the Department of Animal Health and FAO ECTAD Viet Nam issued a joint press release ‘Stay vigilant with Highly Pathogenic Avian Influenza A(H5N1)’ upon the detection of new re-assorted HPAI A(H5N1) viruses in chickens and Muscovy ducks through active surveillance in Viet Nam [link]. In response to the finding, a vaccine efficacy test against the new virus and live bird market (LBM) surveillance is planned waiting the approval of the work plan. FAO ECTAD in Viet Nam participated in a joint risk assessment held on 4 April with inter-sectoral authorities, and provided updates on the HPAI situation in the country.
- Europe and Central Asia
- The Outbreak Costing Tool (OutCosT) to estimate the cost of outbreaks and their control is being adapted to poultry diseases and is being validated with real HPAI outbreak data from countries in three continents.
FAO Alerts
- On 14 September 2022, FAO issued an alert to Chief Veterinary Officers and FAO offices in Central America and South America regions on the risk of introduction and spread of H5NX HPAI [in English, French, and Spanish].
- On 8 April 2022, FAO issued an alert to Chief Veterinary Officers and FAO offices in Asia and the Pacific Region on the risk of a surge and spread of HPAI through increased poultry trade prior to and during Traditional New Year festivities in Asia.
- On 4 March 2022, FAO an alert to Chief Veterinary Officers and FAO offices in the Americas Region on the risk of introduction and spread of H5NX HPAI [in English, French, and Spanish].
- On 18 February 2022, FAO issued an alert to Chief Veterinary Officers, FAO offices, and wild bird partner organizations on the increased risk of HPAI outbreaks in wild bird populations in Africa.
- On 29 October 2021, FAO sent an alert message on the risk of H5Nx HPAI (re-)introduction along migratory flyways to Chief Veterinary Officers globally.
- On 13 November 2020, FAO sent an alert message on the risk of H5Nx HPAI re-introduction to Chief Veterinary Officers and FAO offices of at-risk countries in Africa region.
- On 09 October 2020, FAO sent an alert message on the risk of H5N8 HPAI re-introduction to Chief Veterinary Officers of at-risk countries in Europe, Middle East, and Western and Central Asia regions.
- On 17 January 2020, FAO released an alert on H5N8 HPAI in Eastern Europe to warn the Chief Veterinary Officers and FAO offices about the potential spread of the disease and advise on measures to take for prevention and control.
OFFLU
- Information on the OFFLU avian influenza matching pilot project. [link]
- OFFLU held an online discussion on 5 December 2022 to discuss the avian influenza situation in poultry and wild birds for experts to share experiences on the most recent wave of outbreaks in different countries. A summary is available. [link]
- The OFFLU published reports for the Vaccine Composition Meeting on avian influenza and swine influenza for February – September 2022.
- The Tripartite (FAO- WHO -WOAH) together with the WOAH/FAO Network of Expertise on Animal Influenza (OFFLU) has conducted a joint rapid risk assessment addressing the recent influenza A(H3N8) human infection in China in May 2022. [link]
- The OFFLU annual report for 2021 is now available. [link]
- Avian influenza report of the WOAH/FAO Network of expertise on animal influenzas (OFFLU) covering the period September 2021 – February 2022. [link]
- The OFFLU Network issued a statement on 24 December 2021 addressing the recent introduction of H5N1 HPAI in Canada. [link]
- The OFFLU network issued an avian influenza statement on 10 November 2021 addressing recent H5Nx high pathogenicity avian influenza virus reassortments. [link]
- The OFFLU Network issued the summary of the OFFLU call for avian influenza global situation held on 8 November 2021. [link]
- Avian influenza report of the OIE/FAO Network of expertise on animal influenzas (OFFLU) covering the period March – September 202. [link]
- As part of the OIE/FAO Network of Expertise on Animal Influenzas, FAO attended the Zoonotic Influenza Sessions of the WHO Vaccine Composition Meeting held from 2 to 4 March 2021. The report is now available online. [link]
- On 26 February 2021, the OFFLU issued a statement on High Pathogenicity Avian Influenza in the Russian Federation relating to its detection in poultry workers. [link]
- On 26 October 2020, the OFFLU issued a report on Highl Pathogenicity Avian Influenza in Kazakhstan describing the genetic characteristics of the latest H5N8 HPAI viruses detected recently in the country. [link]
