Mr. F. LUMARE
Aquaculture is expanding on a worldwide level and although the shrimp-culture production is comparatively low it tends to show a tremendous uprise trend. Table 1 compares the world aquaculture production in 1975 (Pillay, 1976) for yields obtained in the years 1979–80 and 1982–83 (Pedini, 1984). The data shows an increase of 688,1% for Crustaceans.
Penaeids, mainly, play a relevant role in Crustacean production at present.
The increase in prawn farming is due to the solutions found for many technological production problems, which may be summarized as follows:
The productive shrimp culture is developed on different systems according to the environmental availability, the socio-economic structure, the shrimp species available and their biological characteristics and, finally, the technological level of the country. Table 2 shows the main different shrimp culture systems in the world.
A very effective system of the shrimp-culture process is based on different steps : 1) sexual maturity in captivity; 2) reproduction or spawning; 3) larval and post-larval rearing; 4) pre-growing and 5) ongrowing to marketable size. This system has a closed cycle and it is completely controlled at each step; like this the system gives the assurance of success because it is able to give constant results in terms of number, quality and cost of the fry production. But in this case the cost productions are, generally speaking, higher than in the fourth system (see table 2) based on the fry collection in the wild (a shrimp culture system adopted in Central America). In this last condition the cost production of fry is very cheap and it may positively affect the whole shrimp-culture process. But this system may also be affected by a very problematic point which is the uncertainty of the fry collection. This is what happened in Ecuador, where, in March 1985, abnormally cold coastal water began to disrupt the spawning of Penaeus vannamei and P. stylirostris, greatly reducing the supply of wild-post-larvae and gravid females. As a result 40–70% of Ecuador's ponds were dry in early August 1985 because of the shortage of seedstock.
In the traditional shrimp-culture system, as seen in Japan (table 2), sexual matured females with well developped gonads (stage IV; table 3) are caught between April and September in spawning grounds, at a depth of about 10 m and then the gravid females with spermatophores (table.4) are transferred into spawning tanks. Here the spawners are stimulated to release eggs by increasing the temperature of the water (thermic stimulation) from natural conditions ( 18–21°C ) to the requested degree ( 24–26°C ).
In Italy, research on prawn reproduction and breeding began in 1970 with the Mediterranean species Penaeus kerathurus. But this Penaeid appeared unsuitable for culture due to the following causes :
P. kerathurus requires a long period ( about 17 months ) to reach a good marketable size ( 30g);
it has low resistance to cold and it dies at 5–6°C. Taking into consideration these points P. japonicus was introduced into Italy in 1979 for culture purposes because :
The introduction of Asiatic kuruma prawn raised the problem that as P.japonicus is not endemic along the Mediterranean coasts it required a reproduction technology settlement.
The successive experiments emphasized the reproductive biology of P. japonicus the role of unilateral eyestalk ablation ( tab.5,6,7,8,9,10, 11, and 12) the influence of the feeding quality ( table 13), the effects of the photoperiods and the importance of the environmental parameter, manipulation.
At present a technology has been perfected by which the sexual maturity of P. japonicus is not merely induced but controlled, so that the reproduction of the breeders can be synchronized ( tab.14). This means that the production of high numbers can be obtained on large scale. To give an idea of the potential of the controlled system, one single maturity controlled module ( 6m in diameter) contains an average of 300 females : for every reproduction cycle, about 40% of the female population is ready to spawn. From these, 85% spawn on average 35,000 eggs at a time, releasing 50,000 eggs per cycle ( 3–4 months), during which each female spawns 1.4 times. over the total reproductive period ( one spawning cycle for each month) 15,000,000 eggs are produced in one single module which means about 3.8 million eggs in the single module for each reproductive cycle. A commercial hatchery with at least 12 controlled tanks ( table 15) can produce 45.5 million eggs per cycle and about 182 million eggs over the total reproductive period (only four spawning cycles). By increasing the number of reproductive cycles, using the same broodstock, this figure can be doubled each season.
The methodology for the mass production of fry has also been designed with constant features and with high survival rates averaging about 60%, although in extreme cases the survival rate might range between 40 and 100%. The final sizes of the P22 post-larvae average 24 mg in weight and 17 mm in length. The final production density of the fry is currently more than 12 specimens ( P22 )/litre. Table 16 shows reproduction characteristics of Penaeids in main shrimp-culture areas.
One important aspect of the reproduction of P. japonicus in Italy
is the decrease in the hatch rate of eggs according to the progression of the
F generations (table 17).
In 1985 the hatch rate was 18% which is much lower than the 50% which can
generally be obtained from wild spawners. This low hatching rate might be
due to the reduction of the genetic variability when compared to the initial
stock of Asian Penaeid, according to research carried out on ongoing.
The population used in Italy in 1985 had already reached generation F7.
Despite this negative aspect, this can easily be overcame by important new
seed stock for broodstock reconstitution or by crossing old sub-stocks at high
numbers, the problem does not affect the large scale production of larvae
because of the plentifulness of eggs.
Another aspect regarding the production of larvae and post-larvae, (tab.18,19,20,21,22,23,24) must be pointed out. The thorough research carried out for about 10 years by specialized institutions on shrimp-pathology has made it possible to plan the disease resolution while defining specific prophylactic measures based on antibiotics and chemotherapy which prevent or reduce the action of the most common pathogens.
The larvae and post-larvae feeding is a basic aspect of the hatchery production. Table 25 shows general sequence scheme of foods supplied in Penaeid culture all over the world.
Table 26 gives the feeding sequence in the larvae and post-larvae rearing with the production costs in the Italian system which uses artificial food from P2–3 ( post-larvae 2–3 days old) to P22.