WHO Vaccine Composition Meeting (VCM)
- Report of the WHO Vaccine Composition Meeting – February 2023. [link]
- Report of the WHO Vaccine Composition Meeting – September 2022. [link]
- Report of the WHO Vaccine Composition Meeting – February 2022. [link]
- Report of the WHO Vaccine Composition Meeting – September 2021. [link]
- Report of the WHO Vaccine Composition Meeting – February 2021. [link]
- Report of the WHO Vaccine Composition Meeting – Sept/Oct 2020. [link]
- Report of the WHO Vaccine Composition Meeting – February 2018. [link]
Global level
- International Alliance for Biological Standardization (IABS) held a meeting on 25-26 October 2022 addressing High Pathogenicity Avian Influenza Vaccination Strategies to prevent and control HPAI: Removing unnecessary barriers for usage. Conclusions and recommendations are now available. [link]
Regional/country level
America
- FAO organized a webinar on HPAI laboratory testing, under the framework of the GF-TADs and ‘Ask the experts’ for animal health laboratory staff in the region in March 2023. [link]
- Between 21 and 23 March 2023, FAO held a meeting in Santiago, Chile with the participation of the heads of official veterinary services from 8 Latin American countries that are part of the TCP project, as well as specialists, discussed the epidemiological situation of HPAI in their territories and the control measures implemented.
- The GF-TADs for the Americas hosted a technical meeting on HPAI vaccination: Approach, tools, knowledge and experience for the Americas held virtually in March 2023. [link]
- The first virtual meeting of the Standing Group of Experts on Avian Influenza (SGE-IA) took place online on 14 December 2022. Recommendations from this meeting can be found here. [link]
- FAO’s emergency Technical Cooperation Programme (TCP) project provides support to manage the outbreak of avian influenza in the region, as well as its impact on the most vulnerable households in the affected countries.
- FAO collated risk communication materials available at FAO in other regions globally and shared with FAO RLC.
- The first virtual meeting of the Standing Group of Experts on Avian Influenza (SGE-IA) took place in December 2022. Recommendations available [link] Dec 2022.
- FAO activated coordination and response protocols for the avian influenza outbreaks in the region. [link]
- FAO conducted a qualitative risk assessment for introduction of the H5N1 HPAI clade 2.3.4.4b virus from currently known infected countries in the Americas has been conducted.
- FAO is monitoring the situation closely through its network of decentralized offices and Reference Centers for Influenza to maintain close communication with members in Latin America and the Caribbean providing technical assistance and support as well as risk communication strategies and collaborating with resource partners to enhance preparedness and control of AI in the region. [link]
Asia
- FAO participated to the 7th World One Health Congress held on 8-11 November 2022 and presented preliminary results of the Qualitative Risk Assessment addressing H5 HPAI risk of introduction in Central America, South America, and the Caribbean. [link]
- International Alliance for Biological Standardization (IABS) held a meeting on 25-26 October 2022 addressing High Pathogenicity Avian Influenza Vaccination Strategies to prevent and control HPAI: Removing unnecessary barriers for usage. Conclusions and recommendations are now available [link].
- FAO ECTAD RAP organized a quarterly coordination call on 16 February 2023 with ECTAD countries in Asia to discuss progresses and challenges around avian influenza surveillance in the region.
- FAO RAP organized a quarterly coordination call on 8 December 2022 with ECTAD countries in Asia to discuss progresses and challenges around avian influenza surveillance in the region.
- The FAO-ECTAD Team in Viet Nam prepared a report entitled Economic analysis of enhanced biosecurity practices in three types of chicken farms in Northern Viet Nam [link].
- FAO RAP organized a regional Avian Influenza virtual meeting in November 2021. A summary can be found [link].
- FAO ECTAD RAP organized a quarterly coordination call on 16 February 2023 with ECTAD countries in Asia to discuss progresses and challenges around avian influenza surveillance in the region.
- FAO ECTAD RAP and IPC developed practical guidelines for field sequencing using MinIon.
- FAO ECTAD Indonesia held a Joint Risk Assessment (JRA) training on zoonotic priority diseases in West Java Province and in West Kalimantan Province.
- FAO ECTAD Cambodia organized AI surveillance review to share data from AI surveillance implementing partners, i.e. the results of AI surveillance in live bird markets, influenza-like illness (ILI) and severe acute respiratory infections (SARI) carried from 2020-2022 by CCDC, FAO, IPC, NAHPRI, NIPH and USCDC, to understand the challenges, lesson-learnt, and to do the AI surveillance resource mapping.
- FAO ECTAD Lao organized a refresher training on avian influenza surveillance and response in Louangprabang Province with participants from various partners i.e. provincial livestock and fisheries section involved in the avian influenza surveillance, Central Veterinary Services and laboratory, Division of Veterinary Legislation, public health sector including the Department of Communicable Disease Control, Information Education and Communication Department, and other development partners namely US CDC, WHO, Wildlife Conservation Society.
- FAO ECTAD Viet Nam organized a joint risk assessment (JRA) workshop for H5N6 (Dong Nai Province) and H5N8 (Lang Son Province).
- FAO RAP organized a regional Avian Influenza virtual meeting in November 2021 [report].
- FAO ECTAD Viet Nam published biosecurity handbooks for: Q&A Handbook, Chicken parent flocks, Broiler chicken, Ducks/Muscovy duck parent flocks, and Meat duck.
North Africa and Middle East
- FAO ECTAD Egypt and General Organization for Veterinary Services (GOVS) epidemiology unit updated the AI surveillance plan for January – December 2023 based on surveillance finding and risk mapping in 2022.
- FAO organized a workshop on Highly Pathogenic Avian Influenza in Libya [link].
Sub-Saharan Africa
- FAO ECTAD assisted Gambia sending samples to the reference laboratory (IZSVe-Italy) for sequencing. H5N1 2.3.4.4b was detected and the phylogenetic analyses confirmed that the H5N1 virus clusters with genotype BB recently detected in northern Italy in June 2023, suggesting a possible back-and-forth movement of viruses between Europe and Africa.
- FAO Emergency Centre for Transboundary Animal Diseases (ECTAD) regional offices in Eastern and Southern Africa (ESA) and West and Central Africa (WCA) organized 5 day regional training courses on Infectious Substances Shipment in Nairobi (June), Abidjan (July), Abuja (August). A total of 32 particpants from 13 countries successfully completed the training and were certified to ship infectious substances by air, in compliance with the applicable international regulations.
- FAO Ethiopia in collaboration with the Ethiopian Agricultural Research Institute has prepared a biosecurity brochure covering three key areas - conceptual, structural, and operational biosecurity measures. The brochure provides guidance to small and medium commercial poultry farms on implementing effective biosecurity measures for increased productivity, and a more sustainable and profitable industry, and is aimed to be used by Farmers Field School (FFS).
- In Kenya, FAO is supporting Kenya Animal Biosurveillance system (KABS) disease reporting platform roll-out for syndromic surveillance and the refresher trainings.
- FAO Burkina Faso trained 175 staff on HPAI epidemiological surveillance; conducted Training of Trainers (ToT) course on on good poultry farming practices, hygiene and biosecurity measures on farms; the 46 trainers conducted sensitization of 300 model poultry farmers from 10 regions, and also 30 communicators and journalists of the press on HPAI under TCP project.
- In Togo, FAO is supported HPAI outbreak response by providing technical assistance and supporting field outbreak investigation missions. A training of 25 agents on disease reporting / early warning using FAO Event Mobile Application (EMA-i) takes place soon.
- FAO Emergency Management Center (EMC-AH) expert mission in May 2023 visited Saint-Louis as a part of support to control HPAI emergency.
- FAO ECTAD Côte d’Ivoire supported the disinfection of poultry markets in Abidjan.
- FAO EMC-AH conducted field mission in Gabon from 4 to 8 July 2022 in response to the recent H5N1 HPAI outbreaks in Estuaire Province.
- FAO ECTAD continues supporting annual proficiency testing schemes of national and sub-national level laboratories for AI diagnosis in Central East, and West Africam countries through USAID funded GHSA programme.
- FAO ECTAD West and Central Africa Region, in collaboration with EMC-AH, supported the Government of Guinea to undertake a HPAI risk assessment mission to identify risk factors for introduction and spread.
- FAO participated in the 2nd virtual meeting of Regional Incident Coordination Group (ICG) for West Africa on HPAI organized by FAO ECTAD-WCA in collaboration with ECOWAS Regional Animal Health Centre (RAHC) held in March 2022.