Table 15 shows a draft of hatchery facilities for the production of 1.4. million Penaeus japonicus fry while comparing the controlled sexual maturity system (A) and the stimulating system (B) only. This point is basic because it influences the cost production of P22 ( table 27) and then the effectiveness of releasing and culture purposes. Table 28 shows the main characteristics of larval and post-larval rearing and the pre-growing of penaeids in the world.
| 1975 | 79–80 | 82–83 | % INCREASE OVER THE LAST TWO YEARS | % INCREASE OVER 1975 | |
| FISH | 3,980,492 | 3,227,810 | 4,447,946 | 11.1 | 11.7 |
| MOLLUSCS | 1,051,341 | 1,908,016 | 1,957,570 | 0.9 | 86.2 |
| CRUSTACEAN | 15,663 | 71,224 | 123,445 | 20.1 | 688.1 |
| SEAEED | 1,054,793 | 2,206,484 | 2,393,782 | 2.8 | 126.9 |
TAB1. - Estimated world aquaculture production (t) in 1975 (Pillay,1976) and the years 1979–80 and 1982–83 (Pedini, 1984)
| P. japonicus; | Italy, France | Sexual maturity in captivity |  | Reproduction |  | Larval and post-larval culture |  | Growing Restocking | ||
| P. japonicus; | Brazil | Sexual maturity in semi-captivity |  | Reproduction |  | Larval and post-larval culture |  | Pre-growing |  | Growing |
| P. japonicus; | Japan | |||||||||
| P. monodon; | South-East of Asia | Reproduction |  | Larval and post-larval culture |  | Pre-growing |  | Growing Restocking | ||
| P. semisulcatus; | Kuwait | |||||||||
| P. vannamei; | Central | Pre-growing |  | Growing | ||||||
| P. stylirostris; | America | |||||||||
| P. monodon; | ||||||||||
| P. indicus; | South-East of Asia | Growing | ||||||||
| P. orientalis | ||||||||||
| Metapenaeus ensis; | ||||||||||
| M. monoceros; | ||||||||||
Table 2 - Scheme of main management steps of shrimp-culture in the world.

UNDEVELOPED (U) OR I STAGE |  |
DEVELOPING (D) OR II STAGE |  |
YELLOW (Y) OR III STAGE | 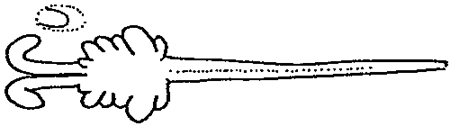 |
RIPE (R) OR IV STAGE |  |
SPENT (S) OR V STAGE | 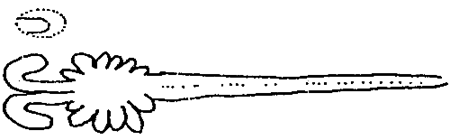 |
TABLE 3 - DIFFERENT STAGES OF OVARY DEVELOPMENT IN PENAEIDS. ON TOP, THE OVARY EXTENSION IN PENAEID.
 | 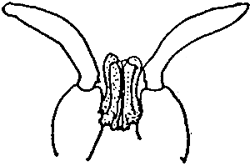 | ||
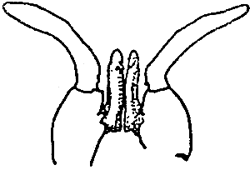
| PENAEUS KERATHURUS | |||
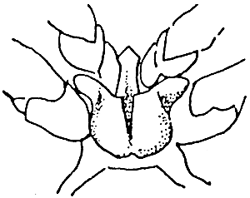
| Female. Thelycum between the bases of the 4th and 5th pairs of thoracic legs. | Male. Petasma on the first pairs of pleopods. | ||
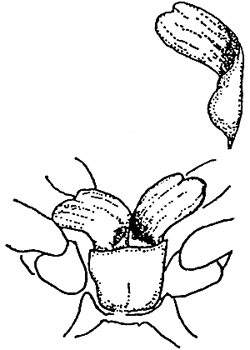 |  | ||
| PENAEUS JAPONICUS | |||
| Female. Thelycum with spermatophore and stoppers outside. Complete spermatophore on top. | Male. Petasma. | ||
Table 4 - Exterior genitals in Penaeids.
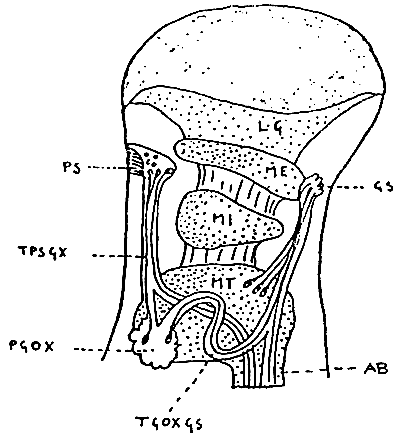
Tab. 5 - Eyestalk of Penaeid (Decapoda, Natantia) showing pars ganglionaris X-organ (PGOX), sensory pore X-organ (PS), sinus gland (GS), the X-organ-sinus gland tract (TGOXGS) and the X-organ-sensory pore tract (TPSGX). AB, axonal tract of brain neurosecretary cells. LG, ME, MI and MT represents respectively the lamina ganglionaris, Medulla externa, Medulla interna and Medulla terminalis, all parts of the optic lobe peduncle (from Carlisle 1953, reported in Adiyodi K.G. and R.G.Adiyodi 1970.).
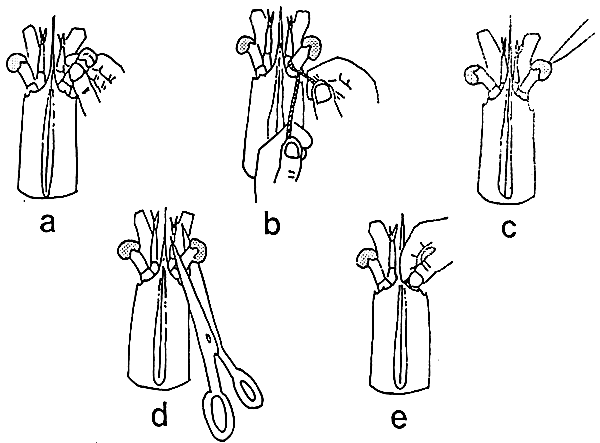
TAB. 6 - Methods of eyestalk removal in Penaeids: a) eyeball incision and squeezing; b) ligation or tying c) electrocautery or using a silver nitrate bar; d) cutting; e) pinching-crushing.