- Taking stock of FAO-USAID partnership to control health threats in Kenya [link].
- Stopping Avian Influenza in Togo [link].
Figure 1. Number of countries reported HPAI since 01 October 2023 by subtype (left) and by region (right) as of 22 May 2024 (territory/area for sub/Antarctic zone)
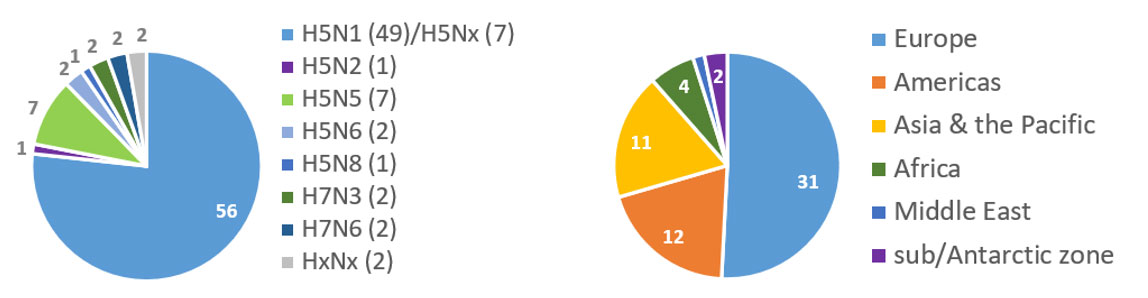
Source: WOAH WAHIS portal, government and publications.
Table 3. Epidemiological overview for avian influenza viruses viruses known to have caused zoonotic infections in the past 20 years
|
Subtype |
Epidemiological situation overview |
|---|---|
|
H5Nx Gs/GD* HPAI (1996) |
High pathogenicity avian influenza viruses within the Goose/Guangdong/1/96- lineage (Gs/GD) were first detected in geese in Guangdong Province, China in 1996. They have persisted, as high pathogenicity viruses, since then and have caused outbreaks in poultry across all regions globally other than Oceania. The initial viruses in this lineage were of the A(H5N1) subtype but other subtypes (including H5N2, H5N3 H5N5, H5N6, H5N8) have emerged, mainly in the past 10 years, as a result of reassortment with other avian influenza viruses. The common feature of these viruses is an HA gene related back to the original Gs/GD/96 virus. The HA gene of these viruses has evolved over the past 28 years, initially into 10 clades (clade 0 to 9) of which descendents of clade 2 viruses are the only ones that continue to circulate. Multiple 5th order clades persist such as the one that is currently dominant globally – clade 2.3.4.4b - whereas others have emerged and disappeared. Multiple genotypes carrying different combinations of the eight influenza A segmented genes have emerged presumably as a result of co-infection of birds with different avian inluenza viruses that also facilitated reassortment. Some important genotypes have been named unofficially (e.g. Z genotype in Hong Kong SAR, China in 2002, BB genotype derived from gulls in Europe in 2022 onwards). Of considerable significance has been reassortment with enzootic A(H9N2) viruses. Some of these Gs/GD viruses have produced severe zoonotic infections in humans, first identified in 1997 when an A(H5N1) clade 0 virus in the Gs/GD lineage in Hong Kong SAR, China caused disease outbreaks in poultry in farms and markets as well as severe disease in humans. In several cases there was some evidence of limited onward transmission in humans and this event raised concerns that it might be the beginning of a human influenza pandemic. Despite the successful efforts to eradicate this particular strain, other viruses within this lineage persisted and evolved in China, becoming more adept at infecting domestic ducks. By 2003 spread of these viruses via wild birds and live bird trade occurred across East and Southeast Asia, resulting in additional zoonotic infection in humans [link]. The important role of wild birds in the transmission of these viruses over long distances became apparent in 2005 when a Gs/GD virus (clade 2.2) spread, primarily via wild birds, across Eurasia, and parts of Africa from western China. Most high-income countries eliminated this virus from poultry, but it persisted in several low and middle- income countries. Viruses within the Gs/GD lineage continued to evolve and spread. Additional intercontinental waves of transmission have occurred with the two most significant being those in 2014 (clade 2.3.4.4c) and from 2016 onwards (clade 2.3.4.4b) that also resulted in spread of these viruses to North America (2014-15 and 2021-22), with the latest outbreak extending through central and South America and to sub-Antarctic islands. These waves involved multiple N subtypes. In 2022/2023, H5N1 2.3.4.4b caused extensive infection in coastal seabirds and mass die- offs of numerous ecologically important wild bird species. For an updated list of bird species affected with A(H5Nx) see HERE In 2024, H5N1 2.3.4.4b caused infection in goats (1 farm) and dairy cattle (51 farms) in the United States of America. See HERE. USDA shared