TABLE 7
Responses to unilateral eyestalk ablation in Penaeus kerathurus ( tank nos 1 and 2, Table 1). Only the females surviving the latency period are considered.
| Spawning sequence | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Females spawning1 | 24 | 16 | 10 | 7 | 3 | 1 | 1 | 1 | |
| Females spawning | |||||||||
fertile eggs | 18 | 14 | 8 | 5 | 2 | 1 | 1 | 1 | |
| Number of fertile | x | 83850 | 72330 | 80390 | 84100 | 80500 | 61000 | 69500 | 74500 |
eggs | e | 17640 | 10610 | 17930 | 18860 | - | - | - | - |
spawned2 | n | 10 | 12 | 8 | 4 | - | - | - | - |
| Number of eggs | x | 41060 | 45820 | 64440 | 69500 | 1000003 | 770003 | 48000 | 29000 |
hatched2 | e | 8424 | 5680 | 17880 | 16200 | 43000 | - | - | - |
| n | 10 | 12 | 8 | 4 | 2 | 1 | 1 | 1 |
1 Including females spawning only unfertile eggs.
2 x, mean; e; standard error; n, number of females examined.
3 Negative difference between number of fertile eggs spawned and of eggs hatched due to errors caused by counting method.
| Tank No. | Impregnated females (%) | Females with maturing gonads (II–IV stages;%) | Unilateral eyestalk ablation | Period(day) until first spawning | Number of spawning days in 19 days |
| 1 | 77.3 | 22.7 | No | - | 0 |
| 2 | 58.3 | 8.3 | No | - | 0 |
| 3 | 91.3 | 17.4 | Yes | 5 | 4 |
| 4 | 78.3 | 26.1 | Yes | 5 | 5 |
| 5 | 69.3 | 8.7 | Yes | 6 | 5 |
Table 8 - Responses to experimental conditions of Penaeus japonicus over a short period.
| Tank No. | Unilateral eyestalk ablation | Light intensity lux | Number of spawning day | Fertilized eggs rate(%) (2) | Total number of eggs | Mean no. of eggs spawning days | Spawning index (SI) (3) | Period(day) before first spawning | Period(day) before starting regular spawning | |||
| fertile (1) | unfertile | total | mean | range | ||||||||
| 1 | No | 1,250 | 3 | 0 | 3 | 72.00 | 36.4–97.8 | 11,500 | 3,833 | 0,08 | 202 | 202 |
| 2 | No | 2,200 | 15 | 6 | 21 | 77.39 | 50.0–100 | 61,000 | 2,904 | 0,50 | 39 | 143 |
| 3 | Yes | 1,500 | 61 | 53 | 114 | 42.45 | 5.0–100 | 231,500 | 2,030 | 2,83 | 5 | 31 |
| 4 | Yes | 2,000 | 61 | 29 | 90 | 59.28 | 5.0–100 | 224,000 | 2,488 | 2.57 | 5 | 5 |
| 5 | Yes | 3,500 | 69 | 52 | 121 | 61.14 | 10.0–100 | 465,000 | 3,883 | 4.40 | 6 | 6 |
Table 9 - Data on the spawning of Penaeus japonicus throughout the whole conditioning period (in 225 days)
(1) Spawning was considered fertile when fertilized and unfertilized eggs were found in the tanks.
(2) Fertilized egg rate (%) is considered on only fertile spawnings.
| Examination date | 26.2.80 | 28.3.80 | 18.5.80 | 25.6.80 | 12.8.80 | 7/8/9.10.80 | Mean impregnation rate (%) over whole conditioning period |
| Tank No. | |||||||
1 | 77.3 | 40.9 | 35.0 | 80.0 | 66.7 | 37.5 | 56.2 |
2 | 58.3 | 50.0 | 47.4 | 78.9 | 94.7 | 94.7 | 70.8 |
3 | 91.3 | 19.0 | 4.8 | 15.0 | 30.0 | 30.8 | 31.8 |
4 | 78.3 | 42.8 | 21.0 | 47.4 | 70.6 | 54.5 | 52.4 |
5 | 69.9 | 14.3 | 18.7 | 62.5 | 83.3 | 77.8 | 54.4 |
| Mean impregnation rate (%) on examination | 75.0 | 33.4 | 25.4 | 56.8 | 69.1 | 59.1 |
Table - 10 Impregnation rate (%) (females with spermatophores) during conditioning period.
| Tank No. | Unilateral eyestalk ablation | Number of moults | Intermouling period (I) | ||||
| females | males | females | males | ||||
1 | No | 114 | 92 | 33.15 | 40.90 | ||
2 | No | 116 | 68 | 47.54 | 34.77 | ||
3 | Yes | 133 | 103 | 38.78 | 38.07 | ||
4 | Yes | 117 | 75 | 47.81 | 32.92 | ||
5 | Yes | 97 | 89 | 46.09 | 46.52 | ||
Table 11 - Intermouling periods (I0 in Penaeus japonicus affected by different experimental conditions and for both males and females.
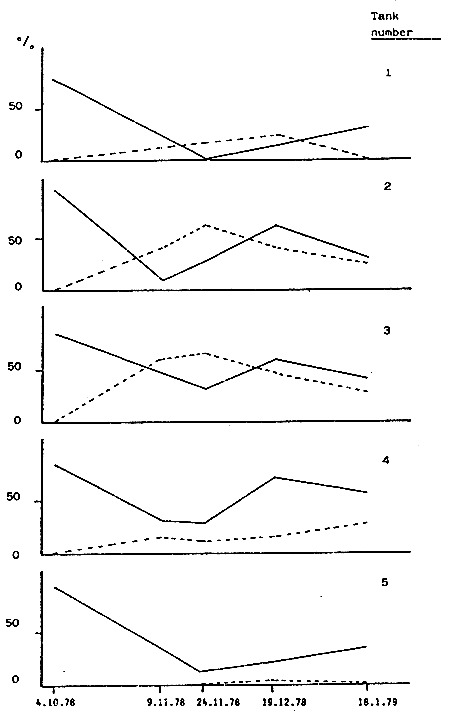
Tab. 12 - Penaeus kerathurus. Flactuations in the mating of ovaries rate of the breeder stocks. mating rate; = ovaries maturity rate.
| Tank number | Eyestalk ablation | Photoperiod Light source | Lux | Feeding (1) |
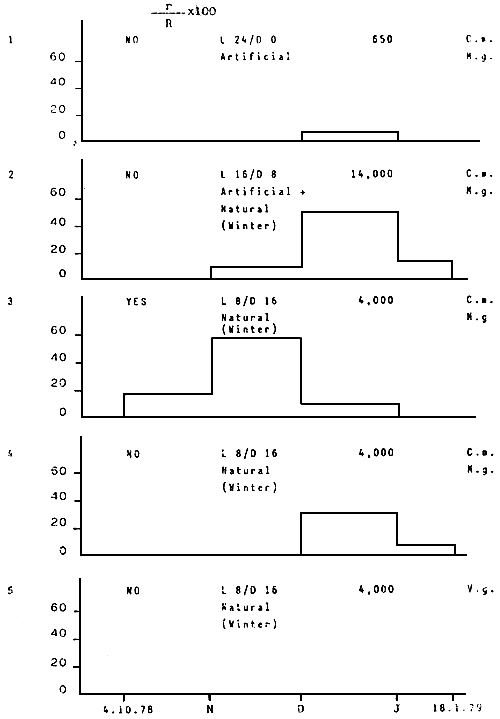
Tab. 13 - Penaeus kerathurus. Distribution of monthly fertile spawnings expressed as r/R.100; r = fertile spawnings, R = breeders;
C.m. = Carcinus moenas; M.g. = Mytilus galloprovincialis; V.g. = Venus gallina
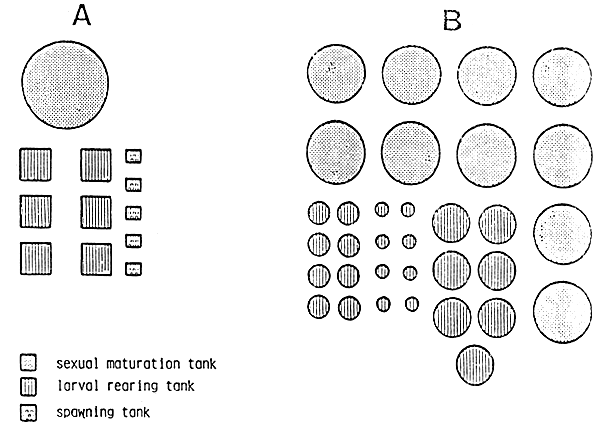
Tab. 14 - Draft of hatchery facilities for 1.4 million Penaeus Joponicus fry according to controlling sexual maturity and spawning system (A) and to stimulating system (B).
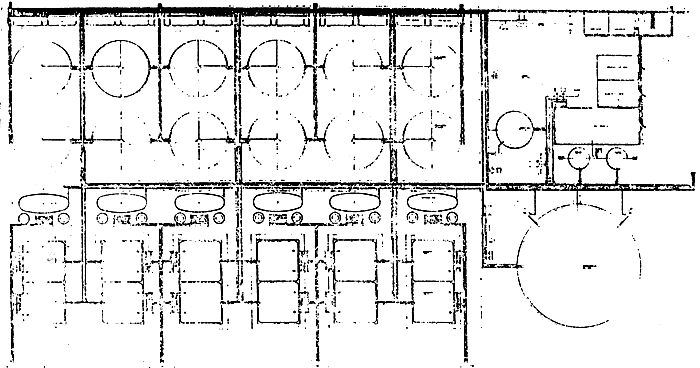
TABLE 15 - Commercial hatchery plant for the production of 20 million post-larvae P22 of Penaeus japonicus.
| SPECIES | AREA | TREATEMENT | TANK MATERIAL | SHAPE/SIZE (m) | VOLUME (m3) | SPAWNED FEMALE RATE CACH CYCLE (%) | SPAWNING NUMBER FOR EACH FEMALE MONTH | ECO NUMBERS RELEASED BY EACH FEMALE FOR SPAWNING | HATCHING RATE (%) |
| P. japonicus | Brazil | Environmental conditioning selected food | Cement | Circular(Æ 5) | 20–16 | 50–70 | 1 | 100,000 | 50 |
| Tetragonal | |||||||||
| (4×4×1) | |||||||||
| P. japonicus | France | Conditioning | PVC | Cylinder-conical | 0.5–2 | Not comparable data | 0.6 | 15,000 | 58 |
| Photoperiodism | Fiberglass | (0.7 Æ) | |||||||
| (1.82 Æ) | |||||||||
| P. japonicus | Italy | Environmental conditioning Unilateral eyestalk ablation | Cement | Tetragonal | 4–7.5 | 98 | 0.4 | 21,500 | 50-15 |
| (2×2×1) | |||||||||
| (2×2×1.8) | |||||||||
| P. japonicus | Japan | Thermic | Cement | Tetragonal | 50–200 | 30–50 | 1 | 400,000 | 50 |
| stimulation | Rectangular | ||||||||
| (10×10×2) | |||||||||
| (10×5×2) | |||||||||
| P. monodon | South-East Asia | Thermic Stimulation Environmental conditioning Unilateral eyestalk ablation | Fiberglass | Circular | 0.2–0.4 | 100 | 1 | 1,000,000 | 60-30 |
| Cement | (0.7 Æ ) | 100.000 | |||||||
| P. semisulcatus | Kuwait | Thermic stimulation | Cement | Tetragonal | 15–531 | 43–90 | 1 | 195,436 (a) | N.a.d. |
| Rectangular | |||||||||
| 20×8×9×1.6) | |||||||||
| 30×0×1.8) | |||||||||
| P. aztecus | North America | Thermic stimulation Unilateral eyestalk ablation and without Unilateral eyestalk ablation and not | Cement | Circular | 15-2 | 50 | 1 | 71,000 | 72.8 |
| P. ducranum | Fiberglass | (1.8–2 Æ) | 20 | 231,000 | |||||
| P. setiferus | Marine | Tetragonal | 309,000 | ||||||
| play-wood | (5×2×2) | ||||||||
| P. vannamei | Central and Southern America | Metal plastic | Circular | 0.5–3.5 | 1 | N.a.d. | 100,000 | 45–80 | |
| P. stylirostris | (0.6–3.7Æ) | 200,000 | |||||||
TAB. 16 Rproduction characteristics of Penaeids in main shrim-culture areas.(a) The figure refers to average nauplis numbers hatched for spawning. N;a.d. = Not available data
Year | Generation | Number of breeding pairs of previous generation | Number of hatchery produced larvae | Mean hatching rate (%) |
| 1980 | F2 | 2 | 2,000 | 50 |
| 1981 | F3 | 50 | 80,000 | 40 |
| 1982 | F4 | 100 | 500,000 | 32 |
| 1983 | F5 | 300 | 750,000 | 33 |
| 1984 | F6 | 300 | 1.250,000 | 20 |
| 1985 | F7 | 300 | 1.400,000 | 18 |
Table 17 - Data concerning lesina broodstock of Penaeus japonicus at various generations.