the Whole Genome Sequences, see HERE. Among the other Gs/GD virus clades that remain endemic in specific areas are clade 2.3.2.1a H5N1 viruses that have persisted in South Asia since 2010 and rarely associated with disease in humans. Clade 2.3.2.1c/e viruses have been present in Indonesia since 2012 and related viruses are still circulating in Cambodia, Viet Nam and Lao People’s Democratic Republic. A novel reassortant influenza A(H5N1) virus has been detected in poultry in Cambodia (since 2023), Lao People's Democratic Republic and Viet Nam (since 2022) and was also detected in the human cases reported from Cambodia since late 2023 and Viet Nam in 2024. This virus contains the surface proteins from clade 2.3.2.1c that has circulated locally, but internal genes from a more recent clade 2.3.4.4b virus [link]. Of the ten recent human cases, five of which were fatal, recorded in Cambodia (4) and Viet Nam (1). For an updated list of confirmed human cases with A(H5N1) see HERE In addition, more than 80 human cases have been associated with clade 2.3.4.4b A(H5Nx/y) and 2.3.4.4h A(H5N6) viruses with most of these occurring in 2021 and 2022. Clade 2.3.4.4b A(H5N1) viruses have caused few human cases but have resulted in multiple mammalian cases including aquatic mammals. For an updated list of mammalian species affected with A(H5Nx) see HERE |
| Avian origin H3N8 LPAI |
An Influenza A(H3N8) virus lineage emerged in live bird markets in southern China in mid 2021 [link]. Since then, three human cases of Influenza A(H3N8) have been reported: In April 2022, the first human clinical case associated with this lineage was reported in Henan Province, China and was associated with severe disease. In May 2022, a 5-year-old boy was diagnosed with a mild influenza A(H3N8) infection in Changsha City, Hunan Province, China. On 27 March 2023, a third human case was reported from Guangdong Province, China in a 56-year-old female with underlying illneses who subsequently died. One of the A(H3N8) viruses isolated from a human was found to be transmissible by air in ferrets [link] but no evidence of sustained human transmission has been reported. |
|
H7N4 LPAI (2017) |
One human case in China with reported exposure to poultry. |
|
H7N9 LPAI (2013) & HPAI (2017) |
Reported only in China with over 1 000 human cases between 2013 and 2017 with a marked increase in 2017 compared to previous waves. Most human cases exposed in live bird markets. Nation-wide vaccination campaign in poultry since Sep 2017: Last reported human case in 2019 [link]. See FAO H7N9 situation update |
| H9N2 LPAI |
First human case reported in 1998. To date, about 100 influenza A(H9N2) human cases diagnosed worldwide, many of them were reported from China since December 2015. Most cases mild and involving children. Only two fatal cases reported [link] Endemic in multiple countries in Africa and Asia, a cause of significant production losses and mortalities in poultry production systems. Three major lineages and multiple genotypes. |
| H10Nx LPAI |
To date, three influenza A(H10N3) human infections have been reported globally [link]. In May 2021, the first case in Jiangsu Province, China [link], in September 2022, a second case in Zhejiang Province, China [link], in February 2024, the third case in Yunnan Province, China [link]. The first influenza A(H10N5) human infection was reported in Zhejiang Province, China [link]. |
Recommendations for affected countries and those at risk
FAO recommends intensified surveillance and awareness raising by national authorities.
General recommendations
It is important to report sick or dead birds – both wild birds and poultry - or wild mammals to local authorities (veterinary services, public health officials, community leaders etc.). These should be tested for avian
influenza viruses.
Recommendations to poultry producers
Farmers and poultry producers should step up their biosecurity measures in order to prevent potential virus introduction from wild birds or their faeces.