 |  |
| N1 | N2 |
 |  |
| N3 | N4 |
 |  |
| N5 | N6 |
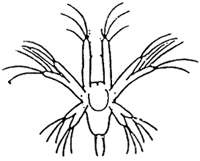
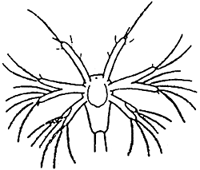
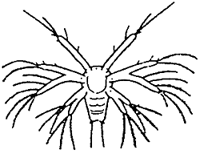
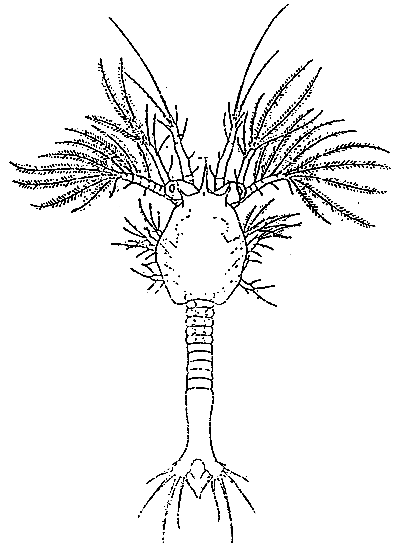
TAB. 18 - Naupliar stages of Penaeus japonicus.
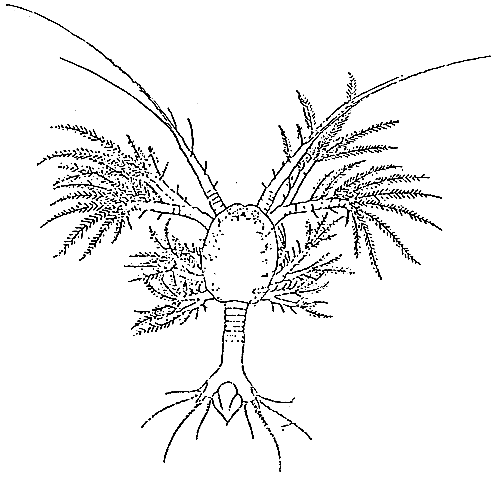
TAB. 19 - PROTÖZEA I
TAB. 20 - PROTOZÖEA II
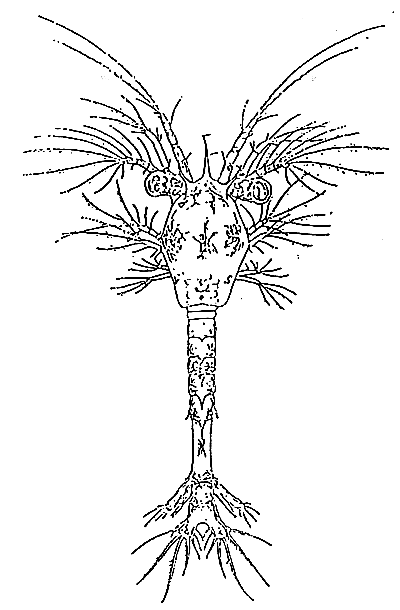
TAB. 21 - PROTOZÖEA III
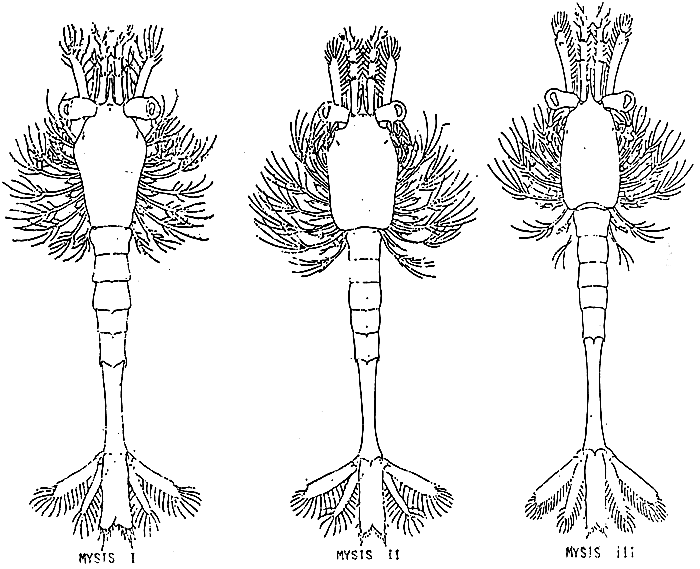
TAB. 22
TAB. 23 - MYSIS IV
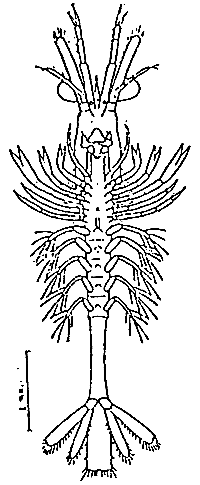
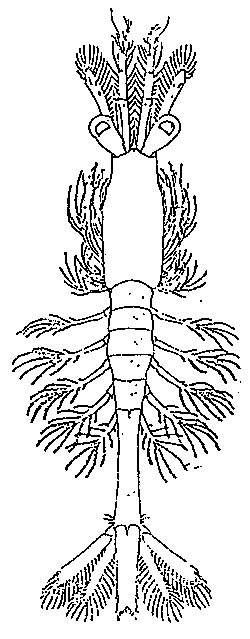
TAB. 24 - POST-LARVA I

TAB. 25- Sequence of foods supplied in the culture of larvae and post-larvae of Penaeids.
| × 1,000,000 di | P22: | |
| Fito | 800,000 lit | |
| Artemia cysts | 13 kg (226,000 lit/kg) | |
| Artificial diet | 60 kg (20,000 lit/kg) | |
| - | Feeding cost incidence for each post-larva P22 = 4.9 lit | |
| - | Management cost and other items incidence for each post-larva P22 = 19–28.9 lit | |
| - | Analysis costs referred to hatchery capacity of 20 millions P22 | |
TAB. 26 - Feeding sequence and production costs in the intensive culture of Penaeus japonicus post-larvae.
| SYSTEM A | SYSTEM B | |||
| Number of tanks employed | 1 | 10 | ||
| Conditioning | Tank volume (m3) | 17 | 12 | |
and | Number of sea water changes | 2 | 2 | |
| sexual | Total utilized sea water volumes | 34 | 240 | |
| maturation | Cost production incidence of sea water heating and recirculation on each P22 (Lit) | 10.38 | 74.68 | |
| Total tank volume (m3) | 33 | 70 | ||
| Egg incubation, larval and post-larval rearing | Number of sea water changes | 1 | 1 | |
| Cost production incidence of sea water heating and recirculation on each P22 (Lit) | 3.99 | 8.46 | ||
| General services of water and aeration | 0.54 | 3.78 | ||
| Phytoplancton | 0.84 | 0.84 | ||
| Artemia nauplia | 2.94 | 2.94 | ||
Feeding | Frozen Artemia | 11.75 | ||
| Artificial diet | 1.20 | |||
| Feeding cost incidence on each P22 (Lit) | 4.98 | 15.53 | ||
| Labour cost | 8.87 | 8.87 | ||
| General cost Incidence on each P22 (Lit) | 28.96 | 111.32 | ||
TAB. 27- Camparative cost incidence on Penaeus japonicus fry production between controlled sexual maturity and spawning system (A) and the stimulated one only (B). Healing cost figures in A system refers to 10° C average temperature in the sexualmaturity conditioning and hatchery production period (November-July) and then with a thermic expensive regime on the northern Adriatic Italian coasts; in the Southern Italian areas the figures correlated to heating costs may be cut by 50 per cent.
| LARVAL AND POST-LARVAL REARING | PER-GROWING | ||||||||||||||||||
| SPECIES | AREA | TANKS | DENSITY (SP/m2) | FINAL STAGE | FINAL SURVIVAL RATE | FINAL WEIGHT (mg) | REARING PERIOD (DAYS) | TANK MATERIAL | SURFACE (ha) | DENSITY SP/m2) | INITIAL STAGE | INITIAL WEIGHT (mg) | FINAL STAGE | FINAL WEIGHT | SURVIVAL RATE (I) | PRE-GROWING PERIOD (DAYS) | |||
| MATERIAL | VOLUME (m3) | START | END | START | END | ||||||||||||||
| P.japonicus | Brazil | Cement | 16–20 | N.a.d. | N.a.d. | P8–10 | 70 | 15 | 18–20 | Earth | 3.0 | 40–50 | 36 | P40–58 | 1–2 | 56 | 30–50 | ||
| 42 | |||||||||||||||||||
| P.japonicus | France | P.V.C. | 0.5–2 | 200,000 | 20,000 | P1–2 | 60–40 | 10–15 | 10–35 | ||||||||||
| Fiberglass | 8 | 250,000 | 50,000 | P5–10 | 47 | ||||||||||||||
| P20 | |||||||||||||||||||
| P.japonicus | Italy | Cement | 4–7.5 | 20,000 | 12,000 | P22–25 | 60 | 24 | 30 | ||||||||||
| P.japonicus | Japan | Cement | 50–200 | 10,000 | 5,000 | P20 | 20–63 | 10–20 | 30 | Sand | 0.4 | 90–175 | 80 | P35–65 | 1–2 | 17 | 15–45 | ||
| 20,000 | 10,000 | 350–600 | 400 | 54 | |||||||||||||||
| P.monodon | South-East Asia | Cement | 4–25 | 2,000 | P5 | 30–40 | N.a.d. | 15–30 | Cement | 2,000 | 1,800 | P45 | 0.2–1.5 | 6 | 30–35 | ||||
| 32 | 50,000 | 3,000 | P12 | Clay | 0.015 | 5,000 | 38 | ||||||||||||
| P20 | 10–30 | Sand | 0.2 | ||||||||||||||||
| Fiberglass | 0.01 | ||||||||||||||||||
| Marine | 0.02 | ||||||||||||||||||
| ply-wood | |||||||||||||||||||
| P.semisulcatus | Kuwait | Cement | 15 | 30,000 | 7,500 | P20 | 2.3 | 29.1 | 30–50 | ||||||||||
| 530 | 57,500 | P40 | 21.2 | ||||||||||||||||
| P.aztecus | North America | Cement | 1–2–20 | 100,000 | 21,000 | P1–2 | 38–50 | N.a.d. | 13 | ||||||||||
| P. ducrarum | Fiberglass | 500,000 | 86,000 | ||||||||||||||||
| P. setiferus | Marine | ||||||||||||||||||
| P. vannamei | ply-wood | ||||||||||||||||||
| P. vannamei | Central and Southern America | Fiberglass | 1.5–10–20 | 40,000 | 20,000 | P14 | 20–40 | N.a.d. | 10–24 | Clay-sand | 0.5–4 | 50 | 25–100 | P1–10 | 0.005–0.60 | P45–50 | 0.5–1 | 25–60 | 20–65 |
| P. stylirostris | 100,000 | 50,000 | P5–6 | 50 | 200 | P14 | |||||||||||||
TAB.28 - MAIN CHARACTERISTICS IN LARVAL AND POST-LARVAL REARING AND PRE-GROWING OF PENAEIDS. N.a.d. - No available data
Mr. G. LE MOULAC, Mr. C. DE LA POMELIE
1. INTRODUCTION
The control in the reproduction of certain Peneidae species is the outcome of en important effort made in research over the past ten years.