Recommendations to hunters
Hunting associations and wildlife authorities should be aware that avian influenza viruses might be present in waterfowl and some other species hunted and that hunting, handling and dressing of shot game carries the risk
of spreading avian influenza viruses to susceptible poultry.
Recommendations to national authorities
Increase surveillance efforts for the early detection of influenza viruses in poultry and dead wild species including certain mammals.
For full recommendations including non-avian species please see [link].
Important links
- The list of wild bird and mammalian species which have been infected with H5Nx HPAI [link]
- Joint FAO/WHO/WOAH preliminary assessment of recent influenza A(H5N1) viruses
- Scientific Task Force on Avian Influenza and Wild Birds H5N1 High pathogenicity avian influenza in wild birds - Unprecedented conservation impacts and urgent needs
- EFSA Avian influenza overview December–March 2023
- EFSA Bird Flu Rader and Migration Mapping Tool
- ECOWAS, FAO, USAID Report on the Regional Consultative Meeting on the Prevention and Control of Highly Pathogenic Avian Influenza (HPAI) in West Africa. Accra, Ghana, 30 May – 2 June 2023
- OFFLU website
- OFFLU Diagnostic guidance: HPAI dairy cattle
- OFFLU HPAI Detections in Livestock
- OFFLU statement on high pathogenicity avian influenza in dairy cows
- OFFLU Avian influenza matching (OFFLU-AIM) report
- OFFLU Continued expansion of high pathogenicity avian influenza H5 in wildlife in South America and incursion into the Antarctic region
- FAO Carcass management guidelines
- FAO guidelines for veterinary laboratory testing protocols for priority zoonotic diseases in Africa
- WHO EPI-WIN Webinar: Public health risk of avian influenza A(H5N1) detected recently in dairy cattle
- WHO influenza at the human animal interface monthly risk assessments
- WHO Public health resource pack for countries experiencing outbreaks of influenza in animals
- WOAH High Pathogenicity Avian Influenza in Cattle
- WOAH High Pathogenicity Avian Influenza (HPAI)- Situation Report
- FAO-WHO-WOAH-UNEP One Health Joint Plan of Action, 2022–2026
Updates on avian influenza infection in dairy cattle have been shared by USDA, US CDC and US FDA through the following sites:
FAO publication
- Approaches to controlling, preventing and eliminating H5N1 highly pathogenic avian influenza in endemic countries
- Preparing for highly pathogenic avian influenza. Revised edition
- Biosecurity guide for live poultry markets
- Biosecurity for highly pathogenic avian influenza
- Prevention and Control of Avian Flu in small scale poultry ::: A guide for veterinary paraprofessionnals in Vietnam, English, French, Indonesian
- Guiding principles for the design of avian influenza active surveillance in Asia: Designing active, comprehensive, risk-based avian influenza surveillance
- Highly-Pathogenic avian influenza and sustainable livelihoods (HPAI): Managing risk and developing options
- Questions & Answers handbook for good management practices and biosecurity in small and medium-scale poultry hatcheries
- Manual for the management of operations during an animal health emergency
- Good emergency management practice – The essentials: A guide to preparing for animal health emergencies, third edition
- The global strategy for prevention and control of h5n1 highly pathogenic avian influenza (2007)
- Report of the global programme for the prevention and control of HPAI. 2008, 2009, 2010, 2011
- Poultry in the 21st century avian influenza and beyond. – Risks and Challenges for Poultry Production
EMPRES Watch/Focus On
- Focus On, 2018: 2016–2018 Spread of H5N8 highly pathogenic avian influenza (HPAI) in sub-Saharan Africa: epidemiological and ecological observations
- Focus On, 2017: Highly Pathogenic H5 Avian Influenza in 2016 and 2017 – Observations and future perspectives
- Focus On, 2016: Updated situation of Highly Pathogenic Avian Influenza (H5N1) in Asia
- EMPRES Watch, 2016: H5N8 highly pathogenic avian influenza (HPAI) of clade 2.3.4.4 detected through surveillance of wild migratory birds in the Tyva Republic, the Russian Federation – potential for international spread
- EMPRES Watch, 2014: Avian influenza A(H5N6): the latest addition to emerging zoonotic avian influenza threats in East and Southeast Asia.