Many studies have been made on the control of maturation (CAUBERE, LAUBIER-BONNICHON). The MEREA team (PALAVAS Station) along with the AQUACOP team (Centre Océanologique IFREMER du Pacifique) have got under control the larvae rearing of many peneidae species especially:
- In temperate waters : P. japonicus
- In hot waters (Intertropical or subtropical zones) : P. monodon, P. stylirostris, P. vannamei, these two later species can be of interest for the hotter zones of the Mediterranean strip.
The hatchery phase for all these species covers three principal stages: Stocking-maturation, larvae spawning-rearing; and eventually first fattening.
The example of the P. japonicus is given here.
2. STOCKING
The production of a large number of post-larvae during the productive season is directly linked to the state of the broodstock at this precise moment.
A protocol was implemented so as to avoid mass mortalities linked with the pathogenic agent Fusarium solani and in these conditions, there is an excellent survival rate throughout the nine months of stocking and spownings take place the whole year round (Diagram 1 and 2).
3. MATURATION
Maturation takes place at 18° C all year with a natural photoperiod without epedonculation (Diagram 2)
The maturation rate obtained from controlled spawning is 50 %.
4. SPAWNING
Spawning is provoked by thermic shock. From 18° C, the animals at a maturation stage of 4.5 – 5, were submitted to 25° C.
Spawning took place around 3 days afterwards. The number of eggs collected per female was 200 000.
70 % of nauplii is then recovered.
5. LARVAE REARING
Larvae rearing is carried out under strict supervision. The control of the feeding sequence of the food quality and the pathological prevention, allows survivals rates of 70 % with final loads in rearing (110 to 150 P 3 per liter) (Diagram 3).
The speed of development of the larvae depends on the temperature.
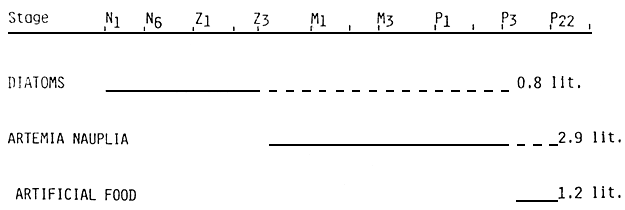
The feeding sequence can be seen in diagram 4.
The principal pathogenic agents remarked in larvae rearing are the “imperfect” fungus (Logidinium calinectes) and bacteria. The former is controlled with a fungicide (trifluraline) which is used as a means of prevention, and inhibits the sporulation of this fungus. This product is used continuously, from egg to P 1 stage. Bacteria is controlled with the use of an antibacterian (furazolidone) which is employed from Z 1 to M 1 stage.
6. FIRST FATTENING
The rearing is carried out for another 20 days, in clear water with a good water renewal.
The average survival rate, during this period, is 70 %.
The feeding sequence (Diagram 6) is established for a range of temperature of 23 – 25° C.
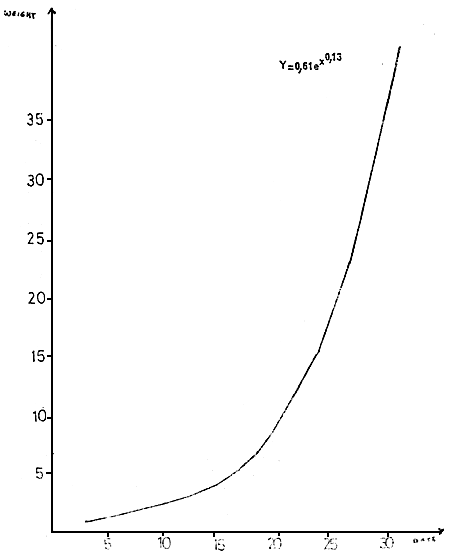
The growth is exponential (Diagram 5)
The post-larvae are sold from P 23 to P 25, between 12 and 15 mg.
There exist no important pathological problems. This period of rearing does not require any treatment.
7. TECHNICO-ECONOMICAL ANALYSIS
The control of all these parameters, the repetition of the results, have permitted the analysis of the production costs for each of the rearing stages in a hatchery (Table 1)
The definition of the production costs permits having at disposal an efficient programmation utensil for the productivity benefits. We discover that some are not as important as believed (efficiency, and not investment).
| Broodstock % | Nauplii | P 3 % | P 23 % | |
| Animals | 11 | 75 | 36 | 25,3 |
| Heating | 27,5 | 10 | 6,5 | 16,8 |
| Pumping | 2,7 | 0,6 | - | 1,8 |
| Air | 0,3 | - | - | 0,9 |
| Food | 5,1 | - | 41 | 50,3 |
| Manual labour | 53,2 | 15,5 | 16,4 | 5 |
| P.H F. H.T. | 120,9 | 1.62/1000 N | 6/1000 P 3 | 34/1000 P23 |
Table 1
8. TRANSFER
The reliability of the technique, along with the technico-economical analysis, shows that the production of shrimp, at post-larvae stage, is profitable. The transfer of this technique was carried out in GAEC “Les Poissons du Soleil” by MM. BALMA and CAUBERE in BALARUC-LES-BAINS. This hatchery which carried out pilot productions for two years (2 000 000 post-larvae) is now building a facility with a capacity of 10 000 post-larvae at first fattening stage. Another hatchery, the SCA Mari-Aude (Mr. LE BITOUX) should start operating this Spring. Finally, in the PALAVAS Station a training course was held for hatchery shrimp technicians and this permitted to answer the demand of transfer for 5 other hatcheries for molluscs or fish.
If the production of post-larvae has developed, it is due to the fact that the results obtained are very encouraging for fattening in the Mediterranean and on the Atlantic coast.
Outside France, France Aquaculture, a sub-company of IFREMER has greatly contributed to the development of shrimp rearing in intertropical zones, while employing three major species: P. monodon, P. stylirostris, P. vannamei.. The two latter species and moreover P. stylirostris could be suitable for the hotter zones of the Mediterranean.
9. HATCHERY DEVELOPMENT IN FRANCE
The production of shrimp should enhance production in hatcheries which already exist.
In France, two hatchery productions exist which are marketable ; fish fry (sea-bass and gilthead sea-bream) and mollusc spat (clam and oyster)
The production season of shrimp post-larvae follows the fry production season of fish. The shrimp production is feasible in these facilities. The only additional facility required is the room for the production of unicellular algae.
In a hatchery that produces mollusc spat, algae is no problem as this production is the food basis for fry.
Another possibility lies in the creation of a monospecific hatchery for shrimp which could integrate a fattening farm.
The cost price of shrimp post-larvae should be around the production cost, as is remarked in hatcheries for mollusc and fish, the depreciation and the expenses have already been taken into account in the cost price of fry and spat.
In the case of a hatchery which integrates a fattening farm, the purchase price of post-larvae is not the same as the commercial price found on the market of post-larvae (0,15 Frs) : the price of the post-larvae will be the cost price without manual labour, as the manual labour comprehends all production (post-larvae and fattening)
Production cost : 0.03 Frs/ P 25
Monospecific hatchery cost price : 0,10 to 0,14 Frs/ P 25.
Figure 1: BROODSTOCK'S SURVIVAL RATE
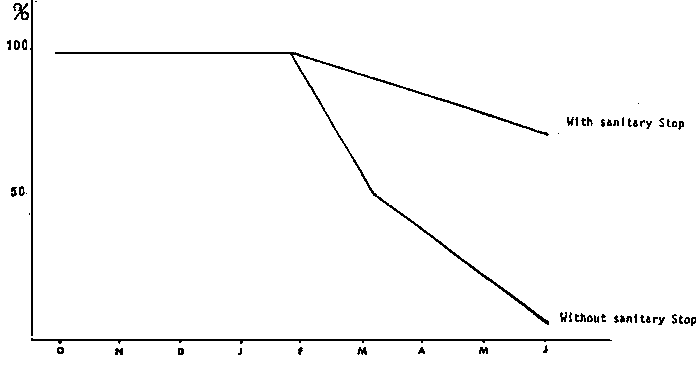
Figure 2: BROADSTOCK SPAWNING
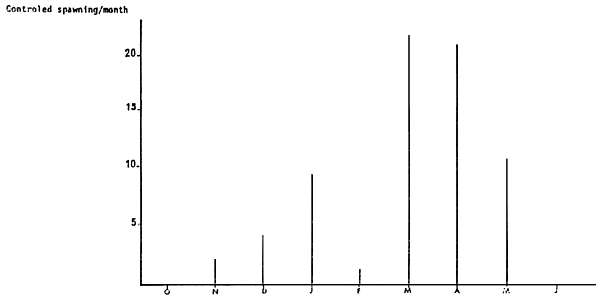
Figure 3 : REARING LARVAE SURVIVAL RATE
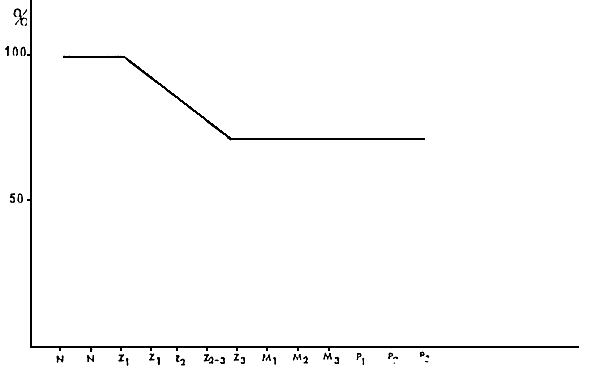
Figure 4 : REARING LARVAE FEEDING
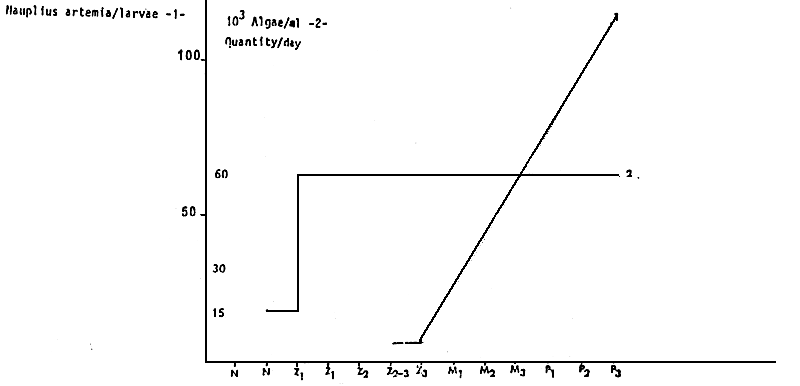
Figure 5 : PREGRANING FEEDING FOR 100 000
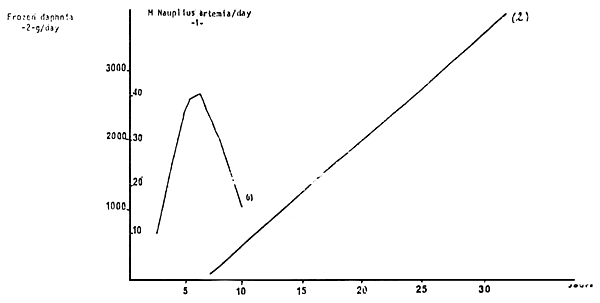
Figure 6 : GROWING CURVE OF POST-LARVAE