Online course/webinar
- Avian Influenza Preparedness Course was held in April/May 2023 [link].
- FAO RNE launched bilingual training course on Avian Influenza preparedness for NENA region on 17 January 2023 [link].
- FAO through its Virtual Learning Center developed an Avian Influenza Preparedness Course in 2022 [link].
- FAO, in collaboration with WOAH, organized a webinar on H5 HPAI occurrence and prevention in North Africa on 20 May 2021.
- WHO developed an online training course entitled “Strengthening collaboration between human and animal health sectors for improved health security”. The course covers the Tripartite Zoonosis Guide and associated tools in Module 2 [link].
- FAO held a webinar entitled Managing HPAI in wild birds on 10 February 2022 – recording part 1 & part 2.
- FAO Webinar: Pros and cons on AI vaccination, presented by Leslie Sims, Ian Brown, Sergei Khomenko, Sophie von Dobschüetz (2018) [link].
- FAO Webinar: Intercontinental spread of H5N8 highly pathogenic avian influenza – Analysis of the current situation and recommendations, for preventive action (2016) [link].
Risk Assessment
- Rapid qualitative risk assessment risk of h5 high pathogenicity avian influenza introduction in Central and South America and the Caribbean (2023)
- Microbiological risk assessment - Guidance for food (2021).
- Chinese-origin H7N9 avian influenza spread in poultry and human exposure - Qualitative risk assessment update. 2017, 2018, 2019.
- Highly pathogenic avian influenza (H5N1 HPAI) spread in the Middle East: risk assessment (2016).
- Addressing avian influenza A(H7N9) Qualitative risk assessment update. Jun 2013, Nov 2013, Jan 2014, Apr 2014, Sep 2015
- H5N8 HPAI in Uganda - Further spread in Uganda and neighbouring countries (2017)
Wild birds/mammals
- FAO Webinar: Managing large-scale highly pathogenic avian influenza outbreaks in wild birds. February 2022. part 1, part 2.
- The Scientific Task Force on Avian Influenza and Wild Birds issued a statement on 21 January 2022 relative to recent mass mortality in some wild bird populations in the United Kingdom and Israel [link].
- Scientific Task Force on Avian Influenza and Wild Birds statement on: H5N8 (and other subtypes) Highly Pathogenic Avian Influenza in poultry and wild birds Winter of 2020/2021 with focus on management of protected areas in the African Eurasian region. February 2021 [link].
- COVID-19 and animals: Information on risk mitigation measures for livestock and agricultural professionals.
- Wild bird highly pathogenic avian influenza surveillance.
Socio-economic / PPP
- Policy Analysis of HPAI Strategy Including Analysis of Collaboration and Partnership between Public and Private Sectors. Gathering Evidence for a Transitional Strategy.(GETS) for HPAI H5N1 Vaccination in Vietnam (2011).
- Family poultry communicatoins Vol.2 (2011).
- Highly pathogenic avian influenza: a rapid assessment of its socio-economic impact on vulnerable households in Egypt (2009)
- Compensation programs for the sanitary emergence of HPAI-H5N1 in Latina America and the Caribbean (2008).
- Smallholder poultry production – livelihoods, food security and sociocultural significance (2010).
- Economic and social impacts of avian influenza (2006).
Tripartite/Quadripartite plan, guide, tool
Next issue: 27 June 2024
Disclaimer
Information provided herein is current as of the date of issue. Information added or changed since the last Global AIV with Zoonotic Potential situation update appears in orange. Human cases are depicted in the geographic location of their report. For some cases, exposure may have occurred in one geographic location but reported in another. For cases with unknown onset date, reporting date was used instead. FAO compiles information drawn from multiple national (Ministries of Agriculture or Livestock, Ministries of Health, Provincial Government websites; Centers for Disease Prevention and Control [CDC]) and international sources (World Health Organization [WHO], World Organisation for Animal Health [WOAH]), as well as peer reviewed scientific articles. FAO makes every effort to ensure, but does not guarantee, accuracy, completeness or authenticity of the information. The designation employed and the presentation of material on the map do not imply the expression of any opinion whatsoever on the part of FAO concerning the legal or constitutional status of any country, territory or sea area, or concerning the delimitation of frontiers.
Contact
If interested in a previous issue please send an email to EMPRES-Animal Health specifying the intended use of the document.
